Abstract
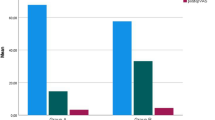
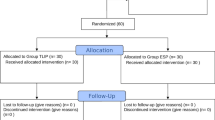
We prospectively studied the effectiveness of the repositioning suture of the erector spinae muscle for lumbar spine surgery using the posterior approach. 393 patients undergoing lumbar spine surgery were randomized to receive the repositioning or conventional suture of the erector spinae muscle. Time to stitch removal and drainage volume was recorded at 24 and 48 h after operation. Hemoglobin loss rate was determined at 48 h post operation and the rate of malunion (redness, swelling and effusion at stitch removal and would disruption after stitch removal) was recorded. Low back pain was evaluated using the visual analog scale (VAS) preoperatively and 6 and 12 months after operation. Time to stitch removal was comparable in lumbar spine surgery patients receiving the repositioning or conventional suture of the erector spinae muscle (P > 0.05). Compared with the conventional suture, the repositioning suture was associated with significantly reduced drainage volume both at 24 (P < 0.01) and 48 h after operation (P < 0.05). Hemoglobin loss rate at 48 h post operation was also markedly lower in lumbar spine surgery patients receiving the repositioning suture than in those receiving the conventional suture (P < 0.01 or 0.05). Furthermore, the malunion rate in lumbar spine surgery patients using the repositioning suture was markedly lower than that in the conventional group (P < 0.05 or 0.001). There was no difference in preoperative VAS scores in both the groups (P > 0.05). Compared with the conventional suture, the repositioning suture was associated with significantly reduced VAS scores both at 24 and 48 h after operation (P < 0.01 in both). The repositioning suture of the erector spinae muscle is superior to the conventional suture in posterior lumbar spine surgery with marked lessened pain and reduced drainage volume.

Similar content being viewed by others
References
Mahar, A., Kim, C., Wedemeyer, M., Mitsunaga, L., Odell, T., Johnson, B., et al. (2007). Short-segment fixation of lumbar burst fractures using pediele fixation at the level of the fracture. Spine, 32, 1503–1507.
Tezeren, G., & Kum, I. (2005). Posterior fixation of thoracolumbar burst fracture: Short-segment pedicle fixation versus long-segment instrumentation. Journal of Spinal Disorders and Techniques, 18, 485–488.
Apfel, C. C., Cakmakkaya, O. S., Martin, W., Richmond, C., Macario, A., George, E., et al. (2010). Restoration of disk height through non-surgical spinal decompression is associated with decreased discogenic low back pain: A retrospective cohort study. BMC Musculoskeletal Disorders, 11, 121–155.
Häkkinen, A., Ylinen, J., Kautiainen, H., Tarvainen, U., & Kiviranta, I. (2005). Effects of home strength training and stretching versus stretching alone after lumbar disk surgery: A randomized study with a 1-year follow-up. Archives of Physical Medicine and Rehabilitation, 86, 865–870.
Pappas, C. T., Harrington, T., & Sonntag, V. K. (1992). Outcome analysis in 654 surgically treated lumbar disc herniations. Neurosurgery, 30, 862–866.
Choi, G., Raiturker, P. P., Kim, M. J., Chung, D. J., Chae, Y. S., & Lee, S. H. (2005). The effect of early isolated lumbar extension exercise program for patients with herniated disc undergoing lumbar discectomy. Neurosurgery, 57, 764–772.
Long, C. J., Brown, D. A., & Engelberg, J. (1980). Intervertebral disc surgery: Strategies for patient selection to improve surgical outcome. Journal of Neurosurgery, 52, 818–824.
Manniche, C., Asmussen, K. H., Vinterberg, H., Rose-Hansen, E. B., Kramhøft, J., & Jordan, A. (1994). Analysis of preoperative prognosticfactors in first-time surgery for lumbar disc herniation, including Finneson’s and modified Spengler’s score systems. Danish Medical Bulletin, 41, 110–115.
Gray, H. (1980). Gray’s Anatomy (36th ed.). London: Warwick and Williams.
Cunningham, D. J. (1981). Textbook of Anatomy (12th ed.). London: Oxford University Press.
Morris, J. M., Lucas, D. B., & Breslar, B. (1962). Role of the trunk in stability of the spine. The Journal of bone and joint surgery, 43, 327–351.
Taylor, H., McGregor, A. H., Medhi-Zadeh, S., Richards, S., Kahn, N., Zadeh, J. A., et al. (2002). The impact of self-retaining retractors on the paraspinal muscles during posterior spinal surgery. Spine, 27, 2758–2762.
Kim, D. Y., Lee, S. H., Chung, S. K., & Lee, H. Y. (2009). Comparison of multifidus muscle atrophy and trunk extension muscle strength: Percutaneous versus open pedicle screw fixation. Spine, 30, 123–129.
Mayer, T. G., Vanharanta, H., Gatchel, R. J., Mooney, V., Barnes, D., Judge, L., et al. (1989). Comparison of CT scan muscle measurements and isokinetic trunk strength in postoperative patients. Spine, 14, 33–36.
Onesti, S. T. (2004). Failed back syndrome. Neurologist, 10, 259–264.
Laasonen, E. M. (1984). Atrophy of sacrospinal muscle groups in patients with chronic, diffusely radiating lumbar back pain. Neuroradiology, 26, 9–13.
Sihvonen, T., Herno, A., Paljärvi, L., Airaksinen, O., Partanen, J., & Tapaninaho, A. (1993). Local denervation atrophy of paraspinal muscles in postoperative failed back syndrome. Spine, 18, 575–581.
Motosuneya, T., Asazuma, T., Tsuji, T., Watanabe, H., Nakayama, Y., & Nemoto, K. (2006). Postoperative change of the cross-sectional area of back musculature after 5 surgical procedures as assessed by magnetic resonance imaging. Journal of Spinal Disorders and Techniques, 19, 318–322.
Schwender, J. D., Holly, L. T., Rouben, D. P., & Foley, K. T. (2005). Minimally invasive transforaminal lumbar interbody fusion (TLIF): Technical feasibility and initial results. Journal of Spinal Disorders and Techniques, 18(Suppl), S1–S6.
Standring, S. (2005). The back. In Gray’s anatomy: The anatomical basis of clinical practice (pp. 733–773). Philadelphia: Elsevier Churchill Livingstone.
Moseley, G. L., Hodges, P. W., & Gandevia, S. C. (2003). External perturbation of the trunk in standing humans differentially activates components of the medial back muscles. Journal of Physiology, 547, 581–587.
Dickx, Nele., Cagnie, Barbara., & Achten, Erik. (2010). Differentiation between deep and superficial fibers of the lumbar multifidus by magnetic resonance imaging. European Spine Journal, 19, 122–128.
Macintosh, J. E., Bogduk, N., & Munro, R. R. (1986). The morphology of the human lumbar multifidus. Clinical Biomechanics, 1, 196–204.
Macdonald, D. A., Lorimer Moseley, G., & Hodges, P. W. (2006). The lumbar multifidus: Does the evidence support clinical beliefs? Manual Therapy, 11, 254–263.
Macintosh, J. E., & Bogduk, N. (1986). The biomechanics of the lumbar multifidus. Clinical Biomechanics, 1, 205–213.
Ward, S. R., Kim, C. W., Eng, C. M., Gottschalk, L. J., 4th, Tomiya, A., Garfin, S. R., et al. (2009). Architectural analysis and intraoperative measurements demonstrate the unique design of the multifidus muscle for lumbar spine stability. Journal of Bone and Joint Surgery. American Volume, 91, 176–185.
Elain, M., Stanley, V., & Gerard, C. (2005). A morphological comparison of the human lumbar multifidus by chemical dissection. The journal of Manual & Manipulative Therapy, 16, 84–92.
Demoulin, C., Crielaard, J. M., & Vanderthommen, M. (2007). Spinal muscle evaluation in healthy individuals and low-back-pain patients: A literature review. Joint Bone Spine, 74, 9–13.
Kjaer, P., Tom, Bendix., Sorensen, J. S., Korsholm, L., & Leboeuf-Yde, C. (2007). Are MRI-defined fat infiltrations is associated with low back pain? BMC Medicine, 52, 1–10.
Conflict of interest
The authors declared no conflict of interest involved in the manuscript and agreed to publish this paper.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Wu, YJ., Fu, ZY., Hu, XP. et al. Repositioning Suture of the Erector Spinae Muscle for Lumbar Spine Surgery via the Posterior Approach: A Prospective Randomized Study. Cell Biochem Biophys 69, 75–80 (2014). https://doi.org/10.1007/s12013-013-9770-x
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12013-013-9770-x