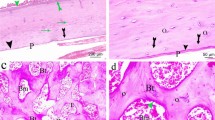
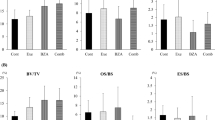
Abstract
Osteoporosis is a growing public health issue for an aging society. Previous studies have found both beneficial and detrimental effects of obesity on bone health. The purpose of this study was to investigate the impact of estrogen deficiency and physical activity on bone and blood concentrations of macrominerals (Ca, P, and Mg) and microminerals (Zn, Se, Cu, and Fe) in a high-fat diet-induced obesity rat model. Forty-eight female Wistar rats were divided into six groups: sham-operated and ovariectomized rats that received a standard diet (SD), high-fat diet (HFD), or HFD accompanied by physical exercise. The effect of ovariectomy on bone minerals varied with diet. Ovariectomy significantly decreased femoral Ca and Mg in sedentary rats receiving a SD; femoral Se, Cu, Zn, and Fe in sedentary rats on HFD; and plasma Fe in both sedentary rats on SD and exercising rats on HFD. The interaction of ovariectomy and diet had the strongest impact on Mg and Se concentrations in femur. In ovariectomized rats, HFD showed to have a protective effect on bone mineralization (femoral Ca and Mg), and a negative one on antioxidant microminerals (femoral Se, Cu, and Zn). Physical activity reduced the decline of Se, Cu, Zn, and Fe in the femur of ovariectomized rats on HFD. In the current state of knowledge, it is difficult to suggest if decreased femoral levels of antioxidant microminerals may contribute to the pathophysiology of osteoporosis in obese individuals or just reflect the mineral status in the body.





Similar content being viewed by others
Data Availability
The datasets generated and analyzed during the current study are available from the corresponding author on reasonable request.
Abbreviations
- BMD:
-
bone mineral density
- HFD:
-
high-fat diet
- Ovx:
-
ovariectomized
- R:
-
running
- S:
-
sedentary
- SD:
-
standard diet
- Sham:
-
sham-operated
References
Sozen T, Ozısık L, Basaran NC (2017) An overview and management of osteoporosis. Eur J Rheumatol 4(1):46–56. https://doi.org/10.5152/eurjrheum.2016.048
Lelovas PP, Xanthos TT, Thoma SE, Lyritis GP, Dontas IA (2008) The laboratory rat as an animal model for osteoporosis research. Comp Med 58(5):424–430
Favus MJ, Bushinsky DA, Lemann J Jr (2006) Regulation of calcium, magnesium, and phosphate metabolism. In: Favus MJ (ed) Primer on the metabolic bone diseases and disorders of mineral metabolism, 6th edn. American Society for Bone and Mineral Research, Washington, DC, pp 76–117
Feng X, McDonald JM (2011) Disorders of bone remodeling. Annu Rev Pathol 6:121–145. https://doi.org/10.1146/annurev-pathol-011110-130203
de Baaij JHF, Joost GJ, Hoenderop JGJ, Bindels RJM (2015) Magnesium in man: implications for health and disease. Physiol Rev 95(1):1–46. https://doi.org/10.1152/physrev.00012.2014
Odabasi E, Turan M, Aydin A, Akay C, Kutlu M (2008) Magnesium, zinc, copper, manganese, and selenium levels in postmenopausal women with osteoporosis. Can magnesium play a key role in osteoporosis? Ann Acad Med Singap 37(7):564–567
Zeng H, Cao JJ, Combs GF Jr (2013) Selenium in bone health: roles in antioxidant protection and cell proliferation. Nutrients 5(1):97–110. https://doi.org/10.3390/nu5010097
Karita K, Yamanouchi Y, Takano T, Oku J, Kisaki T, Yano E (2008) Associations of blood selenium and serum lipid levels in Japanese premenopausal and postmenopausal women. Menopause 15(1):119–124. https://doi.org/10.1097/gme.0b013e31806bf32c
Hoeg A, Gogakos A, Murphy E, Mueller S, Kohrle J, Reid DM, Gluer CC, Felsenberg D, Roux C, Eastell R, Schomburg L, Williams GR (2012) Bone turnover and bone mineral density are independently related to selenium status in healthy euthyroid postmenopausal women. J Clin Endocrinol Metab 97:4061–4070. https://doi.org/10.1210/jc.2012-2121
Pepa GD, Brandi ML (2016) Microelements for bone boost: the last but not the least. Clin Cases Miner Bone Metab 13(3):181–185. https://doi.org/10.11138/ccmbm/2016.13.3.181
Qu X, He Z, Qiao H, Zhai Z, Mao Z, Yu Z, Dai K (2018) Serum copper levels are associated with bone mineral density and total fracture. J Orthop Translat 14:34–44. https://doi.org/10.1016/j.jot.2018.05.001
Yamaguchi M (2010) Role of nutritional zinc in the prevention of osteoporosis. Mol Cell Biochem 338(1-2):241–254. https://doi.org/10.1007/s11010-009-0358-0
Grochans E, Karakiewicz B, Kozielec T, Brodowska A, Brodowski J, Starczewski A, Laszczynska M, Nocen I, Grzywacz A, Samochowiec A, Chlubek D (2011) Serum Mg and Zn levels in postmenopausal women. Magnes Res 24(4):209–214. https://doi.org/10.1684/mrh.2011.0300
D’Amelio P, Cristofaro MA, Tamone C, Morra E, Di Bella S, Isaia G, Grimaldi A, Gennero L, Gariboldi A, Ponzetto A, Pescarmona GP, Isaia GC (2008) Role of iron metabolism and oxidative damage in postmenopausal bone loss. Bone 43(6):1010–1015. https://doi.org/10.1016/j.bone.2008.08.107
Reid IR (2010) Fat and bone. Arch Biochem Biophys 503(1):20–27. https://doi.org/10.1016/j.abb.2010.06.027
Greco EA, Lenzi A, Migliaccio S (2015) The obesity of bone. Ther Adv Endocrinol Metab 6(6):273–286. https://doi.org/10.1177/2042018815611004
Fan Y, Zhang C, Bu J (2017) Relationship between selected serum metallic elements and obesity in children and adolescent in the U.S. Nutrients 9(2):104. https://doi.org/10.3390/nu9020104
Skalnaya MG, Skalny AV, Grabeklis AR, Serebryansky EP, Demidov VA, Tinkov AA (2018) Hair trace elements in overweight and obese adults in association with metabolic parameters. Biol Trace Elem Res 186(1):12–20. https://doi.org/10.1007/s12011-018-1282-5
Blazewicz A, Klatka M, Astel A, Korona-Glowniak I, Dolliver W, Szwerc W, Kocjan R (2015) Serum and urinary selenium levels in obese children: a cross-sectional study. J Trace Elem Med Biol 29:116–122. https://doi.org/10.1016/j.jtemb.2014.07.016
Celik N, Andiran N (2011) The relationship between serum phosphate levels with childhood obesity and insulin resistance. J Pediatr Endocrinol Metab 24(1-2):81–83. https://doi.org/10.1515/jpem.2011.116
Baltaci AK, Mogulkoc R, Baltaci SB (2019) The role of zinc in the endocrine system. Pak J Pharm Sci 32(1):231–239
Aigner E, Feldman A, Datz C (2014) Obesity as an emerging risk factor for iron deficiency. Nutrients 6(9):3587–3600. https://doi.org/10.3390/nu6093587
Ren XH, Yao YS, He LP, Jin YL, Chang WW, Li J, Chen Y, Song XL, Tang H, Ding LL, Guo DX, Li CP (2013) Overweight and obesity associated with increased total serum calcium level: comparison of cross-sectional data in the health screening for teaching faculty. Biol Trace Elem Res 156(1-3):74–78. https://doi.org/10.1007/s12011-013-9856-8
Kapetanovic A, Avdic D (2013) Physical activity and bone mineral density in postmenopausal women without estrogen deficiency in menstrual history. J Health Sci 2(3):205–209. https://doi.org/10.17532/jhsci.2013.108
Alghadir AH, Gabr SA, Al-Eisa ES, Alghadir MH (2016) Correlation between bone mineral density and serum trace elements in response to supervised aerobic training in older adults. Clin Interv Aging 11:265–273. https://doi.org/10.2147/CIA.S100566
Skrypnik D, Bogdanski P, Skrypnik K, Madry E, Karolkiewicz J, Szulinska M, Suliburska J, Walkowiak J (2019) Influence of endurance and endurance-strength training on mineral status in women with abdominal obesity: a randomized trial. Medicine 98(12):e14909. https://doi.org/10.1097/MD.0000000000014909
Elkomy MM, Elsaid FG (2015) Anti-osteoporotic effect of medical herbs and calcium supplementation on ovariectomized rats. JOBAZ 72:81–88. https://doi.org/10.1016/j.jobaz.2015.04.007
Mizoguchi T, Nagasawa S, Takahashi N, Yagasaki H, Ito M (2005) Dolomite supplementation improves bone metabolism through modulation of calcium-regulating hormone secretion in ovariectomized rats. J Bone Miner Metab 23(2):140–146. https://doi.org/10.1007/s00774-004-0552-9
Baltaci AK, Sunar F, Mogulkoc R, Acar M, Toy H (2014) The effect of zinc deficiency and zinc supplementation on element levels in the bone tissue of ovariectomized rats: histopathologic changes. Arch Physiol Biochem 120(2):80–85. https://doi.org/10.3109/13813455.2014.884141
Avila ME, Garcia SA, Salas MR, Antuna S, Lemini C (2009) Effect of the 17β-aminoestrogen pentolame on bone mineral levels in ovariectomized rats. Proc West Pharmacol Soc 52:43–46
Noor Z, Kania N, Setiawan B (2014) Tibia bone properties at different time course of ovariectomized rats. J Diabetes Metab Disord 13:91. https://doi.org/10.1186/s40200-014-0091-4
Wang T, Zhu X, Dai F, Li C, Huang D, Fang Z, Zhang Q, Lu Y (2017) Effects of a standard high-fat diet with or without multiple deficiencies on bone parameters in ovariectomized mature rat. PLoS One 12(9):e0184983. https://doi.org/10.1371/journal.pone.0184983
Zhang Y, Lai WP, Leung PC, Wu CF, Wong MS (2007) Short- to mid-term effects of ovariectomy on bone turnover, bone mass and bone strength in rats. Biol Pharm Bull 30(5):898–903. https://doi.org/10.1248/bpb.30.898
Sonu Y, Avinash SS, Sreekantha, Arun Kumar K, Malathi M, Shivashankara AR (2016) Effect of oestrogen on altering the serum and urinary levels of calcium, phosphate and magnesium in hysterectomised women compared to natural menopausal South Indian women: a case control study. Indian J Clin Biochem 31(3):326–331. https://doi.org/10.1007/s12291-015-0532-y
Seelig MS (1993) Interrelationship of magnesium and estrogen in cardiovascular and bone disorders, eclampsia, migraine and premenstrual syndrome. J Am Coll Nutr 12(4):442–458. https://doi.org/10.1080/07315724.1993.10718335
Vorland CJ, Lachcik PJ, Swallow EA, Metzger CE, Allen MR, Chen NX, Moe SM, Hill Gallant KM (2019) Effect of ovariectomy on the progression of chronic kidney disease-mineral bone disorder (CKD-MBD) in female Cy/+ rats. Sci Rep 9(1):7936. https://doi.org/10.1038/s41598-019-44415-9
Rahnama M, Marciniak A (2002) Influence of estrogen deficiency on the level of magnesium in rat mandible and teeth. Bull Vet Inst Pulawy 46:267–271
Ulas M, Cay M (2011) Effects of 17β-estradiol and vitamin E treatments on blood trace element and antioxidant enzyme levels in ovariectomized rats. Biol Trace Elem Res 139(3):347–355. https://doi.org/10.1007/s12011-010-8669-2
Hariri N, Thibault L (2010) High-fat diet-induced obesity in animal models. Nutr Res Rev 23(2):270–299. https://doi.org/10.1017/S0954422410000168
Shapses SA, Sukumar D (2012) Bone metabolism in obesity and weight loss. Annu Rev Nutr 32:287–309. https://doi.org/10.1146/annurev.nutr.012809.104655
Wang Y, Dellatore P, Douard V, Qin L, Watford M, Ferraris RP, Lin T, Shapses SA (2016) High fat diet enriched with saturated, but not monounsaturated fatty acids adversely affects femur, and both diets increase calcium absorption in older female mice. Nutr Res 36(7):742–750. https://doi.org/10.1016/j.nutres.2016.03.002
Cifuentes M, Morano AB, Chowdhury HA, Shapses SA (2002) Energy restriction reduces fractional calcium absorption in mature obese and lean rats. J Nutr 132(9):2660–2666. https://doi.org/10.1093/jn/132.9.2660
Xiao Y, Cui J, Shi YH, Sun J, Wang ZP, Le GW (2010) Effects of duodenal redox status on calcium absorption and related genes expression in high-fat diet-fed mice. Nutrition 26(11-12):1188–1194. https://doi.org/10.1016/j.nut.2009.11.021
Savvidis C, Tournis S, Dede AD (2018) Obesity and bone metabolism. Hormones 17:205–217. https://doi.org/10.1007/s42000-018-0018-4
Cline-Smith A, Axelbaum A, Shashkova E, Chakraborty M, Sanford J, Panesar P, Peterson M, Cox L, Baldan A, Veis D, Aurora R (2020) ovariectomy activates chronic low-grade inflammation mediated by memory T cells, which promotes osteoporosis in mice. J Bone Miner Res 35(6):1174–1187. https://doi.org/10.1002/jbmr.3966
Galloway P, McMillan DC, Sattar N (2000) Effect of the inflammatory response on trace element and vitamin status. Ann Clin Biochem 37(3):289–297. https://doi.org/10.1258/0004563001899429
Olechnowicz J, Tinkov A, Skalny A, Suliburska J (2018) Zinc status is associated with inflammation, oxidative stress, lipid, and glucose metabolism. J Physiol Sci 68(1):19–31. https://doi.org/10.1007/s12576-017-0571-7
Zofkova I, Davis M, Blahos J (2017) Trace elements have beneficial, as well as detrimental effects on bone homeostasis. Physiol Res 66(3):391–402
Zhou X, Smith AM, Failla ML, Hill KE, Yu Z (2012) Estrogen status alters tissue distribution and metabolism of selenium in female rats. J Nutr Biochem 23(6):532–538. https://doi.org/10.1016/j.jnutbio.2011.02.008
Duntas LH, Hubalewska-Dydejczyk A (2015) Selenium and inflammation - potential use and future perspectives. US Endocrinol 11(2):97–102. https://doi.org/10.17925/USE.2015.11.02.97
Rahnama M (2002) Influence of estrogen deficiency on the copper level in rat teeth and mandible. Ann Univ Mariae Curie Sklodowska Med 57(1):352–356
Tinkov AA, Gatiatulina ER, Popova EV, Polyakova VS, Skalnaya AA, Agletdinov EF, Nikonorov AA, Skalny AV (2017) Early high-fat feeding induces alteration of trace element content in tissues of juvenile male Wistar rats. Biol Trace Elem Res 175(2):367–374. https://doi.org/10.1007/s12011-016-0777-1
Doshi SB, Agarwal A (2013) The role of oxidative stress in menopause. J Midlife Health 4(3):140–146. https://doi.org/10.4103/0976-7800.118990
Marseglia L, Manti S, D’Angelo G, Nicotera A, Parisi E, Di Rosa G, Gitto E, Arrigo T (2014) Oxidative stress in obesity: a critical component in human diseases. Int J Mol Sci 16(1):378–400. https://doi.org/10.3390/ijms16010378
Thomson CD (2004) Assessment of requirements for selenium and adequacy of selenium status: a review. Eur J Clin Nutr 58(3):391–402. https://doi.org/10.1038/sj.ejcn.1601800
Balogh E, Paragh G, Jeney V (2018) Influence of iron on bone homeostasis. Pharmaceuticals (Basel) 11(4):E107. https://doi.org/10.3390/ph11040107
Raso MG, Irace C, Esposito E, Maffettone C, Iacono A, Di Pascale A, Santamaria R, Colonna A, Meli R (2009) Ovariectomy and estrogen treatment modulate iron metabolism in rat adipose tissue. Biochem Pharmacol 78(8):1001–1007. https://doi.org/10.1016/j.bcp.2009.05.034
Lecube A, Carrera A, Losada E, Hernández C, Simo R, Mesa J (2006) Iron deficiency in obese postmenopausal women. Obesity (Silver Spring) 14(10):1724–1730. https://doi.org/10.1038/oby.2006.198
Demerdash HM (2015) Obesity and trace elements. Obes Res Open J 2(3):98–100. https://doi.org/10.17140/OROJ-2-115
Stotzer US, Rodrigues MF, Domingos MM, Silva GH, Duarte FO, Gatto CV, Duarte AC, Shiguemoto GE, Perez SE, Selistre-de-Araujo HS (2015) Resistance training suppresses intra-abdominal fatty acid synthesis in ovariectomized rats. Int J Sports Med 36(3):226–233. https://doi.org/10.1055/s-0034-1390494
Yoon JR, Ha GC, Ko KJ, Kang SJ (2018) Effects of exercise type on estrogen, tumor markers, immune function, antioxidant function, and physical fitness in postmenopausal obese women. J Exerc Rehabil 14(6):1032–1040. https://doi.org/10.12965/jer.1836446.223
Al Dahamsheh Z, Al Rashdan K, Al Hadid A, Jaradat R, Al Bakheet M, Bataineh ZS (2019) The impact of aerobic exercise on female bone health indicators. Mediev Archaeol 73(1):35–38. https://doi.org/10.5455/medarh.2019.73.35-38
Baltaci AK, Uzun A, Kilic M, Mogulkoc R (2009) Effects of acute swimming exercise on some elements in rats. Biol Trace Elem Res 127(2):148–153. https://doi.org/10.1007/s12011-008-8232-6
Acknowledgements
The authors wish to thank Ms Natalia Hladikova for the linguistic corrections of this paper.
Funding
This article was created by the realization of the project “Center of excellence of environmental health”, ITMS No. 26240120033, based on the supporting Operational Research and Development Program financed from the European Regional Development Fund. The work was supported by the EEA and Norwegian Financial Mechanisms and the state budget of the Slovak Republic (Project SK0020).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethics Approval
The experiment design was prepared in accordance with the current legislation on the use of experimental animals in Slovakia. The proposal was approved by the Ethical Committee for Animal Experiment of the Slovak Medical University and by the State Veterinary and Food Authority of the Slovak Republic (Ro-1651/11-221b). All experimental procedures were carried out in animal house in compliance with the European Convention for the Protection of Vertebrate Animals used for Experimental and other Scientific Purposes.
Conflict of Interest
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Masanova, V., Krivosikova, Z., Ursinyova, M. et al. Effects of Ovariectomy and Exercise Training on Mineral Status in a High-Fat Diet-Induced Obesity Rat Model. Biol Trace Elem Res 200, 624–634 (2022). https://doi.org/10.1007/s12011-021-02655-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12011-021-02655-9