Abstract
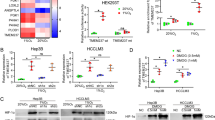
This study aimed to investigate the underlying molecular pathogenic mechanism of Sec62 in hepatocellular carcinoma (HCC). Microarray analysis was conducted to profile the global gene expression in the HCC cell line Huh7 cells transfected with Sec62high vs. NC and Sec62low vs. NC. Ingenuity pathway analysis and gene set enrichment analysis were used to perform Sec62-related signaling pathway analysis from screened differentially expressed genes (DEGs). A protein-protein interaction network was constructed. Experimental validation of the expression of key DEGs was conducted. Hypoxia-induced tube formation was undertaken to investigate the role of Sec62 in angiogenesis. A total of 74 intersected DEGs were identified from Huh7 cells with Sec62high vs. NC and Sec62low vs. NC. Among them, 65 DEGs were correlated with the expression of Sec62. The P53 signaling pathway was found to be enriched in Huh7 cells with Sec62high vs. NC, while the acute phase response signaling pathway was enriched in Huh7 cells with Sec62low vs. NC. DEGs, such as serine protease inhibitor E (SERPINE) and tumor necrosis factor receptor superfamily, member 11B (TNFRSF11B), were not only identified as the lead genes of these enriched pathways, but were also found to be closely related to Sec62. Moreover, knockdown of Sec62 decreased the expression of SERPINE1 (plasminogen activator inhibitor type 1 (PAI-1)) and TNFRSF11B, whereas overexpression of Sec62 had the opposite effects. In addition, knockdown of Sec62 inhibited hypoxia-induced tube formation via PAI-1. Sec62 promoted pro-angiogenesis of HCC under hypoxia by regulating PAI-1, and it may be a crucial angiogenic switch in HCC.



Similar content being viewed by others
Data availability
The datasets used during the present study are available from the corresponding author upon reasonable request.
Abbreviations
- hepatocellular carcinoma:
-
HCC
- ingenuity pathway analysis:
-
IPA
- gene set enrichment analysis:
-
GSEA
- differentially expressed genes:
-
DEGs
- protein-protein interaction:
-
PPI
- serine protease inhibitor E:
-
SERPINE
- tumor necrosis factor receptor superfamily member 11B:
-
TNFRSF11B
- unfolded protein response:
-
UPR
- endoplasmic reticulum:
-
ER
- fold-change:
-
FC
- polymerase chain reaction:
-
PCR
- quantitative PCR:
-
qPCR
- negative control:
-
NC
- standardized enrichment score:
-
NES
- plasminogen activator inhibitor type 1:
-
PAI-1
- glyceraldehyde-3-phosphate dehydrogenase:
-
GAPDH
References
Bray, F., Ferlay, J., Soerjomataram, I., Siegel, R. L., Torre, L. A. & & Jemal, A. (2018). GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA: A Cancer Journal of Clinicians, 68, 394–424. https://doi.org/10.3322/caac.21492.
Matsuura, Y., Wada, H., Eguchi, H., Gotoh, K., Kobayashi, S., Kinoshita, M., Kubo, M., Hayashi, K., Iwagami, Y., Yamada, D., Asaoka, T., Noda, T., Kawamoto, K., Takeda, Y., Tanemura, M., Umeshita, K., Doki, Y., & Mori, M. (2019). Exosomal miR-155 derived from hepatocellular carcinoma cells under hypoxia promotes angiogenesis in endothelial cells. Digestive Diseases and Sciences, 64, 792–802. https://doi.org/10.1007/s10620-018-5380-1.
Moraes, A. O., do Nascimento, E. A., Zubiolo, T. F. M., de Paula, M. F. M., Brito, A. F. B., Petta, B. F. V., Perini, G. M., & Martins, D. R. (2019). Transcatheter arterial chemoembolization of hepatocellular carcinoma in a patient with celiac trunk occlusion: a therapeutic challenge. Journal of Vascular Surgery, 18, e20180090 https://doi.org/10.1590/1677-5449.180090.
Liu, J., Xie, S., Duan, X., Chen, J., Zhou, X., Li, Y., Li, Z., & Han, X. (2020). Assessment of efficacy and safety of the transcatheter arterial chemoembolization with or without apatinib in the treatment of large hepatocellular carcinoma. Cancer Chemotherapy and Pharmacology, 85, 69–76. https://doi.org/10.1007/s00280-019-04004-z.
Morse, M. A., Sun, W., Kim, R., He, A. R., Abada, P. B., Mynderse, M., & Finn, R. S. (2019). The role of angiogenesis in hepatocellular carcinoma. Clinical Cancer Research, 25, 912–920. https://doi.org/10.1158/1078-0432.CCR-18-1254.
Li, X., Zhang, K., & Li, Z. (2011). Unfolded protein response in cancer: the Physician’s perspective. Journal of Hematology & Oncology, 4, 8.
Sano, R., & Reed, J. C. (2013). ER stress-induced cell death mechanisms. Biochimica Et Biophysica Acta Molecular Cell Research, 1833, 3460–3470.
Chipurupalli, S., Kannan, E., Tergaonkar, V., D’Andrea, R. & & Robinson, N. (2019). Hypoxia induced ER stress response as an adaptive mechanism in cancer. International Journal of Molecular Sciences, 20, 749.
Tyedmers, J., Lerner, M., Bies, C., Dudek, J., Skowronek, M.H., Haas, I.G., Heim, N., Nastainczyk, W., Volkmer, J. & Zimmermann, R. (2000). Homologs of the yeast Sec complex subunits Sec62p and Sec63p are abundant proteins in dog pancreas microsomes. Proceedings of the National Academy of Sciences, 97, 7214–7219.
Dudek, J., Greiner, M., Müller, A., Hendershot, L.M., Kopsch, K., Nastainczyk, W. & Zimmermann, R. (2005). ERj1p has a basic role in protein biogenesis at the endoplasmic reticulum. Nature Structural & Molecular Biology, 12, 1008–1014.
Muller, L., De Escauriaza, M. D., Lajoie, P., Theis, M., Jung, M., Muller, A., Burgard, C., Greiner, M., Snapp, E. L., & Dudek, J. (2010). Evolutionary gain of function for the ER membrane protein Sec62 from yeast to humans. Molecular Biology of the Cell, 21, 691.
Hu, S., Ye, H., Cui, Y., & Jiang, L. W. (2020). AtSec62 is critical for plant development and is involved in ER-phagy in Arabidopsis thaliana. Journal Of Integrative Plant Biology, 62, 181–200. https://doi.org/10.1111/jipb.12872.
Linxweiler, M., Schorr, S., Schauble, N., Jung, M., Linxweiler, J., Langer, F., Schafers, H. J., Cavalie, A., Zimmermann, R., & Greiner, M. (2013). Targeting cell migration and the endoplasmic reticulum stress response with calmodulin antagonists: a clinically tested small molecule phenocopy of SEC62 gene silencing in human tumor cells. BMC Cancer, 13, 574. https://doi.org/10.1186/1471-2407-13-574[pii].
Jung, V., Kindich, R., Kamradt, J., Jung, M., Muller, M., Schulz, W. A., Engers, R., Unteregger, G., Stockle, M., Zimmermann, R., & Wullich, B. (2006). Genomic and expression analysis of the 3q25-q26 amplification unit reveals TLOC1/SEC62 as a probable target gene in prostate cancer. Molecular Cancer Research, 4, 169–76. https://doi.org/10.1158/1541-7786.MCR-05-0165[pii]. 4/3/169.
Linxweiler, M., Linxweiler, J., Barth, M., Benedix, J., Jung, V., Kim, Y. J., Bohle, R. M., Zimmermann, R., & Greiner, M. (2012). Sec62 bridges the gap from 3q amplification to molecular cell biology in non-small cell lung cancer. American Journal of Pathology, 180, 473–83. https://doi.org/10.1016/j.ajpath.2011.10.039S0002-9440(11)01062-5[pii].
Greiner, M., Kreutzer, B., Jung, V., Grobholz, R., Hasenfus, A., Stohr, R. F., Tornillo, L., Dudek, J., Stockle, M., Unteregger, G., Kamradt, J., Wullich, B., & Zimmermann, R. (2011). Silencing of the SEC62 gene inhibits migratory and invasive potential of various tumor cells. International Journal of Cancer, 128, 2284–95. https://doi.org/10.1002/ijc.25580.
Weng, L, Du, J, Zhou, Q, Cheng, B, Li, J, Zhang, D & Ling, C (2012) Identification of cyclin B1 and Sec62 as biomarkers for recurrence in patients with HBV-related hepatocellular carcinoma after surgical resection. 11, 39.
Du, J., Zhao, Z., Zhao, H., Liu, D., & Ling, C. (2019). Sec62 promotes early recurrence of hepatocellular carcinoma through activating integrinα/CAV1 signalling. Oncogenesis, 8, 74.
Geis, T., Doring, C., Popp, R., Grossmann, N., Fleming, I., Hansmann, M. L., Dehne, N., & Brune, B. (2015). HIF-2alpha-dependent PAI-1 induction contributes to angiogenesis in hepatocellular carcinoma. Experimental Cell Research, 331, 46–57. https://doi.org/10.1016/j.yexcr.2014.11.018.
Samarakoon, R., & Higgins, P. J. (2002). MEK/ERK pathway mediates cell-shape-dependent plasminogen activator inhibitor type 1 gene expression upon drug-induced disruption of the microfilament and microtubule networks. Journal of Cell Science, 115, 3093–3103.
Samarakoon, R., Overstreet, J. M., Higgins, S. P., & Higgins, P. J. (2012). TGF-β1→SMAD/p53/USF2→PAI-1 transcriptional axis in ureteral obstruction-induced renal fibrosis. Cell & Tissue Research, 347, 117–128.
Higgins, S. P., Samarakoon, R., Higgins, C. E., Freytag, J., Wilkinsport, C. E., & Higgins, P. J. (2009). TGF-β1 -induced expression of the anti-apoptotic PAI-1 protein requires EGFR signaling. Cell Commun Insights, 2, 1–11.
Hideki, K., Xuan, L., Kunio, T., Toshiharu, O., Kazuko, K., Daisuke, M., Naoki, T. & Haruyoshi, Y. (2009). Dexamethasone enhances basal and TNF-α-stimulated production of PAI-1 via the glucocorticoid receptor regardless of 11β-hydroxysteroid dehydrogenase 2 status in human proximal renal tubular cells. Nephrology Dialysis Transplantation, 24, 1759–1765.
Miyagawa, R., Asakura, T., Nakamura, T., Okada, H., Iwaki, S., Sobel, B. E., & Fujii, S. (2010). Increased expression of plasminogen activator inhibitor type-1 (PAI-1) in HEPG2 cells induced by insulin mediated by the 3’-untranslated region of the PAI-1 gene and its pharmacologic implications. Coronary Artery Disease, 21, 144–150.
Mccarty, M. F. (2005). De novo synthesis of diacylglycerol in endothelium may mediate the association between PAI-1 and the insulin resistance syndrome. Medical Hypotheses, 64, 388–393.
Ahn, Y. T., Chua, M. S., Whitlock, J. P., Shin, Y. C., Song, W. H., Kim, Y., Eom, C. Y., & An, W. G. (2010). Rodent-specific hypoxia response elements enhance PAI-1 expression through HIF-1 or HIF-2 in mouse hepatoma cells. International Journal of Oncology, 37, 1627–1638.
Sprague, L., Tomaso, H., Mengele, K., Schilling, D., Bayer, C., Stadler, P., Schmitt, M., & Molls, M. (2007). Effects of hypoxia and reoxygenation on the expression levels of the urokinase-type plasminogen activator, its inhibitor plasminogen activator inhibitor type-1 and the urokinase-type plasminogen activator receptor in human head and neck tumour cells. Oncology Reports, 17, 1259–1268.
Robinson, M. D., & Oshlack, A. (2010). A scaling normalization method for differential expression analysis of RNA-seq data. Genome biology, 11, 1–9.
Du, J., Zhao, Z., Zhao, H., Liu, D., Liu, H., Chen, J., Cheng, B., Zhai, X., Yin, Z., Zhang, Y., & Ling, C. (2019). Sec62 promotes early recurrence of hepatocellular carcinoma through activating integrinalpha/CAV1 signalling. Oncogenesis, 8, 74. https://doi.org/10.1038/s41389-019-0183-6[pii].
Fang, Q., Yao, S., Luo, G. & & Zhang, X. (2018). Identification of differentially expressed genes in human breast cancer cells induced by 4-hydroxyltamoxifen and elucidation of their pathophysiological relevance and mechanisms. Oncotarget, 9, 2475–2501.
KjøLler, L., Kanse, S. M., Kirkegaard, T., Rodenburg, K. W., RNne, E., Goodman, S. L., Preissner, K. T., Ossowski, L., & Andreasen, P. A. (1997). Plasminogen activator inhibitor-1 represses integrin- and vitronectin-mediated cell migration independently of its function as an inhibitor of plasminogen activation. Experimental Cell Research, 232, 420–9.
Szklarczyk, D., Franceschini, A., Wyder, S., Forslund, K., Heller, D., Huerta-Cepas, J., Simonovic, M., Roth, A., Santos, A., & Tsafou, K. P. (2015). STRING v10: protein–protein interaction networks, integrated over the tree of life. Nucleic Acids Research, 43, D447–D452.
Shannon, P., Markiel, A., Ozier, O., Baliga, N. S., Wang, J. T., Ramage, D., Amin, N., Schwikowski, B., & Ideker, T. (2003). Cytoscape: a software environment for integrated models of biomolecular interaction networks. Genome Research, 13, 2498–2504.
Bandettini, W. P., Kellman, P., Mancini, C., Booker, O. J., Vasu, S., Leung, S. W., Wilson, J. R., Shanbhag, S. M., Chen, M. Y., & Arai, A. E. (2012). MultiContrast Delayed Enhancement (MCODE) improves detection of subendocardial myocardial infarction by late gadolinium enhancement cardiovascular magnetic resonance: a clinical validation study. Journal of Cardiovascular Magnetic Resonance Official Journal of the Society for Cardiovascular Magnetic Resonance, 14, 83–83.
Lin, Z., Niu, Y., Wan, A., Chen, D., Liang, H., Chen, X., Sun, L., Zhan, S., Chen, L., Cheng, C., Zhang, X., Bu, X., He, W. & Wan, G. (2020) RNA m(6) A methylation regulates sorafenib resistance in liver cancer through FOXO3-mediated autophagy. EMBO Journal, 39:e103181. https://doi.org/10.15252/embj.2019103181
Tsai, Y. P., & Wu, K. J. (2012). Hypoxia-regulated target genes implicated in tumor metastasis. Journal of Biomedical Science, 19, 102 https://doi.org/10.1186/1423-0127-19-102[pii].
Li, H., Ge, C., Zhao, F., Yan, M., Hu, C., Jia, D., Tian, H., Zhu, M., Chen, T., Jiang, G., Xie, H., Cui, Y., Gu, J., Tu, H., He, X., Yao, M., Liu, Y., & Li, J. (2011). Hypoxia-inducible factor 1 alpha-activated angiopoietin-like protein 4 contributes to tumor metastasis via vascular cell adhesion molecule-1/integrin beta1 signaling in human hepatocellular carcinoma. Hepatology, 54, 910–9. https://doi.org/10.1002/hep.24479.
Huang, Q., Li, J., Xing, J., Li, W., Li, H., Ke, X., Zhang, J., Ren, T., Shang, Y., Yang, H., Jiang, J., & Chen, Z. (2014). CD147 promotes reprogramming of glucose metabolism and cell proliferation in HCC cells by inhibiting the p53-dependent signaling pathway. Journal of Hepatology, 61, 859–66. https://doi.org/10.1016/j.jhep.2014.04.035.
Ren, X., Wang, C., Xie, B., Hu, L., Chai, H., Ding, L., Tang, L., Xia, Y., & Dou, X. (2017). Tanshinone IIA induced cell death via miR30b-p53-PTPN11/SHP2 signaling pathway in human hepatocellular carcinoma cells. European Journal of Pharmacology, 796, 233–241. https://doi.org/10.1016/j.ejphar.2016.11.046. S0014-2999(16)30760-9 [pii].
Nazmy, E. A., El-Khouly, O. A., Zaki, M. M. A., Elsherbiny, N. M., Said, E., Al-Gayyar, M. M. H., & Salem, H. A. (2019). Targeting p53/TRAIL/caspase-8 signaling by adiponectin reverses thioacetamide-induced hepatocellular carcinoma in rats. Environmental Toxicology and Pharmacology, 72, 103240. https://doi.org/10.1016/j.etap.2019.103240. S1382-6689(19)30111-5 [pii].
Davalieva, K., Kiprijanovska, S., Maleva Kostovska, I., Stavridis, S., Stankov, O., Komina, S., Petrusevska, G. and Polenakovic, M. (2017). Comparative proteomics analysis of urine reveals down-regulation of acute phase response signaling and LXR/RXR activation pathways in prostate cancer. Proteomes, 6. https://doi.org/10.3390/proteomes6010001
Jin, Y., Liang, Z. Y., Zhou, W. X., & Zhou, L. (2020). Expression, clinicopathologic and prognostic significance of plasminogen activator inhibitor 1 in hepatocellular carcinoma. Cancer Biomark, 27, 285–293. https://doi.org/10.3233/CBM-190560.
Divella, R., Lacalamita, R., Tommasi, S., Coviello, M., Daniele, A., Garrisi, V. M., Abbate, I., Simone, G., Gadaleta, C., Paradiso, A., & Quaranta, M. (2008). PAI-1, t-PA and circulating hTERT DNA as related to virus infection in liver carcinogenesis. Anticancer Research, 28, 223–8.
Divella, R., Daniele, A., Abbate, I., Savino, E., Casamassima, P., Sciortino, G., Simone, G., Gadaleta-Caldarola, G., Fazio, V., Gadaleta, C. D., Sabba, C., & Mazzocca, A. (2015). Circulating levels of PAI-1 and SERPINE1 4G/4G polymorphism are predictive of poor prognosis in HCC patients undergoing TACE. Translate Oncology, 8, 273–8. https://doi.org/10.1016/j.tranon.2015.05.002.
Wang, Z., Huang, D., Huang, J., Nie, K., Li, X., & Yang, X. (2020). lncRNA TMPO-AS1 exerts oncogenic roles in HCC through regulating miR-320a/SERBP1 axis. Onco Targets and Therapy, 13, 6539–6551. https://doi.org/10.2147/OTT.S250355.
Matsuoka, H., Sisson, T. H., Nishiuma, T., & Simon, R. H. (2006). Plasminogen-mediated activation and release of hepatocyte growth factor from extracellular matrix. American Journal of Respiratory Cell and Molecular Biology, 35, 705–13. https://doi.org/10.1165/rcmb.2006-0006OC. 2006-0006OC [pii].
Stefansson, S., Petitclerc, E., Wong, M. K., McMahon, G. A., Brooks, P. C., & Lawrence, D. A. (2001). Inhibition of angiogenesis in vivo by plasminogen activator inhibitor-1. Journal of Biological Chemistry, 276, 8135–41. https://doi.org/10.1074/jbc.M007609200. M007609200[pii].
Ehlken, C., Grundel, B., Michels, D., Junker, B., Stahl, A., Schlunck, G., Hansen, L. L., Feltgen, N., Martin, G., Agostini, H. T., & Pielen, A. (2015). Increased expression of angiogenic and inflammatory proteins in the vitreous of patients with ischemic central retinal vein occlusion. PLoS ONE, 10, e0126859. https://doi.org/10.1371/journal.pone.0126859.
Muench, D. E., Olsson, A., Ferchen, K., Pham, G., Serafin, R. A., Chutipongtanate, S., Dwivedi, P., Song, B., Hay, S., Chetal, K., Trump-Durbin, L. R., Mookerjee-Basu, J., Zhang, K., Yu, J. C., Lutzko, C., Myers, K. C., Nazor, K. L., Greis, K. D., Kappes, D. J., Way, S. S., Salomonis, N., & Grimes, H. L. (2020). Mouse models of neutropenia reveal progenitor-stage-specific defects. Nature, 582, 109–114. https://doi.org/10.1038/s41586-020-2227-7[pii].
Dwivedi, P., Muench, D. E., Wagner, M., Azam, M., Grimes, H. L., & Greis, K. D. (2019). Time resolved quantitative phospho-tyrosine analysis reveals Bruton’s Tyrosine kinase mediated signaling downstream of the mutated granulocyte-colony stimulating factor receptors. Leukemia, 33, 75–87. https://doi.org/10.1038/s41375-018-0188-8[pii].
Dwivedi, P., & Greis, K. D. (2017). Granulocyte colony-stimulating factor receptor signaling in severe congenital neutropenia, chronic neutrophilic leukemia, and related malignancies. Experimental Hematology, 46, 9–20. https://doi.org/10.1016/j.exphem.2016.10.008[pii]. S0301-472X(16)30663-4.
Acknowledgements
The present study was supported by the National Natural Scientific Foundation of China (grant no. 81473487 and 82074138 to JD, no.81803929 to Zifei Yin).
Authors’ Contributions
J.D. conceived and designed the study. Microarray analysis and acquisition of data, including qPCR, tube formation and western blot analyses were performed by H.Z. and Y.M. Cells culture, transfection and tube formation were performed by Z.Z. Analysis and interpretation of data, including statistical analysis, biostatistics, computational analysis was performed by Y.M. and J.D. J.D. and H.Z. wrote, reviewed and/or revised the manuscript. All authors read and approved the manuscript and agree to be accountable for all aspects of the research in ensuring that the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interest.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
About this article
Cite this article
Meng, Y., Zhao, H., Zhao, Z. et al. Sec62 promotes pro-angiogenesis of hepatocellular carcinoma cells under hypoxia. Cell Biochem Biophys 79, 747–755 (2021). https://doi.org/10.1007/s12013-021-01008-6
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12013-021-01008-6