Abstract
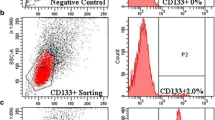
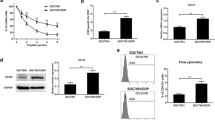
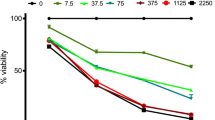
The objective of this study is to investigate the chemoresistance of CD133+ cancer stem cells in Hep-2 cells of laryngeal cancer and detect the expression mRNA and protein levels of BMI-1 in CD133+ cells and CD133− cells. The response of Hep-2 cells to different chemotherapeutic agents was investigated, and the expression of CD133 was studied. Fluorescence-activated cell sorting analysis was used to identify CD133, and the CD133+ subset of cells was separated and analyzed chemotherapy resistance. Colony formation assays were studied and cells were injected subcutaneously into axillary fossa of node mice to measure the tumor-forming ability. RT-PCR and Western blot analyses were used to detect the expression levels of BMI-1 in the different subpopulation cells. It was concluded that chemotherapy enriched the CD133+ subpopulation 2-fourfold, relative to the untreated cells. 1.55 ± 0.28 % of Hep-2 cells were observed to be CD133+ cells. Flow cytometric analysis revealed that after the treatment with these chemotherapeutic agents, the expression of CD133 was up to 5.16 ± 0.86 %, 4.94 ± 0.58 %, 3.66 ± 0.59 %. After 5-FU treatment, the expression of CD133 was 6.7 ± 1.6 % relative to the untreated mice 2.6 ± 0.96 % by nude mice tumor xenograft model. CD133+ cancer stem cells were more resistant to chemotherapy; the proliferation capability and tumor-forming ability were no difference after chemotherapy. Semi-quantitative RT-PCR and Western blot analyses provided strong evidence that BMI-1 expression in CD133+ cells is different from CD133− cells remarkably. Taken together, it was confirmed that CD133+ cancer stem cells were chemoresistant and BMI-1 was highly expressed in these CD133+ cells.






Similar content being viewed by others
References
Chen, H., Zhou, L., Dou, T., Wan, G., Tang, H., & Tian, J. (2011). BMI1’s maintenance of the proliferative capacity of laryngeal cancer stem cells. Head and Neck, 33(8), 1115–1125.
Rubin, B. P., & Duensing, A. (2006). Mechanisms of resistance to small molecule kinase inhibition in the treatment of solid tumors. Laboratory Investigation, 86(10), 981–986.
Raguz, S., & Yague, E. (2008). Resistance to chemotherapy: New treatments and novel insights into an old problem. British Journal of Cancer, 99(3), 387–391.
Burkert, J., Wright, N. A., & Alison, M. R. (2006). Stem cells and cancer: An intimate relationship. The Journal of Pathology, 209(3), 287–297.
Grotenhuis, B. A., Wijnhoven, B. P., & van Lanschot, J. J. (2012). Cancer stem cells and their potential implications for the treatment of solid tumors. Journal of Surgical Oncology, 106(2), 209–215.
Al-Hajj, M., Wicha, M. S., Benito-Hernandez, A., Morrison, S. J., & Clarke, M. F. (2003). Prospective identification of tumorigenic breast cancer cells. Proceedings of the National Academy of Sciences of the United States of America, 100(7), 3983–3988.
Singh, S. K., Hawkins, C., Clarke, I. D., Squire, J. A., Bayani, J., Hide, T., et al. (2004). Identification of human brain tumour initiating cells. Nature, 432(7015), 396–401.
Hemmati, H. D., Nakano, I., Lazareff, J. A., Masterman-Smith, M., Geschwind, D. H., Bronner-Fraser, M., et al. (2003). Cancerous stem cells can arise from pediatric brain tumors. Proceedings of the National Academy of Sciences of the United States of America, 100(25), 15178–15183.
Patrawala, L., Calhoun, T., Schneider-Broussard, R., Li, H., Bhatia, B., Tang, S., et al. (2006). Highly purified CD44+ prostate cancer cells from xenograft human tumors are enriched in tumorigenic and metastatic progenitor cells. Oncogene, 25(12), 1696–1708.
Collins, A. T., Berry, P. A., Hyde, C., Stower, M. J., & Maitland, N. J. (2005). Prospective identification of tumorigenic prostate cancer stem cells. Cancer Research, 65(23), 10946–10951.
Houghton, J., Stoicov, C., Nomura, S., Rogers, A. B., Carlson, J., Li, H., et al. (2004). Gastric cancer originating from bone marrow-derived cells. Science, 306(5701), 1568–1571.
Kim, C. F., Jackson, E. L., Woolfenden, A. E., Lawrence, S., Babar, I., Vogel, S., et al. (2005). Identification of bronchioalveolar stem cells in normal lung and lung cancer. Cell, 121(6), 823–835.
O’Brien, C. A., Pollett, A., Gallinger, S., & Dick, J. E. (2007). A human colon cancer cell capable of initiating tumour growth in immunodeficient mice. Nature, 445(7123), 106–110.
Ricci-Vitiani, L., Lombardi, D. G., Pilozzi, E., Biffoni, M., Todaro, M., Peschle, C., et al. (2007). Identification and expansion of human colon-cancer-initiating cells. Nature, 445(7123), 111–115.
Ma, S., Chan, K. W., Hu, L., Lee, T. K., Wo, J. Y., Ng, I. O., et al. (2007). Identification and characterization of tumorigenic liver cancer stem/progenitor cells. Gastroenterology, 132(7), 2542–2556.
Yang, Z. F., Ngai, P., Ho, D. W., Yu, W. C., Ng, M. N., Lau, C. K., et al. (2008). Identification of local and circulating cancer stem cells in human liver cancer. Hepatology, 47(3), 919–928.
Spillane, J. B., & Henderson, M. A. (2007). Cancer stem cells: A review. ANZ journal of surgery, 77(6), 464–468.
Eramo, A., Lotti, F., Sette, G., Pilozzi, E., Biffoni, M., Di Virgilio, A., et al. (2008). Identification and expansion of the tumorigenic lung cancer stem cell population. Cell Death and Differentiation, 15(3), 504–514.
Hagiwara, S., Kudo, M., Ueshima, K., Chung, H., Yamaguchi, M., Takita, M., et al. (2011). The cancer stem cell marker CD133 is a predictor of the effectiveness of S1+ pegylated interferon alpha-2b therapy against advanced hepatocellular carcinoma. Journal of Gastroenterology, 46(2), 212–221.
Ong, C. W., Kim, L. G., Kong, H. H., Low, L. Y., Iacopetta, B., Soong, R., et al. (2010). CD133 expression predicts for non-response to chemotherapy in colorectal cancer. Modern Pathology, 23(3), 450–457.
Hermann, P. C., Huber, S. L., Herrler, T., Aicher, A., Ellwart, J. W., Guba, M., et al. (2007). Distinct populations of cancer stem cells determine tumor growth and metastatic activity in human pancreatic cancer. Cell Stem Cell, 1(3), 313–323.
Zhao, P., Lu, Y., Jiang, X., & Li, X. (2011). Clinicopathological significance and prognostic value of CD133 expression in triple-negative breast carcinoma. Cancer Science, 102(5), 1107–1111.
Zhou, L., Wei, X., Cheng, L., Tian, J., & Jiang, J. J. (2007). CD133, one of the markers of cancer stem cells in Hep-2 cell line. The Laryngoscope, 117(3), 455–460.
Wei, X. D., Zhou, L., Cheng, L., Tian, J., Jiang, J. J., & Maccallum, J. (2009). In vivo investigation of CD133 as a putative marker of cancer stem cells in Hep-2 cell line. Head and Neck, 31(1), 94–101.
Liu, G., Yuan, X., Zeng, Z., Tunici, P., Ng, H., Abdulkadir, I. R., et al. (2006). Analysis of gene expression and chemoresistance of CD133+ cancer stem cells in glioblastoma. Molecular cancer, 5, 67.
Ma, S., Lee, T. K., Zheng, B. J., Chan, K. W., & Guan, X. Y. (2008). CD133+ HCC cancer stem cells confer chemoresistance by preferential expression of the Akt/PKB survival pathway. Oncogene, 27(12), 1749–1758.
Sparmann, A., & van Lohuizen, M. (2006). Polycomb silencers control cell fate, development and cancer. Nature Reviews Cancer, 6(11), 846–856.
Valk-Lingbeek, M. E., Bruggeman, S. W., & van Lohuizen, M. (2004). Stem cells and cancer; the polycomb connection. Cell, 118(4), 409–418.
Park, I. K., Qian, D., Kiel, M., Becker, M. W., Pihalja, M., Weissman, I. L., et al. (2003). Bmi-1 is required for maintenance of adult self-renewing haematopoietic stem cells. Nature, 423(6937), 302–305.
Molofsky, A. V., Pardal, R., Iwashita, T., Park, I. K., Clarke, M. F., & Morrison, S. J. (2003). Bmi-1 dependence distinguishes neural stem cell self-renewal from progenitor proliferation. Nature, 425(6961), 962–967.
Lessard, J., & Sauvageau, G. (2003). Bmi-1 determines the proliferative capacity of normal and leukaemic stem cells. Nature, 423(6937), 255–260.
Liu, S., Dontu, G., Mantle, I. D., Patel, S., Ahn, N. S., Jackson, K. W., et al. (2006). Hedgehog signaling and Bmi-1 regulate self-renewal of normal and malignant human mammary stem cells. Cancer Research, 66(12), 6063–6071.
Prince, M. E., Sivanandan, R., Kaczorowski, A., Wolf, G. T., Kaplan, M. J., Dalerba, P., et al. (2007). Identification of a subpopulation of cells with cancer stem cell properties in head and neck squamous cell carcinoma. Proceedings of the National Academy of Sciences of the United States of America, 104(3), 973–978.
Tan, T. T., Degenhardt, K., Nelson, D. A., Beaudoin, B., Nieves-Neira, W., Bouillet, P., et al. (2005). Key roles of BIM-driven apoptosis in epithelial tumors and rational chemotherapy. Cancer Cell, 7(3), 227–238.
Bouillet, P., Purton, J. F., Godfrey, D. I., Zhang, L. C., Coultas, L., Puthalakath, H., et al. (2002). BH3-only Bcl-2 family member Bim is required for apoptosis of autoreactive thymocytes. Nature, 415(6874), 922–926.
Richter-Larrea, J. A., Robles, E. F., Fresquet, V., Beltran, E., Rullan, A. J., Agirre, X., et al. (2010). Reversion of epigenetically mediated BIM silencing overcomes chemoresistance in Burkitt lymphoma. Blood, 116(14), 2531–2542.
Weir, H. K., Thun, M. J., Hankey, B. F., Ries, L. A., Howe, H. L., Wingo, P. A., et al. (2003). Annual report to the nation on the status of cancer, 1975–2000, featuring the uses of surveillance data for cancer prevention and control. Journal of the National Cancer Institute, 95(17), 1276–1299.
Dean, M., Fojo, T., & Bates, S. (2005). Tumour stem cells and drug resistance. Nature Reviews Cancer, 5(4), 275–284.
Acknowledgments
This work is supported by the National Natural Science Foundation of China (No.30973288), the Natural Science Foundation of Jilin Province of China (No.20130101151JC), and Doctoral Fund of Ministry of Education of China (No. 20120061120092).
Author information
Authors and Affiliations
Corresponding authors
Additional information
Dan Yu and Yan Liu have contributed equally to this work.
Rights and permissions
About this article
Cite this article
Yu, D., Liu, Y., Yang, J. et al. Clinical Implications of BMI-1 in Cancer Stem Cells of Laryngeal Carcinoma. Cell Biochem Biophys 71, 261–269 (2015). https://doi.org/10.1007/s12013-014-0194-z
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12013-014-0194-z