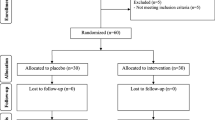
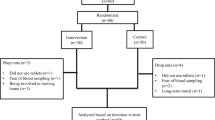
Abstract
Magnesium and melatonin are known to exert multiple beneficial effects including anti-inflammatory and antioxidant actions. This study was designed to determine the effects of magnesium and/or melatonin supplementation on metabolic profiles in women with polycystic ovary syndrome (PCOS). This randomized double-blind, placebo-controlled trial was conducted among 84 subjects with PCOS aged 18–40 years old. Patients were randomly assigned based on the random block procedure to take magnesium, melatonin, magnesium plus melatonin, or placebo for 8 weeks. Fasting blood samples were taken at baseline and after the intervention to quantify related variables. After the 8-week intervention, an insignificant marginal difference was seen in waist circumference (WC) between groups (P = 0.085). Magnesium-melatonin co-supplementation resulted in more reductions in hirsutism compared with other groups (P < 0.001). Serum levels of tumor necrosis factor-α (TNF-α) declined significantly in the melatonin and co-supplementation groups compared to baseline (P < 0.05). Also, magnesium plus melatonin was associated with a more increase in total antioxidant capacity (TAC) levels, as compared to the other treatment groups (P = 0.001). Overall, we found a favorable effect of co-supplementation of magnesium and melatonin for 8 weeks in women with PCOS on hirsutism, serum TNF-α, and TAC levels. Furthermore, melatonin independently contributed to decreased serum values of TNF-α.
Clinical trial registration number http://www.irct.ir: IRCT20191130045556N1, January 2020

Similar content being viewed by others
Availability of Data and Material
The data gathered and analyzed during the current study are available from the corresponding author on reasonable request.
Code Availability
Not applicable.
References
Glueck C, Papanna R, Wang P, Goldenberg N, Sieve-Smith L (2003) Incidence and treatment of metabolic syndrome in newly referred women with confirmed polycystic ovarian syndrome. Metabolism 52:908–915. https://doi.org/10.1016/s0026-0495(03)00104-5
Tarlatzis B, Fauser BC, Legro R et al (2008) Consensus on infertility treatment related to polycystic ovary syndrome. Hum Reprod 23:462–477. https://doi.org/10.1093/humrep/dem426
Group REASPCW (Revised 2003) consensus on diagnostic criteria and long-term health risks related to polycystic ovary syndrome (PCOS). Hum Reprod 19:41–47
Artimani T, Karimi J, Mehdizadeh M et al (2018) Evaluation of pro-oxidant-antioxidant balance (PAB) and its association with inflammatory cytokines in polycystic ovary syndrome (PCOS). Gynecol Endocrinol 34:148–152. https://doi.org/10.1080/09513590.2017.1371691
Shorakae S, Teede H, de Courten B, Lambert G, Boyle J, Moran LJ (2015) The emerging role of chronic low-grade inflammation in the pathophysiology of polycystic ovary syndrome. Semin Reprod Med 33:257–269. https://doi.org/10.1055/s-0035-1556568
Repaci A, Gambineri A, Pasquali R (2011) The role of low-grade inflammation in the polycystic ovary syndrome. Mol Cell Endocrinol 335:30–41. https://doi.org/10.1016/j.mce.2010.08.002
Sathyapalan T, Atkin SL (2010, 2010) Mediators of inflammation in polycystic ovary syndrome in relation to adiposity. Mediat Inflamm. https://doi.org/10.1155/2010/758656
Murri M, Luque-Ramirez M, Insenser MA, Ojeda-Ojeda M, Escobar-Morreale HF (2013) Circulating markers of oxidative stress and polycystic ovary syndrome (PCOS): a systematic review and meta-analysis. Hum Reprod Update 19:268–288. https://doi.org/10.1093/humupd/dms059
Chakraborty P, Ghosh S, Goswami S, Kabir SN, Chakravarty B, Jana K (2013) Altered trace mineral milieu might play an aetiological role in the pathogenesis of polycystic ovary syndrome. Biol Trace Elem Res 152:9–15. https://doi.org/10.1007/s12011-012-9592-5
Zhang D, Luo W-Y, Liao H, Wang C-F, Sun Y (2008) The effects of oxidative stress to PCOS. Sichuan Da Xue Xue Bao Yi Xue Ban 39:421–423
Cagnacci A, Volpe A (2002) A role for melatonin in PCOS? Fertil Steril 77:1089. https://doi.org/10.1016/s0015-0282(02)03070-4
Cassone VM, Natesan AK (1997) Time and time again: the phylogeny of melatonin as a transducer of biological time. J Biol Rhythm 12:489–497. https://doi.org/10.1177/074873049701200602
Reiter RJ, Tan D-X, Fuentes-Broto L (2010) Melatonin: a multitasking molecule. Prog Brain Res 181:127–151. https://doi.org/10.1016/s0079-6123(08)81008-4
Tan DX, Manchester LC, Sainz RM et al (2005) Interactions between melatonin and nicotinamide nucleotide: NADH preservation in cells and in cell-free systems by melatonin. J Pineal Res 39:185–194. https://doi.org/10.1111/j.1600-079x.2005.00234.x
Reiter RJ, Tan DX, Maldonado MD (2005) Melatonin as an antioxidant: physiology versus pharmacology. J Pineal Res 39:215–216. https://doi.org/10.1111/j.1600-079x.2005.00261.x
Tanavde V, Maitra A (2011) In vitro modulation of steroidogenesis and gene expression by melatonin: a study with porcine antral follicles. Endocr Res 29:399–410. https://doi.org/10.1081/erc-120026946
Galano A, Tan DX, Reiter RJ (2011) Melatonin as a natural ally against oxidative stress: a physicochemical examination. J Pineal Res 51:1–16. https://doi.org/10.1111/j.1600-079x.2011.00916.x
Tamura H, Nakamura Y, Korkmaz A et al (2009) Melatonin and the ovary: physiological and pathophysiological implications. Fertil Steril 92:328–343. https://doi.org/10.1016/j.fertnstert.2008.05.016
Pacchiarotti A, Carlomagno G, Antonini G, Pacchiarotti A (2016) Effect of myo-inositol and melatonin versus myo-inositol, in a randomized controlled trial, for improving in vitro fertilization of patients with polycystic ovarian syndrome. Gynecol Endocrinol 32:69–73. https://doi.org/10.3109/09513590.2015.1101444
Jamilian M, Foroozanfard F, Mirhosseini N et al (2019) Effects of melatonin supplementation on hormonal, inflammatory, genetic, and oxidative stress parameters in women with polycystic ovary syndrome. Front Endocrinol 10:273. https://doi.org/10.3389/fendo.2019.00273
Glasdam S-M, Glasdam S, Peters GH (2016) The importance of magnesium in the human body: a systematic literature review. Adv Clin Chem 73:169–193. https://doi.org/10.1016/bs.acc.2015.10.002
Rodríguez-Morán M, Mendía LES, Galván GZ, Guerrero-Romero F (2012) The role of magnesium in type 2 diabetes: a brief based-clinical review. Magnes Res 24:156–162. https://doi.org/10.1684/mrh.2011.0299
Asemi Z, Esmaillzadeh A (2015) DASH diet, insulin resistance, and serum hs-CRP in polycystic ovary syndrome: a randomized controlled clinical trial. Horm Metab Res 47:232–238. https://doi.org/10.1055/s-0034-1376990
Szczuko M, Skowronek M, Zapalowska-Chwyc M, Starczewski A (2016) Quantitative assessment of nutrition in patients with polycystic ovary syndrome (PCOS). Rocz Panstw Zakl Hig 67:419–426
Hamilton KP, Zelig R, Parker AR, Haggag A (2019) Insulin resistance and serum magnesium concentrations among women with polycystic ovary syndrome. Curr Dev Nutr 3:nzz108. https://doi.org/10.1093/cdn/nzz108
Ebrahimi FA, Foroozanfard F, Aghadavod E, Bahmani F, Asemi Z (2018) The effects of magnesium and zinc co-supplementation on biomarkers of inflammation and oxidative stress, and gene expression related to inflammation in polycystic ovary syndrome: a randomized controlled clinical trial. Biol Trace Elem Res 184:300–307. https://doi.org/10.1007/s12011-017-1198-5
Maktabi M, Jamilian M, Asemi Z (2018) Magnesium-zinc-calcium-vitamin D co-supplementation improves hormonal profiles, biomarkers of inflammation and oxidative stress in women with polycystic ovary syndrome: a randomized, double-blind, placebo-controlled trial. Biol Trace Elem Res 182:21–28. https://doi.org/10.1007/s12011-017-1085-0
Moslehi N, Vafa M, Rahimi-Foroushani A, Golestan B (2012) Effects of oral magnesium supplementation on inflammatory markers in middle-aged overweight women. J Res Med Sci 17:607–614
Dou M, Ma AG, Wang QZ et al (2009) Supplementation with magnesium and vitamin E were more effective than magnesium alone to decrease plasma lipids and blood viscosity in diabetic rats. Nutr Res 29:519–524. https://doi.org/10.1016/j.nutres.2009.07.001
Chang W, Ma A, Wang Q, Mao R, Li C (2014) Effects of vitamin E and magnesium on glucolipid metabolism in obese rats. Wei Sheng Yan Jiu 43:713–718
Neter J, Wasserman W, Kutner MH (1989) Applied linear regression models.
Shabani A, Foroozanfard F, Kavossian E et al (2019) Effects of melatonin administration on mental health parameters, metabolic and genetic profiles in women with polycystic ovary syndrome: a randomized, double-blind, placebo-controlled trial. J Affect Disord 250:51–56. https://doi.org/10.1016/j.jad.2019.02.066
Committee IR (2005) Guidelines for data processing and analysis of the International Physical Activity Questionnaire (IPAQ)-short and long forms. http://www.ipaq.ki.se/scoring.pdf. Accessed 1 Dec 2019
Hatch R, Rosenfield RL, Kim MH, Tredway D (1981) Hirsutism: implications, etiology, and management. Am J Obstet Gynecol 140:815–830. https://doi.org/10.1016/0002-9378(81)90746-8
Janero DR (1990) Malondialdehyde and thiobarbituric acid-reactivity as diagnostic indices of lipid peroxidation and peroxidative tissue injury. Free Radic Biol Med 9:515–540. https://doi.org/10.1016/0891-5849(90)90131-2
Miller NJ, Rice-Evans C, Davies MJ, Gopinathan V, Milner A (1993) A novel method for measuring antioxidant capacity and its application to monitoring the antioxidant status in premature neonates. Clin Sci 84:407–412. https://doi.org/10.1042/cs0840407
Duleba AJ, Dokras A (2012) Is PCOS an inflammatory process? Fertil Steril 97:7–12. https://doi.org/10.1016/j.fertnstert.2011.11.023
Hyderali BN, Mala K (2015) Oxidative stress and cardiovascular complications in polycystic ovarian syndrome. Eur J Obstet Gynecol Reprod Biol 191:15–22. https://doi.org/10.1016/j.ejogrb.2015.05.005
González F (2012) Inflammation in polycystic ovary syndrome: underpinning of insulin resistance and ovarian dysfunction. Steroids 77:300–305. https://doi.org/10.1016/j.steroids.2011.12.003
Simental-Mendia LE, Sahebkar A, Rodriguez-Moran M, Zambrano-Galvan G, Guerrero-Romero F (2017) Effect of magnesium supplementation on plasma C-reactive protein concentrations: a systematic review and meta-analysis of randomized controlled trials. Curr Pharm Des 23:4678–4686. https://doi.org/10.2174/1381612823666170525153605
Elin RJ (2010) Assessment of magnesium status for diagnosis and therapy. Magnes Res 23:194–198. https://doi.org/10.1684/mrh.2010.0213
Alfrey AC, Miller NL (1973) Bone magnesium pools in uremia. J Clin Invest 52:3019–3027. https://doi.org/10.1172/jci107500
Shokrpour M, Asemi Z (2019) The effects of magnesium and vitamin E co-supplementation on hormonal status and biomarkers of inflammation and oxidative stress in women with polycystic ovary syndrome. Biol Trace Elem Res 191:54–60. https://doi.org/10.1007/s12011-018-1602-9
Barbagallo M, Dominguez LJ, Galioto A et al (2003) Role of magnesium in insulin action, diabetes and cardio-metabolic syndrome X. Mol Asp Med 24:39–52. https://doi.org/10.1016/s0098-2997(02)00090-0
Liu Y, Guo Y, Wang Z (2007) Effect of magnesium on reactive oxygen species production in the thigh muscles of broiler chickens. Br Poult Sci 48:84–89. https://doi.org/10.1080/00071660601148187
Boujelben M, Ghorbel F, Vincent C et al (2009) Lipid peroxidation and HSP72/73 expression in rat following cadmium chloride administration: interactions of magnesium supplementation. Exp Toxicol Pathol 57:437–443. https://doi.org/10.1016/j.etp.2006.02.012
Tamura H, Takasaki A, Taketani T et al (2012) The role of melatonin as an antioxidant in the follicle. J. Ovarian Res 5:1–9. https://doi.org/10.1186/1757-2215-5-5
Jain P, Jain M, Haldar C, Singh TB, Jain S (2013) Melatonin and its correlation with testosterone in polycystic ovarian syndrome. J Hum Reprod Sci 6:253. https://doi.org/10.4103/0974-1208.126295
Mojaverrostami S, Asghari N, Khamisabadi M, Khoei HH (2019) The role of melatonin in polycystic ovary syndrome: a review. Int J Reprod Biomed 17:865. https://doi.org/10.18502/ijrm.v17i12.5789
Dokras A, Sarwer DB, Allison KC et al (2016) Weight loss and lowering androgens predict improvements in health-related quality of life in women with PCOS. J Clin Endocrinol Metab 101:2966–2974. https://doi.org/10.1210/jc.2016-1896
Lin YW, Lee LM, Lee WJ et al (2016) Melatonin inhibits MMP-9 transactivation and renal cell carcinoma metastasis by suppressing Akt-MAPK s pathway and NF-κB DNA-binding activity. J Pineal Res 60:277–290. https://doi.org/10.1111/jpi.12308
Mohan N, Sadeghi K, Reiter RJ, Meltz ML (1995) The neurohormone melatonin inhibits cytokine, mitogen and ionizing radiation induced NF-kappa B. Int J Biochem Mol Biol 37:1063–1070
Akbari M, Ostadmohammadi V, Tabrizi R et al (2018) The effects of melatonin supplementation on inflammatory markers among patients with metabolic syndrome or related disorders: a systematic review and meta-analysis of randomized controlled trials. Inflammopharmacology 26:899–907. https://doi.org/10.1007/s10787-018-0508-7
Pakravan H, Ahmadian M, Fani A, Aghaee D, Brumanad S, Pakzad B (2017) The effects of melatonin in patients with nonalcoholic fatty liver disease: a randomized controlled trial. Adv Biomed Res 6:40
Karamitri A, Jockers R (2019) Melatonin in type 2 diabetes mellitus and obesity. Nat Rev Endocrinol 15:105–125. https://doi.org/10.4103/2277-9175.204593
Acknowledgements
The authors wish to thank Mr. Meisam Barati, Mr. Seyed Javad Mousavi and Mrs. Marziyeh Keshizadeh for their assistance and the staff of the Diagnostic Laboratory Department at 29 Bahman Hospital (Tabriz, Iran) for their technical help. We also thank all of the study participants.
Funding
The present study was funded by the vice chancellor for research of Ahvaz Jundishapur University of Medical Science (Grant No.: NRC-9815) and Tabriz University of Medical science (Grant No.: 64999).
Author information
Authors and Affiliations
Contributions
MK, MA, and RM were involved in the conception and design of the study. LH, RN, and HBR contributed in methodology. MAJ and RM performed the data analysis and interpretation. RM participated in the conduction of the study and manuscript drafting. MK supervised the development of the work. Mk and MA conducted the critical review. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics Approval
The ethical committee of Ahvaz Jundishapur University of Medical Sciences confirmed the study and the approval of the Ethics Committee was obtained (IR.AJUMS.REC.1398.637).
Consent to Participate
Signed informed consent was obtained from all participants.
Consent to Publish
Not applicable.
Conflict of Interest
The authors declare no conflict of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Mousavi, R., Alizadeh, M., Asghari Jafarabadi, M. et al. Effects of Melatonin and/or Magnesium Supplementation on Biomarkers of Inflammation and Oxidative Stress in Women with Polycystic Ovary Syndrome: a Randomized, Double-Blind, Placebo-Controlled Trial. Biol Trace Elem Res 200, 1010–1019 (2022). https://doi.org/10.1007/s12011-021-02725-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12011-021-02725-y