Abstract
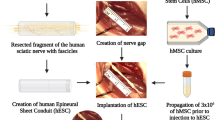
This study is intended to explore the role of human umbilical-cord-derived mesenchymal stem cells (HUC-MSCs) in nerve end-to-side anastomosis, as well as in the induction and promotion of growth of nerve lateral bud. The chitosan nerve conduit was prepared based on the biological characteristics of chitosan, and the nerve conduit was filled with HUC-MSCs, and was used to bridge the nerve end-to-side anastomotic stoma. The experimental animals were randomly assigned into three groups (10 in each group), and the nerve end-to-side anastomosis was conducted: (1) group A (control group): traditional tibial nerve–common peroneal nerve end-to-side anastomosis; (2) group B (experimental group 1): tibial nerve–common peroneal nerve end-to-side anastomotic stoma bridged with chitosan nerve conduit; (3) group C (experimental group 2): tibial nerve–common peroneal nerve end-to-side anastomotic stoma bridged by chitosan nerve conduit filled with HUC-MSCs. General morphological observation, nerve electrophysiology, and anti-S-100 immunohistochemistry were performed. All experimental animals survived, and no infections were found at operative incisions. The nerve continuity was in good condition through visual observation when sampling, which is mild adhesion to the surrounding tissue and easy to be separated. 12 W HUC-MSCs chitosan composite nerve conduits were degraded completely after operation. Electrophysiological test showed that the nerve conduction velocity (NCV) in group C was significantly higher than that in group A or group B (p < 0.01). There were no significant differences between NCVs of group A and group B. Toluidine blue staining and transmission electron microscope showed that the number of the medullated fibers and the myelin sheath thickness in group C were larger than those in group A or B. There were no significant differences between the numbers of the medullated fibers and between the myelin sheath thicknesses of groups A and B. By means of anti-S-100 immunohistochemistry, the arrangement of a large number of brown-red proliferating schwann cells around the regenerated nerve fibers in group C could be found, while fewer and sparse brown-red matters and very poor growth of schwann cells could be observed in groups A and B. Slightly more favorable situation could be observed in group B compared with group A. HUC-MSCs play obviously an important role in promoting nerve regeneration during the nerve end-to-side anastomosis, which induces the growth of axis bud, accelerates the growth velocity of regenerated fiber, and promotes the growth and maturity of schwann cells.




Similar content being viewed by others
References
Voterbo, F., Trindade, J. C., Hoshin, K., et al. (1994). End-to-side neurorrhaphy with removal of the epineural sheath: Experimental study in rats. Plastic and Reconstructive Surgery, 94(7), 1038–1047.
Giovasidis, G., Watanabe, O., Mackinnon, S. E., et al. (2000). Functional and morphometric evaluation of end-to-side neurorrhaphy for muscle reinnervation. Plastic and Reconstructive Surgery, 106(4), 383–392.
Song, C., Oswald, T., Yan, H., et al. (2009). Repair of partial nerve injury by bypass nerve grafting with end-to-side neurorrhaphy. Journal of Reconstructive Microsurgery, 25(8), 483–495.
Spyropoulou, G. A., Lykoudis, E. G., Batistatou, A., et al. (2007). New pure motor nerve experimental model for the comparative study between end-to-end and end-to-side neurorrhaphy in free muscle flap neurotization. Journal of Reconstructive Microsurgery, 23(7), 391–398.
Zhang, Z., Sourcacos, P. N., Bo, J., et al. (1999). Evaluation of collateral sprouting after end-to-side nerve coaptation using a fluorescing double-labeling technique. Microsurgery, 19(6), 281–286.
Zhu, Q., Zhu, J., Lao, Z., et al. (1999). Morphological study on collateral regeneration after nerve end-to-side anastomosis. Chinese Journal of Microsurgery, 22(2), 117–119.
Kim, J. K., Chung, M. S., & Baek, G. H. (2011). The origin of regenerating axons after end-to-side neurorrhaphy without donor nerve injury. Journal of Plastic, Reconstructive & Aesthetic Surgery, 64, 255–260.
Viterbo, F., Trindade, J. C., Hoshino, et al. (1992). Lateroterminal neurorrhaphy without removal of the epineural sheath.experimental study in rats. Revista Paulista de Medicina, 110(6), 267–275.
Mennen, U. (2003). End-to-side nerve suture in clinical practice. The Journal of Hand Surgery, 8(1), 33–42.
Sherif, M., & Amr, A. H. (2010). Intrinsic hand muscle reinnervation by median-ulnar end-to-side bridge nerve graft: Case report. The Journal of Hand Surgery, 35(3), 446–450.
Zhang, Y., Chen, D., Huang, C., et al. (2003). Study on the clinical application and effect of nerve end-to-side anastomosis. Youjiang Medical Journal, 31(5), 429.
Fan, C. G., Zhang, Q. J., & Zhou, J. R. (2011). Therapeutic potentials of tiles-enchymal stem cells derived from human umbilical cord. Stem Cell Reviews and Reports, 7(1), 195–207.
Serge, R., Martin, S., Michal, S., et al. (2007). Spinal cord injury: Time to move. The Journal of Neuroscience, 27(44), 11782–11792.
Wang, Y., Han, Z. B., Ma, J., Zuo, C., Geng, J., Gong, W., et al. (2012). A toxicity study of multiple-administration human umbilical cord mesenchymal stem cells in cynomolgus monkeys. Stem Cells and Development, 21(9), 1401–1408.
Lund, R. D., Wang, S., Lu, B., et al. (2007). Cells isolated from umbilical cord tissue rescue photoreceptors and visual functions in a rodent model of retina1 disease. Stem Cells, 25(3), 602–611.
Baksh, D., Yao, R., & Tuan, R. S. (2007). Comparison of proliferative and multilineage differentiation potential of human mesenchymal stem cells derived from umbilical cord and bone marrow. Stem Cells, 25(6), 1384–1392.
Karahuseyinoglu, S., Cinar, O., Kilic, E., et al. (2007). Biology of the stem cells in human umbilical cord stroma: In situ and in vitro surveys. Stem Cells, 25(2), 319–331.
Ma, L., Feng, X. Y., Cui, B. L., et al. (2005). Human umbilical cord Wharton’s jelly-derived mesenchymal stem cells differentiation into nerve-like cells. Chinese Medicine Journal, 118(23), 1987–1993.
Shetty, P., Cooper, K., & Viswanathan, C. (2010). Comparison of proliferative and multilineage differentiation potentials of cord matrix, cord blood, and bone marrow mesenchymal stem cells. Asian Journal of Transfusion Science, 4(1), 14–24.
Kermani, A. J., Fathi, F., & Mowla, S. J. (2008). Characterization and genetic manipulation of human umbilical cord vein mesenchymal stem cells: Potential application in cell-based gene therapy. Rejuvenation Research, 11(2), 379–386.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Xiao, Q., Zhang, X. & Wu, Y. Experimental Research on Differentiation-Inducing Growth of Nerve Lateral Bud by HUC-MSCs Chitosan Composite Conduit. Cell Biochem Biophys 73, 305–311 (2015). https://doi.org/10.1007/s12013-015-0578-8
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12013-015-0578-8