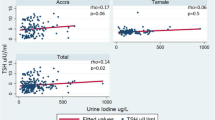
Abstract
Iodine nutrition during pregnancy can affect newborn thyroid-stimulating-hormone concentration (TSH). Associations of newborn TSH with the neurodevelopment and growth of children are inconsistent. The aim of the study was to systematically review the literature on the associations between newborn TSH and childhood neurodevelopment and growth. Databases including PubMed, Scopus, CINAHL, Embase, PsycINFO, WHO, and Iodine Global Network were searched for eligible studies. Seventeen studies were included. Neurodevelopment was assessed using different tools in children aged 1–12 years of age. The associations between newborn TSH and cognitive development were negative in studies from iodine deficient populations, while a null association was found in studies from iodine sufficient populations. A null association between TSH and psychomotor development was observed regardless of iodine status of the study populations. There was no evidence of an association between newborn TSH and child anthropometry, but evidence of negative association was found between newborn TSH and birthweight. Although the associations between newborn TSH and neurodevelopment may differ based on the iodine status of populations, most of the included studies did not adjust for the key confounders and had a small sample size. Quality data-linkage studies that utilize newborn TSH data from newborn screening with adequate adjustment for potential confounders are warranted to better understand the relationship between newborn TSH and neurodevelopment and growth in children. CRD42020152878





Similar content being viewed by others
Data Availability
Not applicable as this is a review paper.
Code Availability
Not applicable.
References
Rousset B, Dupuy C, Miot F, Dumont J (2000) Chapter 2 thyroid hormone synthesis and secretion. In: Feingold KR, Anawalt B, Boyce A et al. (eds) Endotext. South Dartmouth (MA),
Chiamolera MI, Wondisford FE (2009) Minireview: thyrotropin-releasing hormone and the thyroid hormone feedback mechanism. Endocrinology 150(3):1091–1096. https://doi.org/10.1210/en.2008-1795
Nazeri P, Mirmiran P, Kabir A, Azizi F (2016) Neonatal thyrotropin concentration and iodine nutrition status of mothers: a systematic review and meta-analysis. Am J Clin Nutr 104(6):1628–1638. https://doi.org/10.3945/ajcn.116.131953
European Society for Paediatric Endocrinology (1999) Revised guidelines for neonatal screening programmes for primary congenital hypothyroidism. Working Group on Neonatal Screening of the European Society for Paediatric Endocrinology. Horm Res 52(1):49–52. https://doi.org/10.1159/000023433
Lain SJ, Bentley JP, Wiley V, Roberts CL, Jack M, Wilcken B, Nassar N (2016) Association between borderline neonatal thyroid-stimulating hormone concentrations and educational and developmental outcomes: a population-based record-linkage study. Lancet Diabetes Endocrinol 4(9):756–765. https://doi.org/10.1016/S2213-8587(16)30122-X
Korzeniewski SJ, Soto-Rivera CL, Fichorova RN, Allred EN, Kuban KC, O'Shea TM, Paneth N, Agus M, Dammann O, Leviton A (2014) Are preterm newborns who have relative hyperthyrotropinemia at increased risk of brain damage? J Pediatr Endocrinol Metab 27(11-12):1077–1088. https://doi.org/10.1515/jpem-2014-0059
Cuestas E, Gaido MI, Capra RH (2015) Transient neonatal hyperthyrotropinemia is a risk factor for developing persistent hyperthyrotropinemia in childhood with repercussion on developmental status. Eur J Endocrinol 172(4):483–490. https://doi.org/10.1530/EJE-13-0907
Delange F (1999) Neonatal thyroid screening as a monitoring tool for the control of iodine deficiency. Acta Paediatr Suppl 88(432):21–24
Bougma K, Aboud FE, Harding KB, Marquis GS (2013) Iodine and mental development of children 5 years old and under: a systematic review and meta-analysis. Nutrients 5(4):1384–1416. https://doi.org/10.3390/nu5041384
Lain S, Trumpff C, Grosse SD, Olivieri A, Van Vliet G (2017) Are lower TSH cutoffs in neonatal screening for congenital hypothyroidism warranted? Eur J Endocrinol 177(5):D1–D12. https://doi.org/10.1530/EJE-17-0107
Sterne JA, Hernan MA, Reeves BC, Savovic J, Berkman ND, Viswanathan M, Henry D, Altman DG, Ansari MT, Boutron I, Carpenter JR, Chan AW, Churchill R, Deeks JJ, Hrobjartsson A, Kirkham J, Juni P, Loke YK, Pigott TD, Ramsay CR, Regidor D, Rothstein HR, Sandhu L, Santaguida PL, Schunemann HJ, Shea B, Shrier I, Tugwell P, Turner L, Valentine JC, Waddington H, Waters E, Wells GA, Whiting PF, Higgins JP (2016) ROBINS-I: a tool for assessing risk of bias in non-randomised studies of interventions. BMJ 355:i4919. https://doi.org/10.1136/bmj.i4919
Association of newborn thyroid stimulating hormone (TSH) concentration with child growth and neurodevelopment: a systematic review and meta-analysis (2020) PROSPERO https://www.crd.york.ac.uk/prospero/display_record.php?ID=CRD42020152878. Accessed 28/11/2020
Gaudino R, Garel C, Czernichow P, Leger J (2005) Proportion of various types of thyroid disorders among newborns with congenital hypothyroidism and normally located gland: a regional cohort study. Clin Endocrinol 62(4):444–448. https://doi.org/10.1111/j.1365-2265.2005.02239.x
Rose SR, Brown RS (2006) Update of newborn screening and therapy for congenital hypothyroidism. Pediatrics 117(6):2290–2303. https://doi.org/10.1542/peds.2006-0915
WHO (2004) Iodine status worldwide : WHO Global Database on Iodine Deficiency, Geneva
De Benoist B, McLean E, Andersson M, Rogers L (2008) Iodine deficiency in 2007: global progress since 2003. Food and Nutrition Bulletin, vol 29, no 3 © 2008, The United Nations University 29 (3)
Iodine Global Network (2017) Global Scorecard of Iodine Nutrition in 2017 in the general population and in pregnant women (PW). Iodine Global Network Zurich, Switzerland
Iodine Global Network (2015) Global Iodine Nutrition Scorecard Iodine Global Network. Zurich, Switzerland
Borenstein M, Higgins JP, Hedges LV, Rothstein HR (2017) Basics of meta-analysis: I(2) is not an absolute measure of heterogeneity. Res Synth Methods 8(1):5–18. https://doi.org/10.1002/jrsm.1230
Holger Schünemann, Jan Brożek, Gordon Guyatt, Oxman A (2013) GRADE Handbook. Handbook for grading the quality of evidence and the strength of recommendations using the GRADE approach. The GRADE Working Group,
Higgins JP, Thompson SG, Deeks JJ, Altman DG (2003) Measuring inconsistency in meta-analyses. BMJ 327(7414):557–560. https://doi.org/10.1136/bmj.327.7414.557
Hedges LV (2016) Distribution theory for Glass's estimator of effect size and related estimators. J Educ Stat 6(2):107–128. https://doi.org/10.3102/10769986006002107
Cumming G (2013) Understanding the new statistics: effect sizes, confidence intervals, and meta-analysis. Routledge,
Altman DG, Bland JM (2011) How to obtain the confidence interval from a P value. BMJ 343:d2090. https://doi.org/10.1136/bmj.d2090
Cohen J (2013) Statistical power analysis for the behavioral sciences. Routledge,
Zhou SJ, Gibson RA, Crowther CA, Baghurst P, Makrides M (2006) Effect of iron supplementation during pregnancy on the intelligence quotient and behavior of children at 4 y of age: long-term follow-up of a randomized controlled trial. Am J Clin Nutr 83(5):1112–1117. https://doi.org/10.1093/ajcn/83.5.1112
Zhou SJ, Condo D, Ryan P, Skeaff SA, Howell S, Anderson PJ, McPhee AJ, Makrides M (2019) Association between maternal iodine intake in pregnancy and childhood neurodevelopment at age 18 months. Am J Epidemiol 188(2):332–338. https://doi.org/10.1093/aje/kwy225
Walter T, De Andraca I, Chadud P, Perales CG (1989) Iron deficiency anemia: adverse effects on infant psychomotor development. Pediatrics 84(1):7–17
Riano Galan I, Sanchez Martinez P, Pilar Mosteiro Diaz M, Rivas Crespo MF (2005) Psycho-intellectual development of 3 year-old children with early gestational iodine deficiency. J Pediatr Endocrinol Metab 18(Suppl 1):1265–1272. https://doi.org/10.1515/jpem.2005.18.s1.1265
Murcia M, Rebagliato M, Iniguez C, Lopez-Espinosa MJ, Estarlich M, Plaza B, Barona-Vilar C, Espada M, Vioque J, Ballester F (2011) Effect of iodine supplementation during pregnancy on infant neurodevelopment at 1 year of age. Am J Epidemiol 173(7):804–812. https://doi.org/10.1093/aje/kwq424
Belcari F, Placidi G, Guzzetta A, Tonacchera M, Ciampi M, Bartoli A, Scaramuzzo RT, Frumento P, Cioni G, Pinchera A, Boldrini A, Ghirri P (2011) Thyroid-stimulating hormone levels in the first days of life and perinatal factors associated with sub-optimal neuromotor outcome in pre-term infants. J Endocrinol Investig 34(10):e308–e313. https://doi.org/10.3275/7795
Costeira MJ, Oliveira P, Santos NC, Ares S, Saenz-Rico B, de Escobar GM, Palha JA (2011) Psychomotor development of children from an iodine-deficient region. J Pediatr 159(3):447–453. https://doi.org/10.1016/j.jpeds.2011.02.034
Trumpff C, De Schepper J, Vanderfaeillie J, Vercruysse N, Van Oyen H, Moreno-Reyes R, Tafforeau J, Vanderpas J, Vandevijvere S (2015) Thyroid-stimulating hormone (TSH) concentration at birth in Belgian neonates and cognitive development at preschool age. Nutrients 7(11):9018–9032. https://doi.org/10.3390/nu7115450
Trumpff C, De Schepper J, Vanderfaeillie J, Vercruysse N, Tafforeau J, Van Oyen H, Vandevijvere S (2016) No association between elevated thyroid-stimulating hormone at birth and parent-reported problem behavior at preschool age. Front Endocrinol (Lausanne) 7:161. https://doi.org/10.3389/fendo.2016.00161
Trumpff C, De Schepper J, Vanderfaeillie J, Vercruysse N, Van Oyen H, Moreno-Reyes R, Tafforeau J, Vandevijvere S (2016) Neonatal thyroid-stimulating hormone concentration and psychomotor development at preschool age. Arch Dis Child 101(12):1100–1106. https://doi.org/10.1136/archdischild-2015-310006
Leviton A, Allred EN, Fichorova RN, Kuban KC, Michael O'Shea T, Dammann O, investigators Es (2016) Systemic inflammation on postnatal days 21 and 28 and indicators of brain dysfunction 2years later among children born before the 28th week of gestation. Early Hum Dev 93:25–32. https://doi.org/10.1016/j.earlhumdev.2015.11.004
Kuban KC, Joseph RM, O'Shea TM, Heeren T, Fichorova RN, Douglass L, Jara H, Frazier JA, Hirtz D, Rollins JV, Paneth N, Extremely low gestational age newborn study I (2017) Circulating inflammatory-associated proteins in the first month of life and cognitive impairment at age 10 years in children born extremely preterm. J Pediatr 180:116–123. https://doi.org/10.1016/j.jpeds.2016.09.054
Calaciura F, Mendorla G, Distefano M, Castorina S, Fazio T, Motta RM, Sava L, Delange F, Vigneri R (1995) Childhood IQ measurements in infants with transient congenital hypothyroidism. Clin Endocrinol 43(4):473–477. https://doi.org/10.1111/j.1365-2265.1995.tb02620.x
Azizi F, Afkhami M, Sarshar A, Nafarabadi M (2001) Effects of transient neonatal hyperthyrotropinemia on intellectual quotient and psychomotor performance. Int J Vitam Nutr Res 71(1):70–73. https://doi.org/10.1024/0300-9831.71.1.70
Choudhury N, Gorman KS (2003) Subclinical prenatal iodine deficiency negatively affects infant development in Northern China. J Nutr 133(10):3162–3165. https://doi.org/10.1093/jn/133.10.3162
Freire C, Ramos R, Amaya E, Fernandez MF, Santiago-Fernandez P, Lopez-Espinosa MJ, Arrebola JP, Olea N (2010) Newborn TSH concentration and its association with cognitive development in healthy boys. Eur J Endocrinol 163(6):901–909. https://doi.org/10.1530/EJE-10-0495
Shields BM, Knight BA, Hill A, Hattersley AT, Vaidya B (2011) Fetal thyroid hormone level at birth is associated with fetal growth. J Clin Endocrinol Metab 96(6):E934–E938. https://doi.org/10.1210/jc.2010-2814
Sareen N, Kapil U, Nambiar V, Pandey RM, Khenduja P (2016) Iodine nutritional status in Uttarakhand State, India. Indian J Endocrinol Metab 20(2):171–176. https://doi.org/10.4103/2230-8210.176363
Kapil U, Pandey RM, Sareen N, Khenduja P, Bhadoria AS (2015) Iodine nutritional status in Himachal Pradesh state, India. Indian J Endocrinol Metab 19(5):602–607. https://doi.org/10.4103/2230-8210.163173
WHO, ICCIDD, UNICEF (1994) Indicators for assessing iodine deficiency disorders and their control through salt iodization. World Health Organization, Geneva
Markou K, Georgopoulos N, Kyriazopoulou V, Vagenakis AG (2001) Iodine-induced hypothyroidism. Thyroid 11(5):501–510. https://doi.org/10.1089/105072501300176462
Chen W, Sang Z, Tan L, Zhang S, Dong F, Chu Z, Wei W, Zhao N, Zhang G, Yao Z, Shen J, Zhang W (2015) Neonatal thyroid function born to mothers living with long-term excessive iodine intake from drinking water. Clin Endocrinol 83(3):399–404. https://doi.org/10.1111/cen.12625
Zhou SJ, Anderson AJ, Gibson RA, Makrides M (2013) Effect of iodine supplementation in pregnancy on child development and other clinical outcomes: a systematic review of randomized controlled trials. Am J Clin Nutr 98(5):1241–1254. https://doi.org/10.3945/ajcn.113.065854
Taylor PN, Okosieme OE, Dayan CM, Lazarus JH (2014) Therapy of endocrine disease: Impact of iodine supplementation in mild-to-moderate iodine deficiency: systematic review and meta-analysis. Eur J Endocrinol 170(1):R1–R15. https://doi.org/10.1530/EJE-13-0651
Levie D, Korevaar TIM, Bath SC, Murcia M, Dineva M, Llop S, Espada M, van Herwaarden AE, de Rijke YB, Ibarluzea JM, Sunyer J, Tiemeier H, Rayman MP, Guxens M, Peeters RP (2019) Association of maternal iodine status with child IQ: a meta-analysis of individual participant data. J Clin Endocrinol Metab 104(12):5957–5967. https://doi.org/10.1210/jc.2018-02559
Ghassabian A, Steenweg-de Graaff J, Peeters RP, Ross HA, Jaddoe VW, Hofman A, Verhulst FC, White T, Tiemeier H (2014) Maternal urinary iodine concentration in pregnancy and children's cognition: results from a population-based birth cohort in an iodine-sufficient area. BMJ Open 4(6):e005520. https://doi.org/10.1136/bmjopen-2014-005520
Velasco I, Carreira M, Santiago P, Muela JA, Garcia-Fuentes E, Sanchez-Munoz B, Garriga MJ, Gonzalez-Fernandez MC, Rodriguez A, Caballero FF, Machado A, Gonzalez-Romero S, Anarte MT, Soriguer F (2009) Effect of iodine prophylaxis during pregnancy on neurocognitive development of children during the first two years of life. J Clin Endocrinol Metab 94(9):3234–3241. https://doi.org/10.1210/jc.2008-2652
WHO, UNICEF, ICCIDD (2007) Assessment of iodine deficiency disorders and monitoring their elimination—a guide for programme managers 3rd edn.,
Acknowledgements
We thank Michael Draper and Angela Mills (Librarians at University of Adelaide) for their assistance in building search strategies of databases.
Funding
MMW was supported by The University of Adelaide, Australian Government Research Training Program.
Author information
Authors and Affiliations
Contributions
MMW and SJZ designed the study. MMW performed literature search, data extraction, analysis, and drafting the manuscript. SJZ oversaw the study conduct and critically reviewed the manuscript. LGS contributed to the design, interpretation of the results, and critically reviewed the manuscript. All authors approved the final manuscript.
Corresponding author
Ethics declarations
Ethics Approval
Not applicable as this is a review paper.
Consent to Participate
Not applicable as this is a review paper.
Consent for Publication
Not applicable as this is a review paper.
Conflict of Interest
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Wassie, M.M., Smithers, L.G. & Zhou, S.J. Association Between Newborn Thyroid-Stimulating-Hormone Concentration and Neurodevelopment and Growth: a Systematic Review. Biol Trace Elem Res 200, 473–487 (2022). https://doi.org/10.1007/s12011-021-02665-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12011-021-02665-7