Abstract
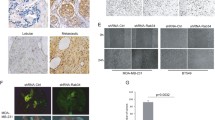
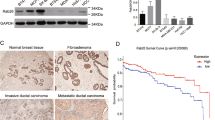
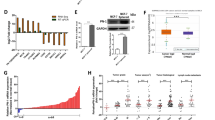
The endocrine system is closely related to the development of the breast cancer. Many studies have shown that FGF1 (Fibroblast growth factor-1) is involved the occurrence and development of the breast cancer. But up to now, the cellular behavior and characteristics of FGF1 in breast cancer have not been fully revealed. In the current study, breast cancer cell was used as an in vitro cell model to investigate FGF’s cell property. The results showed that FGF1 internalized into cells in a time-dependent manner. Further study indicated that both clathrin-mediated and caveolin-mediated endocytic pathway are involved in the internalization of FGF/FGFR (Fibroblast growth factor receptor), and both clathrin-mediated endocytosis and caveolin-mediated endocytosis are involved in the process of FGF1’s nuclear localization. Further study showed that Rab5 also plays an important role in the process of nuclear localization of FGF-1. In addition, we found that FGF1 and FGFR transported to the cell nuclei of breast cancer. Further experimental results indicated that the nuclear-localized FGF1 and/or FGFR is closely associated to cell proliferation of breast cancer cell. Taken together, the current work lays the foundation for exploring the relationship between nuclear-localized FGF1/FGFR and the occurrence and development of breast cancer.






Similar content being viewed by others
Data availability
All data included in this study are available upon request by contact with the corresponding author.
References
Brady, N. J., Chuntova, P., Bade, L. K., & Schwertfeger, K. L. (2013). The fgf/fgf receptor axis as a therapeutic target in breast cancer. Expert Review of Endocrinology & Metabolism, 8(4), 391–402. https://doi.org/10.1586/17446651.2013.811910.
Alberto, O., Hortobagyi, G. N., & Esteva, F. J. (2006). Concomitant versus sequential chemotherapy in the treatment of early-stage and metastatic breast cancer. Clinical Breast Cancer, 6(6), 495–504. https://doi.org/10.3816/CBC.2006.n.002.
Li, L., Wang, Q., & Lv, X. (2015). Expression and Localization of Estrogen Receptor in Human Breast Cancer and Its Clinical Significance. Cell Biochem Biophys, 71, 63–68. https://doi.org/10.1007/s12013-014-0163-6.
Mohammed Y. (2021). Comparative study of logistic regression and artificial neural networks on predicting breast cancer cytology. Indonesian Journal of Electrical Engineering and Computer Science. 21(2). https://doi.org/10.11591/ijeecs.v21.i2.pp1113-1120.
Presta, M., Patrizia Dell’Era, Mitola, S., Moroni, E., Ronca, R., & Rusnati, M. (2005). Fibroblast growth factor/fibroblast growth factor receptor system in angiogenesis. Cytokine & Growth Factor Reviews, 16(2), 159–78. https://doi.org/10.1016/j.cytogfr.2005.01.004.
Willems, W. A., Vanaudenaerde, B. M., & Vos, R. (2013). Azithromycin Attenuates Fibroblast Growth Factors Induced Vascular Endothelial Growth Factor Via p38MAPK Signaling in Human Airway Smooth Muscle Cells. Cell Biochem Biophys, 67, 331–339. https://doi.org/10.1007/s12013-011-9331-0.
Chioni, A. M., & Grose, R. (2012). FGFR1 cleavage and nuclear translocation regulates breast cancer cell behavior. Journal of Cell Biology, 197(6), 801–817. https://doi.org/10.1083/jcb.201108077.
Owczarek, S., Kiryushko, D., Larsen, M. H., Kastrup, J. S., Gajhede, M., & Sandi, C. (2010). Neuroplastin-55 binds to and signals through the fibroblast growth factor receptor. Faseb Journal Official Publication of the Federation of American Societies for Experimental Biology, 24(4), 1139–50. https://doi.org/10.1096/fj.09-140509.
Coleman, S. J., Chioni, A. M., Ghallab, M., Anderson, R. K., Lemoine, N. R., & Grose, R. P. (2013). Nuclear translocation of FGFR1 and FGF2 in pancreatic stellate cells facilitates pancreatic cancer cell invasion. Pancreatology, 13(1), e10–e11. https://doi.org/10.1016/j.pan.2012.12.032.
Miller, S. L., Demaria, J. E., Freier, D. O., Riegel, A. M., & Clevenger, C. V. (2005). Novel association of vav2 and nek3 modulates signaling through the human prolactin receptor. Molecular Endocrinology, 19(4), 939–49. https://doi.org/10.1210/me.2004-0443.
Marie, P. J., Miraoui, H., & Sévère, N. (2012). FGF/FGFR signaling in bone formation: progress and perspectives. Growth Factors, 30(2), 117–23. https://doi.org/10.3109/08977194.2012.656761.
Albiges, L., Quidville, V., Valent, A., Mathieu, M., & Andre, F. (2010). FGFR1 and FGF coamplification in breast cancer. Cancer Research, 69(24 Supplement), 4170–4170. https://doi.org/10.1158/0008-5472.SABCS-09-4170.
Bohrer, L. R., Chuntova, P., Bade, L. K., Beadnell, T. C., Leon, R. P., & Brady, N. J. (2014). Activation of the fgfr-stat3 pathway in breast cancer cells induces a hyaluronan-rich microenvironment that licenses tumor formation. Cancer Research, 74(1), 374–86. https://doi.org/10.1158/0008-5472.CAN-13-2469.
Masaru K., Hitoshi N. (2014). FGF receptors: cancer biology and therapeutics. Medicinal Research Reviews. https://doi.org/10.1002/med.21288.
Anita, L., Bane, D., & Pinnaduwage, S. (2009). Expression profiling of familial breast cancers demonstrates higher expression of fgfr2 in brca2-associated tumors. Breast Cancer Research & Treatment, 117(1), 183–91. https://doi.org/10.1007/s10549-008-0087-1.
Wesche, J., Małecki, J., Wiedłocha, A., Ehsani, M., Marcinkowska, E., Nilsen, T., & Olsnes, S. (2005). Two nuclear localization signals required for transport from the cytosol to the nucleus of externally added FGF-1 translocated into cells. Biochemistry, 44(16), 6071–80. https://doi.org/10.1021/bi047403m.
Bryant, D. M., & Stow, J. L. (2005). Nuclear translocation of cell‐surface receptors: lessons from fibroblast growth factor. Traffic, 6(10), 947–54. https://doi.org/10.1111/j.1600-0854.2005.00332.x.
Guo B., Lv Z., Cui C. (2021). IGF-1R Transported to the Cell Nuclei to Regulate the Proliferation of Breast Cancer Cells. Cell Biochem Biophys. 1–13. https://doi.org/10.1007/s12013-021-00989-8
Santolla, M. F., & Maggiolini, M. (2020). The FGF/FGFR system in breast cancer: oncogenic features and therapeutic perspectives. Cancers, 12(10), 3029 https://doi.org/10.3390/cancers12103029.
Acknowledgements
The author thanks Dr. Y.W. for the necessary information.
Author information
Authors and Affiliations
Contributions
Y.G. and R.-de.G., performed the experiments; Y.G. drafted the paper. J.Y. and Y.W. edited the paper. Y.G. and R.-de.G. conceived of the study, and participated in its design and coordination.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
About this article
Cite this article
Gao, Y., Wang, Y., Yu, J. et al. FGF Exhibits an Important Biological Role on Regulating Cell Proliferation of Breast Cancer When it Transports Into The Cell Nuclei. Cell Biochem Biophys 80, 311–320 (2022). https://doi.org/10.1007/s12013-021-01044-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12013-021-01044-2