Abstract
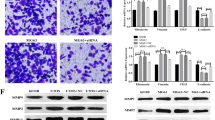
Osteosarcoma is the most common type of solid bone cancer, which is the second leading cause of cancer-related death. Hypoxia is an ordinary phenomenon in solid tumor tissues and can induce cell apoptosis but the specific molecular mechanism remains unclear. In this study, we explored the effect and the molecular mechanism of Transglutaminase 2 (TG2) on cell apoptosis in osteosarcoma U2OS cells under hypoxia. We found the enzymatic activity of TG2 is significantly increased and the expression of TG2 is remarkably up-regulated under hypoxia condition. Cell apoptotic rate is markedly increased upon knockdown of TG2 by siRNA under hypoxia. We further investigated the mechanism of cell apoptosis and found Bax protein is significantly increased after depletion of TG2 under hypoxia. Moreover, our data also show that cytochrome C (Cyt C) is significantly increased in cytoplasm and markedly decreased in mitochondria of U2OS cells after depletion of TG2 under hypoxia. Our results suggest that TG2 can inhibit tumor cell apoptosis through down-regulation of Bax and prevention of release Cyt C from mitochondria into cytoplasm.





Similar content being viewed by others
References
Kakkad, S. M., et al. (2013). Hytoxia tumor environments exhibit disrupted collagen I fibers and low macromolecular transport. PLoS ONE, 8(12), e81869.
Pouyssegur, J., Dayan, F., & Mazure, N. M. (2006). Hypoxia signalling in cancer and approaches to enforce tumour regression. Nature, 441(7092), 437–443.
Siclari, V. A., & Qin, L. (2010). Targeting the osteosarcoma cancer stem cell. Journal of Orthopaedic Surgery and Research, 5, 78.
Kuijjer, M. L., Hogendoorn, P. C., & Cleton-Jansen, A. M. (2013). Genome-wide analyses on high-grade osteosarcoma: Making sense of a genomically most unstable tumor. International Journal of Cancer, 133(11), 2512–2521.
Anninga, J. K., et al. (2011). Chemotherapeutic adjuvant treatment for osteosarcoma: Where do we stand? European Journal of Cancer, 47(16), 2431–2445.
Zhang, Y. Q., et al. (2013). Silencing of Pokemon enhances caspase-dependent apoptosis via fas- and mitochondria-mediated pathways in hepatocellular carcinoma cells. PLoS ONE, 8(7), e68981.
Hsieh, Y. F., et al. (2013). Transglutaminase 2 contributes to apoptosis induction in Jurkat T cells by modulating Ca2+ homeostasis via cross-linking RAP1GDS1. PLoS ONE, 8(12), e81516.
Lauzier, A., et al. (2012). Transglutaminase 2 cross-linking activity is linked to invadopodia formation and cartilage breakdown in arthritis. Arthritis Res Ther, 14(4), R159.
Wang, Y., Ande, S. R., & Mishra, S. (2012). Phosphorylation of transglutaminase 2 (TG2) at serine-216 has a role in TG2 mediated activation of nuclear factor-kappa B and in the downregulation of PTEN. BMC Cancer, 12, 277.
Shibata, T., et al. (2013). Transglutaminase-catalyzed protein-protein cross-linking suppresses the activity of the NF-κB-like transcription factor relish. Sci Signal, 6(285), ra61.
Lee, H. J., & Lee, C. H. (2013). Transglutaminase-2 is involved in expression of osteoprotegerin in MG-63 osteosarcoma cells. Biomolecules & Therapeutics (Seoul), 21(3), 204–209.
Cho, S. Y., et al. (2010). Transglutaminase 2 inhibits apoptosis induced by calcium- overload through down-regulation of Bax. Experimental & Molecular Medicine, 42(9), 639–650.
Jang, G. Y., et al. (2010). Transglutaminase 2 suppresses apoptosis by modulating caspase 3 and NF-κB activity in hypoxic tumor cells. Oncogene, 29(3), 356–367.
Shin, D. M., et al. (2004). Cell type-specific activation of intracellular transglutaminase 2 by oxidative stress or ultraviolet irradiation: Implications of transglutaminase 2 in age-related cataractogenesis. Journal of Biological Chemistry, 279(15), 15032–15039.
Waterhouse, N. J., et al. (2001). The (Holey) study of mitochondria in apoptosis. Methods in Cell Biology, 66, 365–391.
Hong, L., et al. (2009). Mitochondria-initiated apoptosis triggered by oxidative injury play a role in total parenteral nutrition-associated liver dysfunction in infant rabbit model. Journal of Pediatric Surgery, 44(9), 1712–1718.
Ip, S. W., et al. (2008). The role of mitochondria in bee venom-induced apoptosis in human breast cancer MCF7 cells. Vivo, 22(2), 237–245.
Nurminskaya, M. V., & Belkin, A. M. (2012). Cellular functions of tissue transglutaminase. International Review of Cell and Molecular Biology, 294, 1–97.
Sw, F. (2005). Promoting apoptosis as a strategy for cancer drug diacovery. Nature Reviews Cancer, 5(11), 876–885.
Jergens, A., et al. (2014). Bcl-2/Caspase 3 mucosal imbalance favors T cell resistance to apoptosis in dogs with inflammatory bowel disease. Veterinary Immunology and Immunopathology, 158(3–4), 167–174.
Westphal, D., Kluck, R. M., & Dewson, G. (2014). Building blocks of the apoptotic pore: How Bax and Bak are activated and oligomerize during apoptosis. Cell Death and Differentiation, 21(2), 196–205.
Author information
Authors and Affiliations
Corresponding author
Additional information
Wei Wang and Xiao Li have contributed equally to this study.
Rights and permissions
About this article
Cite this article
Wang, W., Li, X., Han, Xz. et al. Transglutaminase-2 is Involved in Cell Apoptosis of Osteosarcoma Cell Line U2OS Under Hypoxia Condition. Cell Biochem Biophys 72, 283–288 (2015). https://doi.org/10.1007/s12013-014-0451-1
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12013-014-0451-1