Abstract
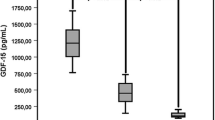
Iron is one of the most important trace elements in the body, and its homeostasis is essential to the normal function of the immune system. Complement component C3, which is the converging of three main pathways of complement system activation, plays a key role in the innate immunity. However, the relationship between iron homeostasis and complement C3 remains unknown. The aim of our study was to analyze the relationship between serum iron and ferritin level and complement C3 and C4. A total of 590 healthy individuals were recruited in our study. Higher serum complement C3 level (p < 0.001) was found in individuals with higher serum ferritin level (> 104.0 μg/L). Moreover, serum iron level and serum ferritin level were positively correlated with complement C3 (r = 0.133, p = 0.001; r = 0.221, p < 0.001) and complement C4 (r = 0.117, p = 0.004; r = 0.123, p = 0.003). The linear regression analysis displayed that both serum iron level and serum ferritin level were linearly correlated with serum complement C3 level (adjusted beta: 2.382, 95% CI: 0.841–3.923; adjusted beta: 42.911, 95% CI: 29.070–56.751). To explore the relationship between iron homeostasis and complement C3 further, the serum samples from C3−/− mice and the wild-type (WT) control mice were obtained. Significantly lower serum iron level and higher ferritin level were found in C3−/− mice than those in WT mice (p < 0.001; p < 0.001), indicating that complement C3 might influence iron distribution and utilization. Overall, these data suggested that serum iron and ferritin levels were correlated with complement C3. The deficiency of complement C3 may disrupt the regular iron metabolism in the body.
Similar content being viewed by others
References
Andrews NC, Schmidt PJ (2007) Iron homeostasis. Annu Rev Physiol 69:69–85. https://doi.org/10.1146/annurev.physiol.69.031905.164337
Abbaspour N, Hurrell R, Kelishadi R (2014) Review on iron and its importance for human health. J Res Med Sci 19(2):164–174
Beard JL (2001) Iron biology in immune function, muscle metabolism and neuronal functioning. J Nutr 131(2s-2):568S–579S; discussion 580S. https://doi.org/10.1093/jn/131.2.568S
Ekiz C, Agaoglu L, Karakas Z, Gurel N, Yalcin I (2005) The effect of iron deficiency anemia on the function of the immune system. Hematol J 5(7):579–583. https://doi.org/10.1038/sj.thj.6200574
Galan P, Thibault H, Preziosi P, Hercberg S (1992) Interleukin 2 production in iron-deficient children. Biol Trace Elem Res 32:421–426. https://doi.org/10.1007/bf02784628
Kuvibidila SR, Kitchens D, Baliga BS (1999) In vivo and in vitro iron deficiency reduces protein kinase C activity and translocation in murine splenic and purified T cells. J Cell Biochem 74(3):468–478
Walker EM Jr, Walker SM (2000) Effects of iron overload on the immune system. Ann Clin Lab Sci 30(4):354–365
Ganz T, Nemeth E (2015) Iron homeostasis in host defence and inflammation. Nat Rev Immunol 15(8):500–510. https://doi.org/10.1038/nri3863
He L, Qian Y, Ren X, Jin Y, Chang W, Li J, Chen Y, Song X, Tang H, Ding L, Guo D, Yao Y (2014) Total serum calcium level may have adverse effects on serum cholesterol and triglycerides among female university faculty and staffs. Biol Trace Elem Res 157(3):191–194. https://doi.org/10.1007/s12011-014-9895-9
Yao Y, He L, Jin Y, Chen Y, Tang H, Song X, Ding L, Qi Q, Huang Z, Wang Q, Yu J (2013) The relationship between serum calcium level, blood lipids, and blood pressure in hypertensive and normotensive subjects who come from a normal university in east of China. Biol Trace Elem Res 153(1–3):35–40. https://doi.org/10.1007/s12011-013-9646-3
Ward PP, Mendoza-Meneses M, Cunningham GA, Conneely OM (2003) Iron status in mice carrying a targeted disruption of lactoferrin. Mol Cell Biol 23(1):178–185. https://doi.org/10.1128/mcb.23.1.178-185.2003
Schubert TE, Bosserhoff AK, Peyssonaux C, Echtenacher B, Knutson M, Hofstädter F, Männel DN (2012) Hypoferraemia during the early inflammatory response is dependent on tumour necrosis factor activity in a murine model of protracted peritonitis. Mol Med Rep 6(4):838–842. https://doi.org/10.3892/mmr.2012.1004
Marques O, Neves J, Horvat NK, Colucci S, Guida C, Muckenthaler MU (2019) Iron-related parameters are altered between C57BL/6N and C57BL/6J Mus musculus wild-type substrains. HemaSphere 3(6):e304. https://doi.org/10.1097/hs9.0000000000000304
Ferreira C, Santambrogio P, Martin ME, Andrieu V, Feldmann G, Hénin D, Beaumont C (2001) H ferritin knockout mice: a model of hyperferritinemia in the absence of iron overload. Blood 98(3):525–532. https://doi.org/10.1182/blood.v98.3.525
He L, Zhang Y, Ru D, Xue B, Wen S, Zhou H (2020) Serum iron levels are negatively correlated with serum triglycerides levels in female university students. Ann Palliat Med. https://doi.org/10.21037/apm.2020.03.02
Brittenham GM (2011) Iron-chelating therapy for transfusional iron overload. N Engl J Med 364(2):146–156. https://doi.org/10.1056/NEJMct1004810
Sheftel AD, Mason AB, Ponka P (2012) The long history of iron in the universe and in health and disease. Biochim Biophys Acta 1820(3):161–187. https://doi.org/10.1016/j.bbagen.2011.08.002
Adams PC, Barton JC (2011) A diagnostic approach to hyperferritinemia with a non-elevated transferrin saturation. J Hepatol 55(2):453–458. https://doi.org/10.1016/j.jhep.2011.02.010
Broedbaek K, Poulsen HE, Weimann A, Kom GD, Schwedhelm E, Nielsen P, Böger RH (2009) Urinary excretion of biomarkers of oxidatively damaged DNA and RNA in hereditary hemochromatosis. Free Radic Biol Med 47(8):1230–1233. https://doi.org/10.1016/j.freeradbiomed.2009.08.004
Akter S, Nanri A, Kuwahara K, Matsushita Y, Nakagawa T, Konishi M, Honda T, Yamamoto S, Hayashi T, Noda M, Mizoue T (2017) Circulating ferritin concentrations and risk of type 2 diabetes in Japanese individuals. J Diabetes Investig 8(4):462–470. https://doi.org/10.1111/jdi.12617
Kell DB, Pretorius E (2014) Serum ferritin is an important inflammatory disease marker, as it is mainly a leakage product from damaged cells. Metallomics : Integr Biometal Sci 6(4):748–773. https://doi.org/10.1039/c3mt00347g
Fernández-Real JM, Peñarroja G, Castro A, García-Bragado F, Hernández-Aguado I, Ricart W (2002) Blood letting in high-ferritin type 2 diabetes: effects on insulin sensitivity and beta-cell function. Diabetes 51(4):1000–1004. https://doi.org/10.2337/diabetes.51.4.1000
Li Y (2015) Iron-induced Complement Dysregulation in the Retinal Pigment Epithelium: Implications for Age-Related Macular Degeneration Publicly Accessible Penn Dissertations 1088
Li Y, Song D, Song Y, Zhao L, Wolkow N, Tobias JW, Song W, Dunaief JL (2015) Iron-induced local complement component 3 (C3) up-regulation via non-canonical transforming growth factor (TGF)-beta signaling in the retinal pigment epithelium. J Biol Chem 290(19):11918–11934. https://doi.org/10.1074/jbc.M115.645903
Tang S, Lai KN, Chan TM, Lan HY, Ho SK, Sacks SH (2001) Transferrin but not albumin mediates stimulation of complement C3 biosynthesis in human proximal tubular epithelial cells. Am J Kidney Dis 37(1):94–103. https://doi.org/10.1053/ajkd.2001.20593
Moen IW, Bergholdt HKM, Mandrup-Poulsen T, Nordestgaard BG, Ellervik C (2018) Increased plasma ferritin concentration and low-grade inflammation-a Mendelian randomization study. Clin Chem 64(2):374–385. https://doi.org/10.1373/clinchem.2017.276055
Hansen JB, Tonnesen MF, Madsen AN, Hagedorn PH, Friberg J, Grunnet LG, Heller RS, Nielsen A, Størling J, Baeyens L, Anker-Kitai L, Qvortrup K, Bouwens L, Efrat S, Aalund M, Andrews NC, Billestrup N, Karlsen AE, Holst B, Pociot F, Mandrup-Poulsen T (2012) Divalent metal transporter 1 regulates iron-mediated ROS and pancreatic β cell fate in response to cytokines. Cell Metab 16(4):449–461. https://doi.org/10.1016/j.cmet.2012.09.001
Nemeth E, Rivera S, Gabayan V, Keller C, Taudorf S, Pedersen BK, Ganz T (2004) IL-6 mediates hypoferremia of inflammation by inducing the synthesis of the iron regulatory hormone hepcidin. J Clin Invest 113(9):1271–1276. https://doi.org/10.1172/jci20945
Trombini P, Paolini V, Pelucchi S, Mariani R, Nemeth E, Ganz T, Piperno A (2011) Hepcidin response to acute iron intake and chronic iron loading in dysmetabolic iron overload syndrome. Liver Int 31(7):994–1000. https://doi.org/10.1111/j.1478-3231.2011.02520.x
Jayaweera J, Reyes M, Joseph A (2019) Childhood iron deficiency anemia leads to recurrent respiratory tract infections and gastroenteritis. Sci Rep 9(1):12637. https://doi.org/10.1038/s41598-019-49122-z
Thurnham DI, McCabe LD, Haldar S, Wieringa FT, Northrop-Clewes CA, McCabe GP (2010) Adjusting plasma ferritin concentrations to remove the effects of subclinical inflammation in the assessment of iron deficiency: a meta-analysis. Am J Clin Nutr 92(3):546–555. https://doi.org/10.3945/ajcn.2010.29284
Acknowledgments
We thank Professor Hong Zhou for his generous support.
Funding
This study was supported by The Natural Science Foundation of the Jiangsu Higher Education Institutions of China (Grant No. 19KJB310007).
Author information
Authors and Affiliations
Contributions
Dongyan Shi and Yeqin Sha contributed to the design of the project. Zhilian Rui, Yuning Zhou, Fengfeng yan, Jingming Hu, Chenhong Si, and Xu Han contributed to data collection. Yeqin Sha, Shuang Wen, and Yinling Li contributed to data analysis. Yeqin Sha and Bicheng Wang wrote the first draft of the manuscript. All authors reviewed the manuscript.
Corresponding author
Ethics declarations
The studies were approved by the Institutional Research Board of Nanjing Medical University and were performed in accordance with the principles of the Declaration of Helsinki. All experimental protocols were approved by the Laboratory Animal Care and Use Committee at Nanjing Medical University (NJMUAC No. 053/2016).
Conflict of Interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Wen, S., Sha, Y., Li, Y. et al. Serum Iron and Ferritin Levels Are Correlated with Complement C3. Biol Trace Elem Res 199, 2482–2488 (2021). https://doi.org/10.1007/s12011-020-02379-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12011-020-02379-2