Abstract
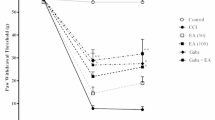
Our objective is to analyze and observe the different administration routes of parecoxib sodium pretreatment on the behavioral improvement of rats with neuropathic pain to provide the preclinical data of parecoxib sodium on neuropathic pain treatment. 30 SD rats were randomly divided into five groups, including model group, sham operation group, intrathecal injection group (IT group), intraperitoneal injection group (IP group), and perineural infiltration group (PI group). The rats in model group and three parecoxib sodium pretreatment groups received spinal nerve ligation (SNL). Heat pain test and 50 % paw mechanical withdrawal threshold test (50 % PMWT) were use to assess the responses after parecoxib sodium pretreatment. 50 % PMWT results of right foot in five groups had no statistical difference (P > 0.05); 50 % PMWT results of left and right feet in three parecoxib sodium pretreatment groups were obviously higher than SNL group at different time points, which was statistically different (P < 0.05); in comparison with three pretreatment groups, the data of left foot in IT group were obviously higher than PI group and IP group, and the comparison among three groups had significant difference (P < 0.05). However, the data of right foot had no significant difference among three groups (P > 0.05). Paw thermal withdrawal latency (PTWL) results of left and right feet in five groups had no significant difference before surgery (P > 0.05); after the establishment of neuropathic model, PTWL results in five groups were significantly decreased; however, PTWL results of left and right feet at 3 days after surgery in IT group were significantly higher than the two other pretreatment groups (P < 0.05); PTWL results of left and right feet at 7 and 14 days after surgery had no significant difference. Parecoxib sodium pretreatment can effectively improve the behaviors caused by neuropathic pain, and intrathecal injection is the most effective route of administration.
Similar content being viewed by others
References
Arle, J. E., et al. (2014). Mechanism of dorsal column stimulation to treat neuropathic but not nociceptive pain: Analysis with a computational model. Neuromodulation, 17(7), 642–655.
Bohren, Y., et al. (2013). Antidepressants suppress neuropathic pain by a peripheral beta2-adrenoceptor mediated anti-TNFalpha mechanism. Neurobiology of Diseases, 60, 39–50.
Kandhare, A. D., et al. (2013). Elucidation of molecular mechanism involved in neuroprotective effect of coenzyme Q10 in alcohol-induced neuropathic pain. Fundamental & Clinical Pharmacology, 27(6), 603–622.
Akcakaya, M. O., et al. (2012). Cervical intradural glass fragment: A rare cause of neuropathic pain. Turkish Neurosurgery, 22(5), 667–670.
Nakagawa, S., et al. (2011). Can milnacipran used for neuropathic pain in patients with advanced cancer cause neuromuscular and somatosensory disorders? Journal of Palliative Medicine, 14(4), 403–405.
Zenonos, G., & Kim, J. E. (2010). A T cell-orchestrated immune response in the adult dorsal spinal cord as a cause of neuropathic pain-like hypersensitivity after peripheral nerve damage: A door to novel therapies? Neurosurgery, 66(4), N24–N25.
Fishbain, D. A., et al. (2009). Does pain cause the perception of fatigue in patients with chronic pain? Findings from studies for management of diabetic peripheral neuropathic pain with duloxetine. Pain Practice, 9(5), 354–362.
Schroder, S., et al. (2013). Can medical herbs stimulate regeneration or neuroprotection and treat neuropathic pain in chemotherapy-induced peripheral neuropathy? Evidence-Based Complement and Alternative Medicine, 2013, 423713.
Eaton, M. J., Berrocal, Y., & Wolfe, S. Q. (2012). Potential for cell-transplant therapy with human neuronal precursors to treat neuropathic pain in models of PNS and CNS injury: Comparison of hNT2.17 and hNT2.19 cell lines. Pain Research and Treatment, 2012, 356412.
Hayashida, K., & Eisenach, J. C. (2008). Multiplicative interactions to enhance gabapentin to treat neuropathic pain. European Journal of Pharmacology, 598(1–3), 21–26.
Lenz, H., et al. (2011). Effects of COX inhibition on experimental pain and hyperalgesia during and after remifentanil infusion in humans. Pain, 152(6), 1289–1297.
Liu, X., et al. (2013). Parecoxib added to ropivacaine prolongs duration of axillary brachial plexus blockade and relieves postoperative pain. Clinical Orthopaedics and Related Research, 471(2), 562–568.
Williams, D. L., Pemberton, E., & Leslie, K. (2011). Effect of intravenous parecoxib on post-craniotomy pain. British Journal of Anaesthesia, 107(3), 398–403.
Bao, Y., et al. (2012). Comparison of preincisional and postincisional parecoxib administration on postoperative pain control and cytokine response after total hip replacement. Journal of International Medical Research, 40(5), 1804–1811.
Akimoto, N., et al. (2013). CCL-1 in the spinal cord contributes to neuropathic pain induced by nerve injury. Cell Death and Disease, 4, e679.
Liu, M., et al. (2012). Analgesic effect of iridoid glycosides from Paederia scandens (LOUR.) MERRILL (Rubiaceae) on spared nerve injury rat model of neuropathic pain. Pharmacology, Biochemistry and Behavior, 102(3), 465–470.
Kim, C. F., & Moalem-Taylor, G. (2011). Interleukin-17 contributes to neuroinflammation and neuropathic pain following peripheral nerve injury in mice. Journal of Pain, 12(3), 370–383.
Guan, Y., et al. (2008). Peripherally acting mu-opioid receptor agonist attenuates neuropathic pain in rats after L5 spinal nerve injury. Pain, 138(2), 318–329.
Mamet, J., et al. (2014). Single intrathecal administration of the transcription factor decoy AYX1 prevents acute and chronic pain after incisional, inflammatory, or neuropathic injury. Pain, 155(2), 322–333.
Alexander, J. K., et al. (2012). Macrophage migration inhibitory factor (MIF) is essential for inflammatory and neuropathic pain and enhances pain in response to stress. Experimental Neurology, 236(2), 351–362.
Zhao, J., et al. (2006). Nociceptor-derived brain-derived neurotrophic factor regulates acute and inflammatory but not neuropathic pain. Molecular and Cellular Neuroscience, 31(3), 539–548.
Quintao, N. L., et al. (2006). Long-lasting neuropathic pain induced by brachial plexus injury in mice: Role triggered by the pro-inflammatory cytokine, tumour necrosis factor alpha. Neuropharmacology, 50(5), 614–620.
Chodakiewitz, Y. G., Bicalho, G. V., & Chodakiewitz, J. W. (2013). Multi-target neurostimulation for adequate long-term relief of neuropathic and nociceptive chronic pain components. Surgical Neurology International, 4(Suppl 3), S170–S175.
Zamponi, G. W. (2011). Antiallodynic effects of a confused alpha-conotoxin: Vc1.1 relieves neuropathic pain via off target actions on GABA(B) receptors and N-type channels. Pain, 152(2), 241–242.
Sakai, A., & Suzuki, H. (2008). NCAM as a target for GDNF-induced analgesia in neuropathic pain. Journal of Nippon Medical School, 75(3), 136–137.
Li, J. Z., et al. (2013). Effects of parecoxib sodium analgesia on serum concentrations of neuron-specific enolase and S-100beta and postoperative cognitive function of elderly patients undergoing acute replacement of femoral head. Zhonghua Yi Xue Za Zhi, 93(27), 2152–2154.
Apfelbaum, J. L., et al. (2008). Multiple-day efficacy of parecoxib sodium treatment in postoperative bunionectomy pain. Clinical Journal of Pain, 24(9), 784–792.
Wiktorowska-Owczarek, A. (2013). The effect of valdecoxib on the production of growth factors evoked by hypoxia and bacterial lipopolysaccharide in HMEC-1 cells. Advances in Clinical and Experimental Medicine, 22(6), 795–800.
Thakral, N. K., et al. (2011). The quest for targeted delivery in colon cancer: Mucoadhesive valdecoxib microspheres. International Journal of Nanomedicine, 6, 1057–1068.
Keshetti, S., & Ciddi, V. (2010). Biotransformation of valdecoxib by microbial cultures. Journal of Microbiology and Biotechnology, 20(4), 809–816.
Zall, A., et al. (2011). NSAID-derived gamma-secretase modulation requires an acidic moiety on the carbazole scaffold. Bioorganic and Medicinal Chemistry, 19(16), 4903–4909.
Valdes, A. M., et al. (2014). History of knee surgery is associated with higher prevalence of neuropathic pain-like symptoms in patients with severe osteoarthritis of the knee. Seminars in Arthritis and Rheumatism, 43(5), 588–592.
Searle, R. D., & Bennett, M. I. (2011). Neuropathic pain after thoracic surgery. Pain, 152(4), 958.
Acknowledgments
This study was funded by 2011 Research Project of Shenyang Science and Technology Bureau (F11-262-9-04).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Cui, W., Yu, X. & Zhang, H. The Study of Different Approaches of Parecoxib Sodium Pretreatment on the Behavior of Rats with Neuropathic Pain. Cell Biochem Biophys 72, 137–140 (2015). https://doi.org/10.1007/s12013-014-0424-4
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12013-014-0424-4