Abstract
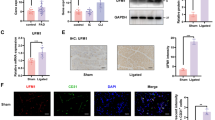
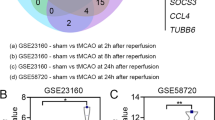
In our previous studies, the results have revealed that circRNA_102046 is significantly upregulated in plasma of patients with ischemic stroke, which closely related to NIHSS score. Human neural stem cells (hNSCs) were used for characterization and subcellular localization of circRNA_102046, and hNSCs OGD/R model was generated. The proliferation of cells was examined by CCK-8 assay. The expression levels of associated molecules were evaluated using RT-qPCR, immunofluorescence staining or western blotting. The binding and co-localization of associated molecules were also evaluated by RIP and FISH assay. Furthermore, MCAO mouse model was established to examine the effects of circRNA_102046 on the progression of ischemic stroke. Expression of circRNA_102046 was detected in the cytoplasma of hNSCs. Then OGD/R cell model was established, where the levels of circRNA_102046 was significantly up-regulated. Furthermore, knockdown of circRNA_102046 was able to enhance the proliferation and differentiation of OGD/R hNSCs. In further downstream molecular studies, the results indicated that circRNA_102046 could participate in the occurrence and development of ischemic stroke through targeting miR-493-5p. In addition, ROCK1 was identified as the putative target of miR-493-5p, and circRNA_102046 regulates the proliferation and differentiation of hNSCs via the miR-493-5p/ROCK1 signaling. More importantly, the infarct volumes of MCAO mice were remarkably reduced after the treatment with sh-circRNA_102046, which also up- and down-regulate the expression of miR-493-5p and ROCK1, respectively. Elucidating this novel pathway provides a theoretical basis for the development of new diagnostic approach and targeted treatment for ischemic stroke.





Similar content being viewed by others
Data Availability
The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.
References
Lancet, T. (2018). Stroke—Acting FAST at all ages. The Lancet, 391(10120), 514. https://doi.org/10.1016/S0140-6736(18)30240-X
Yao, Y., Liu, G., Wang, L., et al. (2019). Disease and disparity in China: A view from stroke and MI disease. International Journal for Equity in Health, 18(1), 15–85. https://doi.org/10.1186/s12939-019-0986-2
Yang, G. Y. (2019). Advancement in stroke research. Stroke and Vascular Neurology, 4(2), 61–62. https://doi.org/10.1136/svn-2019-000253
Malone, K., Amu, S., Moore, A. C., et al. (2019). The immune system and stroke: From current targets to future therapy. Immunology & Cell Biology, 97, 5–16. https://doi.org/10.1111/imcb.12191
Mehta, S. L., Pandi, G., & Vemuganti, R. (2017). Circular RNA expression profiles alter significantly in mouse brain after transient focal ischemia. Stroke, 48, 117–17469. https://doi.org/10.1161/STROKEAHA.117.017469
Knupp, D., & Miura, P. (2018). CircRNA accumulation: A new hallmark of aging? Mechanisms of Ageing & Development, 173, 71–79. https://doi.org/10.1016/j.mad.2018.05.001
Humphreys, D. T., Nicolas, F., Madeleine, D., et al. (2019). Ularcirc: Visualization and enhanced analysis of circular RNAs via back and canonical forward splicing. Nucleic Acids Research, 47(20), 20. https://doi.org/10.1093/nar/gkz718
Dong, Z., Deng, L., Peng, Q., et al. (2019). CircRNA expression profiles and function prediction in peripheral blood mononuclear cells of patients with acute ischemic stroke. Journal of Cellular Physiology. https://doi.org/10.1002/jcp.29165
Bazan, H. A., Hatfield, S. A., Brug, A., et al. (2017). Carotid plaque rupture is accompanied by an increase in the ratio of serum circR-284 to miR-221 levels. Clinical Perspective. Circulation Cardiovascular Genetics, 10(4), e1720. https://doi.org/10.1161/CIRCGENETICS.117.001720
Ostolaza, A., Blanco-Luquin, I., Urdánoz-Casado, A., et al. (2020). Circular RNA expression profile in blood according to ischemic stroke etiology. Cell & Bioscience, 10, 34. https://doi.org/10.1186/s13578-020-00394-3
Wang, G., Han, B., Shen, L., et al. (2020). Silencing of circular RNA HIPK2 in neural stem cells enhances functional recovery following ischaemic stroke. EBioMedicine, 52, 102660. https://doi.org/10.1016/j.ebiom.2020.102660
Zhu, X., Ding, J., Wang, B., Wang, J., & Xu, M. (2019). Circular RNA DLGAP4 is down-regulated and negatively correlates with severity, inflammatory cytokine expression and pro-inflammatory gene miR-143 expression in acute ischemic stroke patients. International Journal of Clinical and Experimental Pathology, 12(3), 941–948. https://doi.org/10.3390/jpm13050839
Zang, J., Lu, D., & Xu, A. (2020). The interaction of circRNAs and RNA binding proteins: An important part of circRNA maintenance and function. Journal of Neuroscience Research, 98(1), 87–97. https://doi.org/10.1002/jnr.24356
Wang, A., Jin, C., Li, H., et al. (2018). LncRNA ADAMTS9-AS2 regulates ovarian cancer progression by targeting miR-182-5p/FOXF2 signaling pathway. International Journal of Biological Macromolecules, 120(Pt B), 1705–1713. https://doi.org/10.1016/j.ijbiomac.2018.09.179
Mu, Y., Li, N., & Cui, Y. L. (2018). The lncRNA CCAT1 upregulates TGFbetaR1 via sponging miR-490-3p to promote TGFbeta1-induced EMT of ovarian cancer cells. Cancer Cell International, 18, 145. https://doi.org/10.1111/1759-7714.13758
Zhu, R., Wang, Q. W., Zhao, J., Liu, X., & He, Z. (2020). miR-149 and miR-499 gene polymorphism and the incident of ischemic stroke in the Asian population: From a case-control study to meta-analysis. Clinical Neurology and Neurosurgery, 193, 105789. https://doi.org/10.1016/j.clineuro.2020.105789
Sun, P., Zhang, K., Hassan, S. H., Zhang, X., Tang, X., Pu, H., Stetler, R. A., Chen, J., & Yin, K. J. (2020). Endothelium-targeted deletion of microRNA-15a/16-1 promotes post-stroke angiogenesis and improves long-term neurological recovery. Circulation Research. https://doi.org/10.1161/CIRCRESAHA.119.315886
Li, Q., He, Q., Baral, S., Mao, L., Li, Y., Jin, H., Chen, S., An, T., Xia, Y., & Hu, B. (2016). MicroRNA-493 regulates angiogenesis in a rat model of ischemic stroke by targeting MIF. The FEBS Journal, 283(9), 1720–1733. https://doi.org/10.1111/febs.13697
Tuladhar, A., Obermeyer, J. M., Payne, S. L., Siu, R. C. W., Zand, S., Morshead, C. M., & Shoichet, M. S. (2020). Injectable hydrogel enables local and sustained co-delivery to the brain: Two clinically approved biomolecules, cyclosporine and erythropoietin, accelerate functional recovery in rat model of stroke. Biomaterials, 235, 119794. https://doi.org/10.1016/j.biomaterials.2020.119794
Hc, T., Jh, W., Ya, C., et al. (2020). A self-assembled layer-by-layer surface modification to fabricate the neuron- rich model from neural stem/precursor cells. Journal of the Formosan Medical Association = Taiwan yi zhi, 119, 430–438. https://doi.org/10.1016/j.jfma.2019.08.004
Wu, F., Han, B., Wu, S., Yang, L., Leng, S., Li, M., Liao, J., Wang, G., Ye, Q., Zhang, Y., Chen, H., Chen, X., Zhong, M., Xu, Y., Liu, Q., Zhang, J. H., & Yao, H. (2019). TLK1 circular RNA aggravates neuronal injury and neurological deficits after ischemic stroke via miR-335-3p/TIPARP. The Journal of Neuroscience, 39(37), 7369–7393. https://doi.org/10.1523/JNEUROSCI.0299-19.2019
Zhao, F., Xing, Y., Jiang, P., et al. (2021). LncRNA MEG3 inhibits the proliferation of neural stem cells after ischemic stroke via the miR-493-5P/MIF axis. Biochemical and Biophysical Research Communications, 568, 186–192. https://doi.org/10.1016/j.bbrc.2021.06.033
Niu, M., Li, H., Li, X., et al. (2021). Circulating exosomal miRNAs as novel biomarkers perform superior diagnostic efficiency compared with plasma miRNAs for large-artery atherosclerosis stroke. Frontiers in Pharmacology, 12, 791644. https://doi.org/10.3389/fphar.2021.791644
Lin, Y., Zhao, Z., Hu, M., et al. (2023). Inhibition of interaction between ROCK1 and Rubicon restores autophagy in endothelial cells and attenuates brain injury after prolonged ischemia. Journal of Neurochemistry, 164(2), 172–192. https://doi.org/10.1111/jnc.15721
Acknowledgements
None.
Funding
This project was funded by Natural Science Foundation of Chongqing (Grant No. cstc2020jcyj-msxmX0358).
Author information
Authors and Affiliations
Contributions
CY designed the study, and all authors carried out the experiments and data analysis. All authors drafted the manuscript and approved the final version of the paper.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical Approval
This study was approved by the Ethics Committee of the third affiliated hospital of Chongqing Medical University (Chongqing, China).
Consent for Publication
Not applicable.
Additional information
Handling Editor: Y. Robert Li.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
He, W., Cheng, Y. & Lai, Y. CircRNA_102046 Affects the Occurrence and Development of Ischemic Stroke by Regulating the miR-493-5p/ROCK1 Signaling. Cardiovasc Toxicol 24, 280–290 (2024). https://doi.org/10.1007/s12012-024-09831-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12012-024-09831-y