Abstract
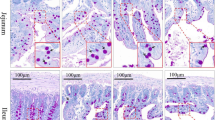
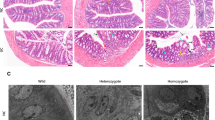
To investigate fluoride (F)-induced intestine barrier damage and the role of estrogen deficiency in this progress, a rat model of estrogen deficiency was established through bilateral surgical removal of ovaries. The F exposure model was then continued by adding sodium fluoride (0, 25, 50, and 100 mg/L, calculated on a fluorine ion basis) to drinking water for 90 days. Afterward, intestinal mucosal structure, barrier function, and inflammatory cytokines were evaluated. The results showed that excessive F decreased the developmental parameters (crypt depth) of the cecum and rectum and inhibited the proliferation capacity of the intestinal epithelia, which are more obvious in the state of estrogen deficiency. The distribution of goblet cells and glycoproteins in the intestinal mucosa decreased with the increase in F concentration, and estrogen deficiency led to a further decline, especially in the rectum. Using the immunofluorescence method, the study showed that excessive F caused interleukin-17A (IL-17A) significantly decrease in the cecum and increase in the rectum. Meanwhile, F treatment remarkably upregulated the expression of intestinal IL-1β, IL-23, and IL-22, while the level of IL-6 was downregulated. In addition, estrogen deficiency increased IL-1β, IL-6, IL-23, and IL-22, but decreased IL-17A expression in the cecum and rectum. Collectively, F exposure damaged intestinal morphological structure, inhibited epithelial cell proliferation and mucus barrier function, and resulted in the disturbance of T helper (Th) 17 cell-related cytokines expression. Estrogen deficiency may further aggravate F-induced damage to the cecum and rectum.







Similar content being viewed by others
Data Availability
All data generated or analyzed during this study are included in this published article.
Code Availability
Not applicable.
References
Peterson LW, Artis D (2014) Intestinal epithelial cells: regulators of barrier function and immune homeostasis. Nat Rev Immunol 14:141–153. https://doi.org/10.1038/nri3608
Chelakkot C, Ghim J, Ryu SH (2018) Mechanisms regulating intestinal barrier integrity and its pathological implications. Exp Mol Med 50:1–9. https://doi.org/10.1038/s12276-018-0126-x
Okumura R, Takeda K (2017) Roles of intestinal epithelial cells in the maintenance of gut homeostasis. Exp Mol Med 49:e338. https://doi.org/10.1038/emm.2017.20
Catalioto R-M, A. Maggi C, Giuliani S, (2011) Intestinal epithelial barrier dysfunction in disease and possible therapeutical interventions. Curr Med Chem 18:398–426. https://doi.org/10.2174/092986711794839179
Guan Q (2019) A comprehensive review and update on the pathogenesis of inflammatory bowel disease. J Immunol Res 2019:1–16. https://doi.org/10.1155/2019/7247238
Mowat AM, Agace WW (2014) Regional specialization within the intestinal immune system. Nat Rev Immunol 14:667–685. https://doi.org/10.1038/nri3738
Maloy KJ, Powrie F (2011) Intestinal homeostasis and its breakdown in inflammatory bowel disease. Nature 474:298–306. https://doi.org/10.1038/nature10208
Goodman WA, Bedoyan SM, Havran HL et al (2020) Impaired estrogen signaling underlies regulatory T cell loss-of-function in the chronically inflamed intestine. Proc Natl Acad Sci 117:17166–17176. https://doi.org/10.1073/pnas.2002266117
Rizzetto L, Fava F, Tuohy KM, Selmi C (2018) Connecting the immune system, systemic chronic inflammation and the gut microbiome: the role of sex. J Autoimmun 92:12–34. https://doi.org/10.1016/j.jaut.2018.05.008
Klein SL, Flanagan KL (2016) Sex differences in immune responses. Nat Rev Immunol 16:626–638. https://doi.org/10.1038/nri.2016.90
Fish EN (2008) The X-files in immunity: sex-based differences predispose immune responses. Nat Rev Immunol 8:737–744. https://doi.org/10.1038/nri2394
Kane SV, Reddy D (2008) Hormonal replacement therapy after menopause is protective of disease activity in women with inflammatory bowel disease. Am J Gastroenterol 103:1193–1196. https://doi.org/10.1111/j.1572-0241.2007.01700.x
Gameiro CM, Romão F, Castelo-Branco C (2010) Menopause and aging: changes in the immune system–a review. Maturitas 67:316–320. https://doi.org/10.1016/j.maturitas.2010.08.003
Collins FL, Rios-Arce ND, Atkinson S et al (2017) Temporal and regional intestinal changes in permeability, tight junction, and cytokine gene expression following ovariectomy-induced estrogen deficiency. Physiol Rep 5:e13263. https://doi.org/10.14814/phy2.13263
Wei W, Pang S, Sun D (2019) The pathogenesis of endemic fluorosis: research progress in the last 5 years. J Cell Mol Med 23:2333–2342. https://doi.org/10.1111/jcmm.14185
Mohammadi AA, Yousefi M, Yaseri M et al (2017) Skeletal fluorosis in relation to drinking water in rural areas of West Azerbaijan. Iran Sci Rep 7:17300. https://doi.org/10.1038/s41598-017-17328-8
Mumtaz N, Pandey G, Labhasetwar PK (2015) Global fluoride occurrence, available technologies for fluoride removal, and electrolytic defluoridation: a review. Crit Rev Env Sci Tec 45(21):2357–2389. https://doi.org/10.1080/10643389.2015.1046768
Johnston NR, Strobel SA (2020) Principles of fluoride toxicity and the cellular response: a review. Arch Toxicol 94:1051–1069. https://doi.org/10.1007/s00204-020-02687-5
Zuo H, Chen L, Kong M et al (2018) Toxic effects of fluoride on organisms. Life Sci 198:18–24. https://doi.org/10.1016/j.lfs.2018.02.001
Dionizio A, Uyghurturk DA, Melo CGS et al (2021) Intestinal changes associated with fluoride exposure in rats: integrative morphological, proteomic and microbiome analyses. Chemosphere 273:129607. https://doi.org/10.1016/j.chemosphere.2021.129607
Zhu SQ, Liu J, Han B et al (1987) (2022) Fluoride exposure cause colon microbiota dysbiosis by destroyed microenvironment and disturbed antimicrobial peptides expression in colon. Environ Pollut Barking Essex 292:118381. https://doi.org/10.1016/j.envpol.2021.118381
Liu S, Zhao J, Tian WS et al (2022) Estrogen deficiency aggravates fluorine ion-induced renal fibrosis via the TGF-β1/Smad signaling pathway in rats. Toxicol Lett 362:26–37. https://doi.org/10.1016/j.toxlet.2022.04.005
Yu YM, Zhou BH, Yang YL et al (2022) Estrogen deficiency aggravates fluoride-induced liver damage and lipid metabolism disorder in rats. Biol Trace Elem Res 200:2767–2776. https://doi.org/10.1007/s12011-021-02857-1
Wang HW, Liu J, Zhao WP et al (2019) Effect of fluoride on small intestine morphology and serum cytokine contents in rats. Biol Trace Elem Res 189:511–518. https://doi.org/10.1007/s12011-018-1503-y
Ando Y, Yang G-X, Tsuda M et al (2012) The immunobiology of colitis and cholangitis in interleukin-23p19 and interleukin-17A deleted dominant negative form of transforming growth factor beta receptor type II mice. Hepatol Baltim Md 56:1418–1426. https://doi.org/10.1002/hep.25803
Wang HW, Zhao WP, Liu J et al (2017) Fluoride-induced oxidative stress and apoptosis are involved in the reducing of oocytes development potential in mice. Chemosphere 186:911–918. https://doi.org/10.1016/j.chemosphere.2017.08.068
Zhou BH, Zhao J, Liu J et al (2015) Fluoride-induced oxidative stress is involved in the morphological damage and dysfunction of liver in female mice. Chemosphere 139:504–511. https://doi.org/10.1016/j.chemosphere.2015.08.030
Chauhan SS, Mahmood A, Ojha S (2013) Ethanol and age enhances fluoride toxicity through oxidative stress and mitochondrial dysfunctions in rat intestine. Mol Cell Biochem 384:251–262. https://doi.org/10.1007/s11010-013-1804-6
Job JT, Rajagopal R, Alfarhan A, Narayanankutty A (2022) Borassus flabellifer Linn haustorium methanol extract mitigates fluoride-induced apoptosis by enhancing Nrf2/Haeme oxygenase 1-dependent glutathione metabolism in intestinal epithelial cells. Drug Chem Toxicol 45:2269–2275. https://doi.org/10.1080/01480545.2021.1926476
Zhao W, Wang H, Tan P et al (2018) Fluoride exposure changed the structure and the function of sperm in the testis and epididymis of male rats. Fluoride 51:340–354
Maldonado-Contreras AL, McCormick BA (2011) Intestinal epithelial cells and their role in innate mucosal immunity. Cell Tissue Res 343:5–12. https://doi.org/10.1007/s00441-010-1082-5
Maloy KJ, Kullberg MC (2008) IL-23 and Th17 cytokines in intestinal homeostasis. Mucosal Immunol 1:339–349. https://doi.org/10.1038/mi.2008.28
Wu X, Tian J, Wang S (2018) Insight into non-pathogenic Th17 cells in autoimmune diseases. Front Immunol 9:1112. https://doi.org/10.3389/fimmu.2018.01112
Liang SC, Tan X-Y, Luxenberg DP et al (2006) Interleukin (IL)-22 and IL-17 are coexpressed by Th17 cells and cooperatively enhance expression of antimicrobial peptides. J Exp Med 203:2271–2279. https://doi.org/10.1084/jem.20061308
Slaats J, Ten Oever J, van de Veerdonk FL, Netea MG (2016) IL-1β/IL-6/CRP and IL-18/ferritin: distinct inflammatory programs in infections. PLoS Pathog 12:e1005973. https://doi.org/10.1371/journal.ppat.1005973
Patnaude L, Mayo M, Mario R et al (2021) Mechanisms and regulation of IL-22-mediated intestinal epithelial homeostasis and repair. Life Sci 271:119195. https://doi.org/10.1016/j.lfs.2021.119195
Yasuda K, Takeuchi Y, Hirota K (2019) The pathogenicity of Th17 cells in autoimmune diseases. Semin Immunopathol 41:283–297. https://doi.org/10.1007/s00281-019-00733-8
Lee JS, Tato CM, Joyce-Shaikh B et al (2015) Interleukin-23-independent IL-17 production regulates intestinal epithelial permeability. Immunity 43:727–738. https://doi.org/10.1016/j.immuni.2015.09.003
Wang HW, Zhou BH, Niu RY et al (2017) Analysis of the roles of dietary protein and calcium in fluoride-induced changes in T-lymphocyte subsets in rat. Environ Toxicol 32:1587–1595. https://doi.org/10.1002/tox.22386
Keir M, Yi T, Lu T, Ghilardi N (2020) The role of IL-22 in intestinal health and disease. J Exp Med 217:e20192195. https://doi.org/10.1084/jem.20192195
Biswas SK (2016) Does the interdependence between oxidative stress and inflammation explain the antioxidant paradox? Oxid Med Cell Longev 2016:5698931. https://doi.org/10.1155/2016/5698931
Tian T, Wang Z, Zhang J (2017) Pathomechanisms of oxidative stress in inflammatory bowel disease and potential antioxidant therapies. Oxid Med Cell Longev 2017:4535194. https://doi.org/10.1155/2017/4535194
Diebel ME, Diebel LN, Manke CW, Liberati DM (2015) Estrogen modulates intestinal mucus physiochemical properties and protects against oxidant injury. J Trauma Acute Care Surg 78:94–99. https://doi.org/10.1097/TA.0000000000000499
Díaz A, López-Grueso R, Gambini J et al (2019) Sex differences in age-associated type 2 diabetes in rats-role of estrogens and oxidative stress. Oxid Med Cell Longev 2019:6734836. https://doi.org/10.1155/2019/6734836
Borrás C, Sastre J, García-Sala D et al (2003) Mitochondria from females exhibit higher antioxidant gene expression and lower oxidative damage than males. Free Radic Biol Med 34:546–552. https://doi.org/10.1016/s0891-5849(02)01356-4
López-Grueso R, Gambini J, Abdelaziz KM et al (2014) Early, but not late onset estrogen replacement therapy prevents oxidative stress and metabolic alterations caused by ovariectomy. Antioxid Redox Signal 20:236–246. https://doi.org/10.1089/ars.2012.5112
Yu M, Pal S, Paterson CW et al (2021) Ovariectomy induces bone loss via microbial-dependent trafficking of intestinal TNF+ T cells and Th17 cells. J Clin Invest 131:143137. https://doi.org/10.1172/JCI143137
Lorenzo J (2021) From the gut to bone: connecting the gut microbiota with Th17 T lymphocytes and postmenopausal osteoporosis. J Clin Invest 131:146619. https://doi.org/10.1172/JCI146619
Funding
This work was supported by the National Natural Science Foundation of China (grant no.·32102740) and the Natural Science Foundation of Henan, China (Grant no. 202300410120).
Author information
Authors and Affiliations
Contributions
Xiao-ying Gao: formal analysis, writing the original draft, and preparation of the data presentation. Ye Jin: visualization, validation, and data curation. Jing Zhao: funding acquisition and validation. Yu-ling Zhang: validation and project administration. Hong-wei Wang: ideas, investigation, and critical review. Bian-hua Zhou: funding acquisition and validation, conceptualization, methodology, review and editing, and provision of resources.
Corresponding author
Ethics declarations
Ethics Approval
The experimental design was approved by the Institutional Animal Experiment Committee of Henan University of Science and Technology, China.
Consent to Participate
Written informed consent for publication was obtained from all participants.
Consent for Publication
Written informed consent for publication was obtained from all participants.
Competing Interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Gao, Xy., Jin, Y., Zhao, J. et al. Th17-Related Cytokines Involved in Fluoride-Induced Cecal and Rectal Barrier Damage of Ovariectomized Rats. Biol Trace Elem Res 201, 4497–4507 (2023). https://doi.org/10.1007/s12011-022-03519-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12011-022-03519-6