Abstract
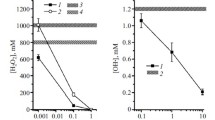
The present study was conducted to explore the protective effect of exogenous gelsolin (GSN) in mice exposed to high-dose of radiation. Changes in the levels of GSNs in peripheral blood of mice and cytoplasm of cultured human intestinal epithelial cells (HIECs) were analyzed after their exposure to different doses of 137Cs γ-rays at a fixed dose rate. The coagulation associated indices, such as prothrombin time (PT) and activated partial thromboplastin time (APTT) were measured. Effect on radiation-mediated oxidative damage was evaluated by estimating the altered glutathione (GSH) and malondialdehyde (MDA) concentrations in the blood. The results showed that radiation induced a pronounced decrease in the pGSN blood levels. However, the cGSN levels of irradiated HIECs were increased in a dose-dependent manner. Administration of recombinant human pGSN to irradiated mice resulted in an ameliorated clotting time as indicated by the PT and the APTT indices. The treatment of mice with hpGSN enhanced the blood levels of GSH while MDA concentrations were decreased indicating an improved antioxidant status. These results suggest that GSNs might play a regulatory role in the suppression of the tissue damage induced by acute radiation exposure.




Similar content being viewed by others
References
Yin, H. L., Kwiatkowski, D. J., Mole, J. E., & Cole, F. S. (1984). Structure and biosynthesis of cytoplasmic and secreted variants of gelsolin. Journal of Biological Chemistry, 259, 5271–5276.
Kwiatkowski, D. J., Mehl, R., Izumo, S., Nadal-Ginard, B., & Yin, H. L. (1988). Muscle is the major source of plasma gelsolin. Journal of Biological Chemistry, 263, 8239–8243.
Silacci, P., Mazzolai, L., Gauci, C., Stergiopulos, N., Yin, H. L., & Hayoz, D. (2004). Gelsolin superfamily proteins: key regulators of cellular functions. Cellular and Molecular Life Sciences, 61, 2614–2623.
Kwiatkowski, D. J., Stossel, T. P., Orkin, S. H., Mole, J. E., Colten, H. R., & Yin, H. L. (1986). Plasma and cytoplasmic gelsolins are encoded by a single gene and contain a duplicated actin-binding domain. Nature, 323, 455–458.
Vouyiouklis, D. A., & Brophy, P. J. (1997). A novel gelsolin isform expressed by oligodendrocytes in the central nervaous system. Neurochem, 69(3), 995–1005.
Tanaka, J., & Sobue, K. (1994). Localization and characterization of gelsolin in nervous tissues: gelsolin is specifically enriched in myelin-forming cells. Journal of Neuroscience, 14, 1038–1052.
Teubner, A., Sobek-Klocke, I., Hinssen, H., & Eichenlaub-Ritter, U. (1994). Distribution of gelsolin in mouse ovary. Cell and Tissue Research, 276, 535–544.
Lueck, A., Brown, D., & Kwiatkowski, D. J. (1998). The actin-binding proteins adseverin and gelsolin are both highly expressed but differentially localized in kidney and intestine. Journal of Cell Science, 111, 3633–3643.
Arai, M., & Kwiatkowski, D. J. (1999). Differential developmentally regulated expression of gelsolin family members in the mouse. Developmental Dynamics, 215, 297–307.
Witke, W., Sharpe, A. H., Hartwig, J. H., Azuma, T., Stossel, T. P., & Kwiatkowski, D. J. (1995). Hemostatic, inflammatory, and fibroblast responses are blunted in mice lacking gelsolin. Cell, 81, 41–51.
Barkalow, K., Witke, W., Kwiatkowski, D. J., & Hartwig, J. H. (1996). Coordinated regulation of platelet actin filament barbed ends by gelsolin and capping protein. Journal of Cell Biology, 134, 389–399.
Kwiatkowski, D. J., Mehl, R., & Yin, H. L. (1988). Genomic organization and biosynthesis of secreted and cytoplasmic forms of gelsolin. Journal of Cell Biology, 106, 375–384.
Mark, J. D. (2008). Plasma gelsolin as a biomarker of inflammation. Arthritis Research and Therapy, 10, 1–2.
Peddada, N., Sagar, A., Ashish, & Garg, R. (2012). Plasma gelsolin: a general prognostic marker of health. Medical Hypotheses, 78, 203–210.
Lee, P. S., Drager, L. R., Stossel, T. P., Moore, F. D., & Rogers, S. O. (2006). Relationship of plasma gelsolin levels to outcomes in critically ill surgical patients. Annals of Surgery, 243, 399–403.
Li, G. H., Arora, P. D., Chen, Y., McCulloch, C. A., & Liu, P. (2012). Multifunctional roles of gelsolin in health and diseases. Medicinal Research Reviews, 32, 999–1025.
Ji, L., Chauhan, A., Muthaiyah, B., Wegiel, J., & Chauhan, V. (2009). Gelsolin levels are increased in the brain as a function of age during normal development in children that are further increased in down syndrome. Alzheimer Disease and Associated Disorders, 23, 319–322.
Ji, L., Chauhan, A., & Chauhan, V. (2010). Upregulation of cytoplasmic gelsolin, an amyloid-beta binding protein, under oxidative stress conditions: involvement of protein kinase C. Journal of Alzheimer’s Disease, 19, 829–838.
Jordan, J. R., Moore, E. E., Damle, S. S., Eckels, P., Johnson, J. L., Roach, J. P., et al. (2007). Gelsolin is depleted in post-shock mesenteric lymph. Journal of Surgical Research, 143, 130–135.
Lee, W. M., & Galbraith, R. M. (1992). The extracellular actin-scavenger system and actin toxicity. New Engl J Med, 326, 1335–1341.
Haddad, J. G., Harper, K. D., Guoth, M., Pietra, G. G., & Sanger, J. W. (1990). Angiopathic consequences of saturating the plasma scavenger system for actin. Proceedings of the National Academy of Sciences of the United States of America, 87, 1381–1385.
Erukhimov, J. A., Tang, Z. L., Johnson, B. A., Donahoe, M. P., Razzack, J. A., Gibson, K. F., et al. (2000). Actin-containing sera from patients with adult respiratory distress syndrome are toxic to sheep pulmonary endothelial cells. American Journal of Respiratory and Critical Care Medicine, 162, 288–294.
Kułakowska, A., Ciccarelli, N. J., Wen, Q., Mroczko, B., Drozdowski, W., Szmitkowski, M., et al. (2010). Hypogelsolinemia, a disorder of the extracellular actin scavenger system, in patients with multiple sclerosis. BMC Neurology, 10, 107–115.
Chauhan, V., Ji, L., & Chauhan, A. (2008). Anti-amylodogenic, anti-oxidant and anti-apoptotic role gelsolin in Alzheimer’s disease. Biogerontology, 9, 381–389.
Carro, E. (2010). Gelsolin as therapeutic target in Alzheimer’s disease. Expert Opinion on Therapeutic Targets, 14, 585–592.
Bucki, R., Levental, I., Kulakowska, A., & Janmey, P. A. (2008). Plasma gelsolin: function, prognostic value, and potential therapeutic use. Current Protein and Peptide Science, 9, 541–551.
Lee, P. S., Sampath, K., Karumanchi, S. A., Tamez, H., Bhan, I., Isakova, T., et al. (2009). Plasma gelsolin and circulating actin correlate with hemodialysis mortality. Journal of the American Society of Nephrology, 20, 1140–1148.
Grabryś, D., Greco, O., Patel, G., Prise, K. M., Tozer, G. M., & Kanthou, C. (2007). Radiation effects on the cytoskeleton of endothelial cells and endothelial monolayer permeability. International Journal of Radiation Oncology Biology Physics, 69, 1553–1562.
Guo, X. C., Luo, B. Y., Li, X. F., Yang, D. G., Zheng, X. N., & Zhang, K. (2011). Plasma gelsolin levels and 1-year mortality after first-ever ischemic stroke. Journal of Critical Care, 26, 608–612.
Lee, P. S., Waxman, A. B., Cotich, K. L., Chung, S. W., Perrella, M. A., & Stossel, T. P. (2007). Plasma gelsolin is a marker and therapeutic agent in animal sepsis. Critical Care Medicine, 35, 849–855.
Meier, U., Gressner, O., Lammert, F., & Gressner, A. M. (2006). Gc-globulin: roles in response to injury. Clinical Chemistry, 52, 1247–1253.
Dahl, B., Schiødt, F. V., Ott, P., Wians, F., Lee, W. M., Balko, J., et al. (2003). Plasma concentration of Gc-globulin is associated with organ dysfunction and sepsis after injury. Critical Care Medicine, 31, 152–156.
Ono, S. (2007). Mechanism of depolymerization and severing of actin filaments and its significance in cytoskeletal dynamics. International Review of Cytology, 258, 1–8.
Christofidou-Solomidou, M., Scherpereel, A., Somolides, C. C., Muzykantov, V. R., Machtay, M., Albelda, S. M., et al. (2002). Changes in plasma gelsolin concentration during acute oxidant lung injury in mice. Lung, 180, 91–104.
Osborn, T. M., Dahlgren, C., Hartwig, J. H., & Stossel, T. P. (2011). Modifications of cellular responses to lysophosphotidic acid and platelet activating factor by plasma gelsolin. American Journal of Physiology: Cell Physiology, 292, C1323–C1330.
Bohgaki, M., Matsumoto, M., Atsumi, T., Kondo, T., Yasuda, S., Horita, T., et al. (2011). Plasma gelsolin facilitates interaction between β2 glycoprotein I and α5β1 integrin. Journal of Cellular and Molecular Medicine, 15, 141–151.
Acknowledgments
The research was supported by the National Natural Science Fund 81372932 and Army Force Fund BWS11J009. We thank Dr.Po-shun Lee (Brigham and Women's hospital, Boston, MA) for valuable suggestions and providing us with the recombiant human gelsolin and also thank Dr. Thomas P.Stossel(Brigham and Women's Hospital, Boston, MA) and Susan Goelz (Biogen,Cambridge, MA) for their interest in this work and for providing useful advice.
Conflict of interest
The authors declare that they have no competing interests, including, but not limited to, conflicts of interest regarding the gift of gelsolin.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Mingjuan Li and Fengmei Cui have contributed equally to this work.
Rights and permissions
About this article
Cite this article
Li, M., Cui, F., Cheng, Y. et al. Gelsolin: Role of a Functional Protein in Mitigating Radiation Injury. Cell Biochem Biophys 71, 389–396 (2015). https://doi.org/10.1007/s12013-014-0210-3
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12013-014-0210-3