Abstract
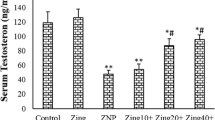
Zinc (Zn) is an important trace element in the human body and plays an important role in growth, development, and male reproductive functions. Marginal zinc deficiency (MZD) is common in the human population and can cause spermatogenic dysfunction in males. Therefore, the aim of this study was to investigate methods to improve spermatogenic dysfunction caused by MZD and to further explore its mechanism of action. A total of 75 4-week-old male SPF ICR mice were randomly divided into five groups (control, MZD, MZD + ZnY2, MZD + ZnY4, and MZD + ZnY8, 15 mice per group). The dietary Zn content was 30 mg/kg in the control group and 10 mg/kg in the other groups. From low to high, the Zn supplementation doses administered to the three groups were 2, 4, and 8 mg/kg·bw. After 35 days, the zinc content, sperm quality, activity of spermatogenic enzymes, oxidative stress level, and apoptosis level of the testes in mice were determined. The results showed that MZD decreased the level of Zn in the serum, sperm quality, and activity of spermatogenic enzymes in mice. After Zn supplementation, the Zn level in the serum increased, sperm quality was significantly improved, and spermatogenic enzyme activity was restored. In addition, MZD reduced the content of antioxidants (copper-zinc superoxide dismutase (Cu–Zn SOD), metallothionein (MT), and glutathione (GSH) and promoted malondialdehyde (MDA) production. The apoptosis index of the testis also increased significantly in the MZD group. After Zn supplementation, the level of oxidative stress decreased, and the apoptosis index in the testis was reduced. Furthermore, quantitative real-time polymerase chain reaction (qRT–PCR) showed that the expression of B-cell lymphoma-2 (Bcl-2) mRNA and Bcl-2/BCL2-associated X (Bax) in the control group decreased in testicular cells, and their expression was restored after Zn supplementation. The results of this study indicated that Zn supplementation can reduce the level of oxidative stress and increase the ability of testicular cells to resist apoptosis, thereby improving spermatogenic dysfunction caused by MZD in mice.








Similar content being viewed by others
Data Availability
All data presented in the main manuscript.
References
Stefanidou M, Maravelias C, Dona A, Spiliopoulou C (2006) Zinc: a multipurpose trace element. Arch Toxicol 80(1):1–9. https://doi.org/10.1007/s00204-005-0009-5
Baltaci AK, Mogulkoc R, Baltaci SB (2019) Review: the role of zinc in the endocrine system. Pak J Pharm Sci 32(1):231–239
Vickram S, Rohini K, Srinivasan S, Nancy VD, Archana K, Anbarasu K, Jeyanthi P, Thanigaivel S, Gulothungan G, Rajendiran N, Srikumar PS (2021) Role of Zinc (Zn) in human reproduction: a journey from initial spermatogenesis to childbirth. Int J Mol Sci 22(4). https://doi.org/10.3390/ijms22042188
Prasad AS (2009) Zinc: role in immunity, oxidative stress and chronic inflammation. Curr Opin Clin Nutr Metab Care 12(6):646–652. https://doi.org/10.1097/MCO.0b013e3283312956
Wessells KR, Brown KH (2012) Estimating the global prevalence of zinc deficiency: results based on zinc availability in national food supplies and the prevalence of stunting. PLoS One 7(11):e50568. https://doi.org/10.1371/journal.pone.0050568
Caro CR, Del CCM, Arrollo J, Martinez G, Majana LS, Sarmiento-Rubiano LA (2016) Zinc deficiency: a global problem that affect the health and cognitive development. Arch Latinoam Nutr 66(3):165–175
Jinno N, Nagata M, Takahashi T (2014) Marginal zinc deficiency negatively affects recovery from muscle injury in mice. Biol Trace Elem Res 158(1):65–72. https://doi.org/10.1007/s12011-014-9901-2
Colagar AH, Marzony ET, Chaichi MJ (2009) Zinc levels in seminal plasma are associated with sperm quality in fertile and infertile men. Nutr Res 29(2):82–88. https://doi.org/10.1016/j.nutres.2008.11.007
Merrells KJ, Blewett H, Jamieson JA, Taylor CG, Suh M (2009) Relationship between abnormal sperm morphology induced by dietary zinc deficiency and lipid composition in testes of growing rats. Br J Nutr 102(2):226–232. https://doi.org/10.1017/S0007114508159037
Allouche-Fitoussi D, Breitbart H (2020) The role of zinc in male fertility. Int J Mol Sci 21(20). https://doi.org/10.3390/ijms21207796
Nishito Y, Kambe T (2018) Absorption mechanisms of iron, copper, and zinc: an overview. J Nutr Sci Vitaminol (Tokyo) 64(1):1–7. https://doi.org/10.3177/jnsv.64.1
Maret W, Sandstead HH (2006) Zinc requirements and the risks and benefits of zinc supplementation. J Trace Elem Med Biol 20(1):3–18. https://doi.org/10.1016/j.jtemb.2006.01.006
Ganger R, Garla R, Mohanty BP, Bansal MP, Garg ML (2016) Protective effects of zinc against acute arsenic toxicity by regulating antioxidant defense system and cumulative metallothionein expression. Biol Trace Elem Res 169(2):218–229. https://doi.org/10.1007/s12011-015-0400-x
Ho E, Ames BN (2002) Low intracellular zinc induces oxidative DNA damage, disrupts p53, NFkappa B, and AP1 DNA binding, and affects DNA repair in a rat glioma cell line. Proc Natl Acad Sci USA 99(26):16770–16775. https://doi.org/10.1073/pnas.222679399
Sakaguchi S, Iizuka Y, Furusawa S, Ishikawa M, Satoh S, Takayanagi M (2002) Role of Zn(2+) in oxidative stress caused by endotoxin challenge. Eur J Pharmacol 451(3):309–316. https://doi.org/10.1016/s0014-2999(02)02223-9
Zhu X, Yu C, Wu W, Shi L, Jiang C, Wang L, Ding Z, Liu Y (2022) Zinc transporter ZIP12 maintains zinc homeostasis and protects spermatogonia from oxidative stress during spermatogenesis. Reprod Biol Endocrinol 20(1):17. https://doi.org/10.1186/s12958-022-00893-7
Chen Y, Yang J, Wang Y, Yang M, Guo M (2020) Zinc deficiency promotes testicular cell apoptosis in mice. Biol Trace Elem Res 195(1):142–149. https://doi.org/10.1007/s12011-019-01821-4
D’Arcy MS (2019) Cell death: a review of the major forms of apoptosis, necrosis and autophagy. Cell Biol Int 43(6):582–592. https://doi.org/10.1002/cbin.11137
Beigi HA, Dahan H, Tahmasbpour E, Bakhtiari KH, Shahriary A (2020) Effects of zinc deficiency on impaired spermatogenesis and male infertility: the role of oxidative stress, inflammation and apoptosis. Hum Fertil (Camb) 23(1):5–16. https://doi.org/10.1080/14647273.2018.1494390
Kumari D, Nair N, Bedwal RS (2011) Testicular apoptosis after dietary zinc deficiency: ultrastructural and TUNEL studies. Syst Biol Reprod Med 57(5):233–243. https://doi.org/10.3109/19396368.2011.584500
Zhang Z, Cheng Q, Liu Y, Peng C, Wang Z, Ma H, Liu D, Wang L, Wang C (2022) Zinc-enriched yeast may improve spermatogenesis by regulating steroid production and antioxidant levels in mice. Biol Trace Elem Res 200(8):3712–3722. https://doi.org/10.1007/s12011-021-02970-1
Peng C, Cheng Q, Liu Y, Zhang Z, Wang Z, Ma H, Liu D, Wang L, Wang C (2022) Marginal zinc deficiency in mice increased the number of abnormal sperm and altered the expression level of spermatogenesis-related genes. Biol Trace Elem Res 200(8):3738–3749. https://doi.org/10.1007/s12011-021-02979-6
Adamo AM, Liu X, Mathieu P, Nuttall JR, Supasai S, Oteiza PI (2019) Early developmental marginal zinc deficiency affects neurogenesis decreasing neuronal number and altering neuronal specification in the adult rat brain. Front Cell Neurosci 13:62. https://doi.org/10.3389/fncel.2019.00062
Croxford TP, Mccormick NH, Kelleher SL (2011) Moderate zinc deficiency reduces testicular Zip6 and Zip10 abundance and impairs spermatogenesis in mice. J Nutr 141(3):359–365. https://doi.org/10.3945/jn.110.131318
Iwaya H, Kashiwaya M, Shinoki A, Lee JS, Hayashi K, Hara H, Ishizuka S (2011) Marginal zinc deficiency exacerbates experimental colitis induced by dextran sulfate sodium in rats. J Nutr 141(6):1077–1082. https://doi.org/10.3945/jn.111.138180
China NHCO (2017) Dietary nutrient reference intake of Chinese residents. http://www.nhc.gov.cn/wjw/yingyang/201710/ef2d42ee35894a46b7726457d08d7e2d.shtml. Accessed 2023/4/20
Sarkar D, Chowdhury JP, Singh SK (2016) Effect of polybrominated diphenyl ether (BDE-209) on testicular steroidogenesis and spermatogenesis through altered thyroid status in adult mice. Gen Comp Endocrinol 239:50–61. https://doi.org/10.1016/j.ygcen.2015.11.009
Duan P, Hu C, Butler HJ, Quan C, Chen W, Huang W, Tang S, Zhou W, Yuan M, Shi Y, Martin FL, Yang K (2017) 4-Nonylphenol induces disruption of spermatogenesis associated with oxidative stress-related apoptosis by targeting p53-Bcl-2/Bax-Fas/FasL signaling. Environ Toxicol 32(3):739–753. https://doi.org/10.1002/tox.22274
Menkveld R (2010) Clinical significance of the low normal sperm morphology value as proposed in the fifth edition of the WHO laboratory manual for the examination and processing of human semen. Asian J Androl 12(1):47–58. https://doi.org/10.1038/aja.2009.14
Wood RJ (2000) Assessment of marginal zinc status in humans. J Nutr 130(5S Suppl):1350S-1354S. https://doi.org/10.1093/jn/130.5.1350S
Yokokawa H, Fukuda H, Saita M, Miyagami T, Takahashi Y, Hisaoka T, Naito T (2020) Serum zinc concentrations and characteristics of zinc deficiency/marginal deficiency among Japanese subjects. J Gen Fam Med 21(6):248–255. https://doi.org/10.1002/jgf2.377
Gaulke CA, Rolshoven J, Wong CP, Hudson LG, Ho E, Sharpton TJ (2018) Marginal zinc deficiency and environmentally relevant concentrations of arsenic elicit combined effects on the gut microbiome. mSphere 3(6). https://doi.org/10.1128/mSphere.00521-18
Huang YL, Supasai S, Kucera H, Gaikwad NW, Adamo AM, Mathieu P, Oteiza PI (2016) Nutritional marginal zinc deficiency disrupts placental 11beta-hydroxysteroid dehydrogenase type 2 modulation. Food Funct 7(1):84–92. https://doi.org/10.1039/c5fo01203a
Nuttall JR, Supasai S, Kha J, Vaeth BM, Mackenzie GG, Adamo AM, Oteiza PI (2015) Gestational marginal zinc deficiency impaired fetal neural progenitor cell proliferation by disrupting the ERK1/2 signaling pathway. J Nutr Biochem 26(11):1116–1123. https://doi.org/10.1016/j.jnutbio.2015.05.007
Disilvestro RA, Dardenne M, Joseph E (2021) Comparison of thymulin activity with other measures of marginal zinc deficiency. Biol Trace Elem Res 199(2):585–587. https://doi.org/10.1007/s12011-020-02159-y
Hess SY, Peerson JM, King JC, Brown KH (2007) Use of serum zinc concentration as an indicator of population zinc status. Food Nutr Bull 28(3 Suppl):S403–S429. https://doi.org/10.1177/15648265070283S303
King JC, Shames DM, Woodhouse LR (2000) Zinc homeostasis in humans. J Nutr 130(5S Suppl):1360S-1366S. https://doi.org/10.1093/jn/130.5.1360S
Dimitrova AA, Strashimirov D, Betova T, Russeva A, Alexandrova M (2008) Zinc content in the diet affects the activity of Cu/ZnSOD, lipid peroxidation and lipid profile of spontaneously hypertensive rats. Acta Biol Hung 59(3):305–314. https://doi.org/10.1556/ABiol.59.2008.3.4
Kimura T, Kambe T (2016) The functions of metallothionein and ZIP and ZnT transporters: an overview and perspective. Int J Mol Sci 17(3):336. https://doi.org/10.3390/ijms17030336
Yu J, Fujishiro H, Miyataka H, Oyama TM, Hasegawa T, Seko Y, Miura N, Himeno S (2009) Dichotomous effects of lead acetate on the expression of metallothionein in the liver and kidney of mice. Biol Pharm Bull 32(6):1037–1042. https://doi.org/10.1248/bpb.32.1037
Justus J, Weigand E (2014) The effect of a moderate zinc deficiency and dietary fat source on the activity and expression of the Delta(3)Delta (2)-enoyl-CoA isomerase in the liver of growing rats. Biol Trace Elem Res 158(3):365–375. https://doi.org/10.1007/s12011-014-9940-8
Mahfouz RZ, du Plessis SS, Aziz N, Sharma R, Sabanegh E, Agarwal A (2010) Sperm viability, apoptosis, and intracellular reactive oxygen species levels in human spermatozoa before and after induction of oxidative stress. Fertil Steril 93(3):814–821. https://doi.org/10.1016/j.fertnstert.2008.10.068
Chia SE, Ong CN, Chua LH, Ho LM, Tay SK (2000) Comparison of zinc concentrations in blood and seminal plasma and the various sperm parameters between fertile and infertile men. J Androl 21(1):53–57
Osadchuk LV, Danilenko AD, Osadchuk AV (2021) An influence of zinc on male infertility. Urologiia 5:84–93
Piechal A, Blecharz-Klin K, Pyrzanowska J, Widy-Tyszkiewicz E (2016) Influence of long-term zinc administration on spatial learning and exploratory activity in rats. Biol Trace Elem Res 172(2):408–418. https://doi.org/10.1007/s12011-015-0597-8
Casper RF, Meriano JS, Jarvi KA, Cowan L, Lucato ML (1996) The hypo-osmotic swelling test for selection of viable sperm for intracytoplasmic sperm injection in men with complete asthenozoospermia. Fertil Steril 65(5):972–976. https://doi.org/10.1016/s0015-0282(16)58271-5
Jeyendran RS, Van der Ven HH, Perez-Pelaez M, Crabo BG, Zaneveld LJ (1984) Development of an assay to assess the functional integrity of the human sperm membrane and its relationship to other semen characteristics. J Reprod Fertil 70(1):219–228. https://doi.org/10.1530/jrf.0.0700219
Zhang B, Lin S (2009) Effects of 3,4-dichloroaniline on testicle enzymes as biological markers in rats. Biomed Environ Sci 22(1):40–43. https://doi.org/10.1016/S0895-3988(09)60020-9
Hodgen GD, Sherins RJ (1973) Enzymes as markers of testicular growth and development in the rat. Endocrinology 93(4):985–989. https://doi.org/10.1210/endo-93-4-985
Turgut G, Abban G, Turgut S, Take G (2003) Effect of overdose zinc on mouse testis and its relation with sperm count and motility. Biol Trace Elem Res 96(1–3):271–279. https://doi.org/10.1385/BTER:96:1-3:271
Sirisena D, Gayashani SW, Neranjan TM, Madusanka RK, Jeong JB, Lee J (2021) A copper-zinc-superoxide dismutase (CuZnSOD) from redlip mullet, Liza haematocheila: insights to its structural characteristics, immune responses, antioxidant activity, and potent antibacterial properties. Dev Comp Immunol 123:104165. https://doi.org/10.1016/j.dci.2021.104165
Ruttkay-Nedecky B, Nejdl L, Gumulec J, Zitka O, Masarik M, Eckschlager T, Stiborova M, Adam V, Kizek R (2013) The role of metallothionein in oxidative stress. Int J Mol Sci 14(3):6044–6066. https://doi.org/10.3390/ijms14036044
Blokhina O, Virolainen E, Fagerstedt KV (2003) Antioxidants, oxidative damage and oxygen deprivation stress: a review. Ann Bot 91(2):179–194. https://doi.org/10.1093/aob/mcf118
Semercioz A, Baltaci AK, Mogulkoc R, Avunduk MC (2017) Effect of zinc and melatonin on oxidative stress and serum inhibin-B levels in a rat testicular torsion-detorsion model. Biochem Genet 55(5–6):395–409. https://doi.org/10.1007/s10528-017-9826-5
Amara S, Abdelmelek H, Garrel C, Guiraud P, Douki T, Ravanat JL, Favier A, Sakly M, Ben RK (2008) Preventive effect of zinc against cadmium-induced oxidative stress in the rat testis. J Reprod Dev 54(2):129–134. https://doi.org/10.1262/jrd.18110
Aitken RJ, Roman SD (2008) Antioxidant systems and oxidative stress in the testes. Oxid Med Cell Longev 1(1):15–24. https://doi.org/10.4161/oxim.1.1.6843
Wang Z, Peng C, Zhang Y, Wang L, Yu L, Wang C (2023) Characteristics of Zn content and localization, Cu-Zn SOD, and MT levels in the tissues of marginally Zn-deficient mice. Biol Trace Elem Res 201(1):262–271. https://doi.org/10.1007/s12011-022-03119-4
Alibek K, Irving S, Sautbayeva Z, Kakpenova A, Bekmurzayeva A, Baiken Y, Imangali N, Shaimerdenova M, Mektepbayeva D, Balabiyev A, Chinybayeva A (2014) Disruption of Bcl-2 and Bcl-xL by viral proteins as a possible cause of cancer. Infect Agent Cancer 9:44. https://doi.org/10.1186/1750-9378-9-44
Delbridge AR, Grabow S, Strasser A, Vaux DL (2016) Thirty years of BCL-2: translating cell death discoveries into novel cancer therapies. Nat Rev Cancer 16(2):99–109. https://doi.org/10.1038/nrc.2015.17
Dewson G, Ma S, Frederick P, Hockings C, Tan I, Kratina T, Kluck RM (2012) Bax dimerizes via a symmetric BH3:groove interface during apoptosis. Cell Death Differ. 19(4):661–70. https://doi.org/10.1038/cdd.2011.138
Fan TJ, Han LH, Cong RS, Liang J (2005) Caspase family proteases and apoptosis. Acta Biochim Biophys Sin (Shanghai) 37(11):719–27. https://doi.org/10.1111/j.1745-7270.2005.00108.x
Xu Y, Li A, Li X, Deng X, Gao XJ (2023) Zinc deficiency induces inflammation and apoptosis via oxidative stress in the kidneys of mice. Biol Trace Elem Res 201(2):739–750. https://doi.org/10.1007/s12011-022-03166-x
Pourhassanali N, Roshan-Milani S, Kheradmand F, Motazakker M, Bagheri M, Saboory E (2016) Zinc attenuates ethanol-induced Sertoli cell toxicity and apoptosis through caspase-3 mediated pathways. Reprod Toxicol 61:97–103. https://doi.org/10.1016/j.reprotox.2016.03.041
Acknowledgements
Thanks to all members of Professor Wang CH’s lab at the School of Public Health, Wuhan University for their generous help. We would like to thank AJE for its professional support in the help of the language polish. At the same time, thanks to the animal facilities of Wuhan University Animal Experiment Center for the maintenance of the mouse population.
Funding
This study was supported by grants from Angel Nutritech Nutrition Fund (Grant No. AF2019004).
Author information
Authors and Affiliations
Contributions
Chunhong Wang and Xiangchao Zeng designed this study. Ziqiong Wang, Lu Yu, Lei Wang, Yueling Liu, and Yuxin Chen raised the animals. Xiangchao Zeng and Ziqiong Wang carried out index detection and wrote the first draft. Chunhong Wang, Lu Yu, and Lei Wang edited the final paper. All authors approved the final version.
Corresponding author
Ethics declarations
Ethics Approval
This study was approved by Committee of the Ethics of Animal Experiments of the Wuhan University School of Medicine (No.: WP2020-08053).
Consent to Participate
Not applicable.
Consent for Publication
Not applicable.
Conflict of Interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zeng, X., Wang, Z., Yu, L. et al. Zinc Supplementation Reduces Testicular Cell Apoptosis in Mice and Improves Spermatogenic Dysfunction Caused by Marginal Zinc Deficiency. Biol Trace Elem Res 202, 1656–1668 (2024). https://doi.org/10.1007/s12011-023-03789-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12011-023-03789-8