Abstract
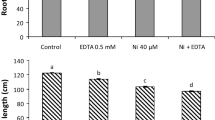
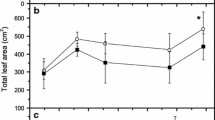
Yerba mate (Ilex paraguariensis St. Hill.) has shown a relatively high capacity for micronutrient absorption and could be a candidate for biofortification and combating a lack of micronutrients. To further evaluate the accumulation capacity of Ni and Zn, yerba mate clonal seedlings were grown in containers under five rates of Ni or Zn (0, 0.5, 2, 10, and 40 mg kg−1) with three soils originating from different parent material (basalt, rhyodacite, and sandstone). After 10 months, plants were harvested, divided into component parts (leaves, branches, and roots), and evaluated for 12 elements. The use of Zn and Ni enhanced seedling growth under rhyodacite- and sandstone-derived soils at the first application rate. Application of Zn and Ni resulted in linear increases based on Mehlich I extractions; recovery of Ni was smaller than Zn. Root Ni concentration increased from approximately 20 to 1000 mg kg−1 in rhyodacite-derived soil and from 20 to 400 mg kg−1 in basalt- and sandstone-derived soils; respective increases in leaf tissue were ~ 3 to 15 mg kg−1 and 3 to 10 mg kg−1. For Zn, the maximum obtained values were close to 2000, 1000, and 800 mg kg−1 for roots, leaves, and branches for rhyodacite-derived soils, respectively. Corresponding values for basalt- and sandstone-derived soils were 500, 400, and 300 mg kg−1, respectively. Although yerba mate is not a hyperaccumulator, this species has a relatively high capacity to accumulate Ni and Zn in young tissue with the highest accumulation occurring in roots. Yerba mate showed high potential to be used in biofortification programs for Zn.









Similar content being viewed by others
Data Availability
All data generated or analyzed during this study are included in this published article [and its supplementary information files].
References
IBGE - Instituto Brasileiro de Geografia e Estatística. Dados PAM e PEVS. Available in: https://sidra.ibge.gov.br/home/pms/brasil. Accessed 20 July 2022
Ministério da Indústria, Comércio Exterior e Serviços. Available in: http://www.mdic.gov.br/index.php/comercioexterior. Accessed 10 July 2022
Mendoza, J (2020) Annual per capita consumption of yerba mate in selected countries in Latin America as of July 2018. Available in: https://www.statista.com/statistics/884925/consumption-yerba-mateper-capita-latin-america/#statisticContainer. Accessed 27 Sept 2022
DERAL- Departamento de economia Rural (Secretaria da Agricultura). Prognóstico Erva-mate. Available in: https://www.agricultura.pr.gov.br. Accessed 28 Aug 2022
Bhutta ZA, Salam RA (2012) Global nutrition epidemiology and trends. Ann Nutr Metab 61(Suppl. 1):19–27
Kumssa DB, Joy EJ, Ander EL, Watts MJ, Young SD, Walker S, Broadley MR (2015) Dietary calcium and zinc deficiency risks are decreasing but remain prevalent. Sci Rep 5(1):1–11
Ulbrich NCM, do Prado LL, Barbosa JZ, Araujo EM, Poggere G, Motta ACV, Prior SA, Magri E, Young SD, Broadley MR (2022) Multi-elemental analysis and health risk assessment of commercial yerba mate from Brazil. Biol Trace Elem Res 200(3):1455–1463. https://doi.org/10.1007/s12011-021-02736-9
Magri E, Valduga AT, Gonçalves IL et al (2021) Cadmium and lead concentrations in yerba mate leaves from agroforestry and plantation systems: an international survey in South America. J Food Compos Anal 96:103702. https://doi.org/10.1016/j.jfca.2020.103702
Motta ACV, Barbosa JZ, Magri E, Pedreira GQ, Santin D, Prior SA, Consalter R, Young SD, Broadley MR, Benedetti EL (2020) Elemental composition of yerba mate (Ilex paraguariensis A.St.-Hil.) under low input systems of southern Brazil. Sci Total Environ 736:139637. https://doi.org/10.1016/j.scitotenv.2020.139637
Magri E, Barboza JZ, Corrêa RS, Auler AC, Valduga AT, Motta ACV (2022) Linking edaphoclimatic conditions with elementary composition of yerba mate leaves in South America. J Food Compos Anal 107:104360. https://doi.org/10.1016/j.jfca.2021.104360
Kabata-Pendias A (2011) Trace elements in soils and plants, 4th edn. Taylor & Francis, London, New York, p 505
Marques JJ, Burille C, Schuster C, Brandoli MAA, Rocha D, Bresolin B, Rodrigues P, Cardoso D, Brock M, Mallmann A, Silveira JS da (2013) Erva-mate: guia para aplicação de boas práticas agrícolas. Lajeado, RS: Emater/RS-Ascar, 80
Benedetti EL, Santin D, de Barros NF, Pereira GL, Martinez HP, Neves JCL (2017) Alumínio estimula o crescimento radicular de erva-mate? Pesq Flor Bras 37(90):139–147. https://doi.org/10.4336/2017.pfb.37.90.983
Magri E, Gugelmin EK, Gragarski FAP, Barbosa JZ, Auler AC, Wendling I, Prior SA, Valduga AT, Motta ACV (2020) Manganese hyperaccumulation capacity of Ilex paraguariensis A. St. Hil. and occurrence of interveinal chlorosis induced by transient toxicity. Ecotoxicol Environ Safety 203:111010. https://doi.org/10.1016/j.ecoenv.2020.111010
Barbosa JZ, Motta ACV, Consalter R, Poggere GC, Santin D, Wendling I (2018) Plant growth, nutrients and potentially toxic elements in leaves of yerba mate clones in response to phosphorus in acid soils. An Acad Bras Ciênc 90(1):557–571. https://doi.org/10.1590/0001-3765201820160701
Toppel FV, Junior AM, Motta ACV, Frigo C, Magri E, Barbosa JZ (2018) Soil chemical attributes and their influence on elemental composition of yerba mate leaves. Floresta 48(3):425–434. https://doi.org/10.5380/rf.v48i3.56677
Pozebon D, Dressler VL, Marcelo MCA et al (2015) Toxic and nutrient elements in yerba mate (Ilex paraguariensis). Food Addit Contam Part B Surveill 8(3):215–220. https://doi.org/10.1080/19393210.2015.1053420
Frigo C, Magri E, Barbosa JZ, Sarteretto LM, Araujo EM, de Melo VF, Prior SA, Motta ACV (2020) Influence of roadways on heavy metal content in soils and yerba mate tissue in southern Brazil. Manag Environ Qual 31(6):1477–1495. https://doi.org/10.1108/MEQ-10-2019-0219
da Marques AC, dos Reis MS, Denardin VF (2019) As paisagens da erva-mate: uso das florestas e conservação socioambiental. Ambiente and Sociedade 22:02822
Reis AR dos, Rodak BW, Putti FF, Moraes MF de (2014) Papel fisiológico do níquel: essencialidade e toxidez em plantas. Encarte de Informações Agronômicas 147. Available in: http://www.ipni.net/publication/iabrasil.nsf/0/0A37B421DA0EA5F383257D660046D009/$FILE/Page10-24-147.pdf. Accessed 14 Dec 2021
Seenivasan S, Anderson TA, Muraleedharan N (2016) Heavy metal content in tea soils and their distribution in different parts of tea plants, Camellia sinensis (L). O. Kuntze. Environ Monit Assess 188(7):428. https://doi.org/10.1007/s10661-016-5440-y
Marques R, Motta ACV (2003) Analise química do solo para fins de fertilidade. In: de Lima MR (ed) Manual de diagnóstico da fertilidade e manejo dos solos agrícolas, 2nd edn. Departamento de Solos e Engenharia Agrícola, Curitiba, pp 81–102
Dane JH, Topp CG, Campbell GS (2002) Particle size analysis. In: Methods of Soil Analysis. Part 4: Physical Methods, 5. Soil Science Society of America Book Series 255-293
Wendling I, Santin D, Nagaoka R, Sturion JA (2017) BRS BLD Aupaba e BRS BLD Yari: cultivares clonais de erva-mate para produção de massa foliar de sabor suave. Embrapa Florestas - Comunicado Técnico, ISSN 1980-3982, Colombo, PR. Available in: https://www.embrapa.br/busca-de-publicacoes/-/publicacao/1083388/brs-bld-aupaba-e-brs-bld-yari-cultivares-clonais-de-erva-mate-para-producao-de-massa-foliar-de-sabor-suave. Accessed 15 Jan 2020
Martins APL, Reissmann CB (2007) Material vegetal e as rotinas laboratoriais nos procedimentos químico analíticos. Sci Agrar 8:1–17
R Core Team (2019) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. Available in: https://www.R.project. Accessed 20 Jan 2022
Abreu CA, Coscione AR, Pires AM, Paz-Ferreiro J (2012) Phytoremediation of a soil contaminated by heavy metals and boron using castor oil plants and organic matter amendments. J Geochem Explor 123:3–7. https://doi.org/10.1016/j.gexplo.2012.04.013
Barman M, Datta SP, Rattan RK, Meena MC (2020) Critical limits of deficiency of nickel in intensively cultivated alluvial soils. J Soil Sci Plant Nutr 20(1):284–292. https://doi.org/10.1007/s42729-019-00141-9
Barrera AAA, Alvarez OC, Rodríguez OAH, Mendoza AB, Melendez LRO, Caballero MCS, Barrios DLO (2022) Do foliar applications of nickel increase urease activity and nutrient levels in pecan leaflets? Plant, Soil Environ 68:129–136. https://doi.org/10.17221/487/2021-PSE
Dixon NE, Gazzola C, Blakeley RL, Zerner B (1975) Jack bean urease (EC 3.5.1.5) a metalloenzyme: simple biological role for nickel. J Am Chem Soc 97(14):4131–4133
Davis MA, Pritchard SG, Boyd RS, Prior SA (2001) Developmental and induced responses of nickel-based and organic defences of the nickel-hyperaccumulating shrub, Psychotria douarrei. New Phytol 150:49–58. https://doi.org/10.1046/j.1469-8137.2001.00067.x
Fabiano CC, Tezotto T, Favarin JL, Polacco JC, Mazzafera P (2015) Essentiality of nickel in plants: a role in plant stresses. Front Plant Sci 6:754. https://doi.org/10.3389/fpls.2015.00754
Fernandes ALT, Calzavara SA, Paseto LA, Silva FH (2011) Avaliação de diferentes doses de níquel (Ni) no desenvolvimento vegetativo e produtivo do cafeeiro irrigado cultivado no Noroeste de Minas Gerais. Trabalho de Conclusão de Curso (Graduação em Agronomia) - Faculdades Associadas de Uberaba
Lopes JF, Coelho FC, Rangel OJP, Rabello WS, de Gravina G, Vieira HD (2014) Adubação foliar com níquel e molibdênio no feijoeiro comum cv. Ouro Vermelho. Rev Ceres 61(2):234–240
Verkleij JAC, Parest JE (1989) Cadmium tolerance and co-tolerance in Silene vulgaris (Moench.) Garcke [= S. cucubalus (L.) wilb.]. New Phytol 111:637–645. https://doi.org/10.1111/j.1469-8137.1989.tb02358.x
Arduini I, Godbold DL, Onnis A (1996) Cadmium and copper uptake and distribution in Mediterranean tree seedlings. Physiol Plant 97:111–117. https://doi.org/10.1111/j.1399-3054.1996.tb00486.x
Yang X, Baligar VC, Martens DC, Clark RB (1996) Plant tolerance to nickel toxicity: 2- nickel effects on influx and transport of mineral nutrients in four plant species. J Plant Nutr 19(2):265–279. https://doi.org/10.1080/01904169609365121
Brooks RR (1980) Accumulation of nickel by terrestrial plants. In: NRIAGU, J. O. Nickel in the environment. New York, 407–430
Welch RM (1981) The biological significance of nickel. J Plant Nutr 3:345–356
Barbosa JZ, Zambon LM, Motta ACV, Wendling I (2015) Composition, hot-water solubility of elements and nutritional value of fruits and leaves of yerba-mate. Ciência e Agrotecnologia 39(6):593–603. https://doi.org/10.1590/S1413-70542015000600006
Zhang J, Yang R, Li YC, Peng Y, Wen X, Ni X (2020) Distribution, accumulation, and potential risks of heavy metals in soil and tea leaves from geologically different plantations. Ecotoxicol Environ Safety 195:110475. https://doi.org/10.1016/j.ecoenv.2020.110475
Li W, Cheng H, Mu Y, Xu A, Ma B, Wang F, Xu P (2021) Occurrence, accumulation, and risk assessment of trace metals in tea (Camellia sinensis): a national reconnaissance. Sci Total Environ 792:148354. https://doi.org/10.1016/j.scitotenv.2021.148354
Wen B, Li L, Duan Y, Zhang Y, Shen J, Xia M, Zhu X (2018) Zn, Ni, Mn, Cr, Pb and Cu in soil-tea ecosystem: the concentrations, spatial relationship and potential control. Chemosphere 204:92–100
Kozhevnikova AD, Seregin IV, Bystrova EI, Belyaeva AI, Kataeva MN, Ivanov VB (2009) The effects of lead, nickel, and strontium nitrates on cell division and elongation in maize roots. Russ J Plant Physiol 56(2):242–250. https://doi.org/10.1134/S1021443709020137
Sadeghipour O (2021) Chitosan application improves nickel toxicity tolerance in soybean. J Soil Sci Plant Nutr 21(3):2096–2104. https://doi.org/10.1007/s42729-021-00505-0
Reeves RD, Baker AJM, Becquer T, Echevarria G, Miranda ZJG (2007) The flora and biogeochemistry of the ultramafic soils of Goiás state, Brazil. Plant And Soil 293(1–2):107–119. https://doi.org/10.1007/s11104-007-9192-x
Ma G, Zhang J, Zhang L, Huang C, Chen L, Wang G, Lu C (2019) Elements characterization of Chinese tea with different fermentation degrees and its use for geographical origins by liner discriminant analysis. J Food Compos Anal 82:103246. https://doi.org/10.1016/j.jfca.2019.103246
Zhong WS, Ren T, Zhao LJ (2016) Determination of Pb (lead), Cd (cadmium), Cr (chromium), Cu (copper), and Ni (nickel) in Chinese tea with high-resolution continuum source graphite furnace atomic absorption spectrometry. J Food Drug Anal 24(1):46–55. https://doi.org/10.1016/j.jfda.2015.04.010
Marcos A, Fisher A, Rea G, Hill Steve J (1998) Preliminary study using trace element concentrations and a chemometrics approach to determine the geographical origin of tea. J Anal At Spectrom 13:521–525. https://doi.org/10.1039/A708658J
Nookabkaew S, Rangkadilok N, Satayavivad J (2006) Determination of trace elements in herbal tea products and their infusions consumed in Thailand. J Agric Food Chem 54:6939–6944. https://doi.org/10.1021/jf060571w
Brzezicha-Cirocka J, Grembecka M, Szefer P (2016) Monitoring of essential and heavy metals in green tea from different geographical origins. Environ Monit Assess 188(3):183. https://doi.org/10.1007/s10661-016-5157-y
Szymczycha-Madeja A, Welna M, Zyrnicki W (2013) Multi-element analysis, bioavailability and fractionation of herbal tea products. J Braz Chem Soc 24(5):777–787. https://doi.org/10.5935/0103-5053.20130102
Uren NC (1992) Forms, reaction and availability of nickel n soils. Adv Agron 48:141–203
Palacios G, Gómez I, Carbonell-Barrachina A, Navarro PJ, Mataix J (1998) Effect of nickel concentration on tomato plant nutrition and dry matter yield. J Plant Nutr 21(10):2179–2191. https://doi.org/10.1080/01904169809365553
Roveda LF, Cuquel FL, Motta ACV, de Melo VF (2014) Composto orgânico com altos teores de níquel e sua biodisponibilidade no sistema solo planta. Rev Bras Eng Agríc Ambient 18(8):819–825. https://doi.org/10.1590/1807-1929/agriambi.v18n08p819-825
da Silva RF, Weirich SW, da Ros CO, Scheid DL, Grolli AL, Viel P (2015) Acumulação e translocação de zinco em mudas de espécies do gênero Eucalyptus e Corymbia. Rev Bras Eng Agríc Ambient 19(11):1114–1120. https://doi.org/10.1590/1807-1929/agriambi.v19n11p1114-1120
Tito GA, Chaves LHG, de Vasconcelos ACF (2016) Acúmulo e translocação de cobre e zinco em plantas de Crambe abyssinica. Rev Verde Agroecologia e Desenvolvimento Sustentável 11(4):12–16. https://doi.org/10.18378/rvads.v11i4.4539
Althaus D, Gianello C, Tedesco MJ, Silva KJD, Bissani CA, Felisberto R (2018) Natural fertility and metals contents in soils of Rio Grande do Sul (Brazil). Rev Bras Ciênc Solo 42:1–15. https://doi.org/10.1590/18069657rbcs20160418
Reissmann CB, Radomski MI, de Quadros RMB (1999) Chemical composition of Ilex paraguariensis St. Hil. under different management conditions in seven localities of Paraná State. Braz Arch Biol Technol 42(2):187–194. https://doi.org/10.1590/S1516-89131999000200009
Giulian R, dos Santos CEI (2009) Shubeita S de M, Silva LM da, Yoneama ML, Dias JF (2009) The study of the influence of industrial processing on the elemental composition of mate tea leaves (Ilex paraguariensis) using the PIXE technique. Food Sci Technol 42(1):74–80. https://doi.org/10.1016/j.lwt.2008.05.007
Funding
This study was funded by the National Council for Scientific and Technological Development (CNPq) (Grant Numbers 306908/2016–6 and 311304/2019–2) and to the Coordination for the Improvement of Higher Education Personnel (CAPES) for scholarship financial support.
Author information
Authors and Affiliations
Contributions
Nayara Caroline Majewski Ulbrich: writing—original draft, conceptualization, methodology, investigation, software, formal analysis. Antônio Carlos Vargas Motta: writing—review & editing, conceptualization, methodology, investigation. Ederlan Magri: writing—review & editing, conceptualization, investigation, software, formal analysis. Stephen A. Prior: writing—review & editing, investigation. Carla Gomes de Albuquerque: review & editing, methodology, investigation. Fabiana Gavelaki: review & editing, methodology, investigation. Julierme Zimmer Barbosa: review & editing, investigation. Ivar Wendling: review & editing, investigation. Giovana Poggere: review & editing, conceptualization, methodology, investigation.
Corresponding author
Ethics declarations
Ethics Approval
Not applicable.
Consent to Participate
Not applicable.
Consent for Publication
Not applicable.
Competing Interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Ulbrich, N.C.M., Motta, A.C.V., Magri, E. et al. Accumulation Capacity of Nickel and Zinc in Yerba Mate Cultivated in Soils with Contrasting Parent Materials. Biol Trace Elem Res 201, 5468–5480 (2023). https://doi.org/10.1007/s12011-023-03593-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12011-023-03593-4