Abstract
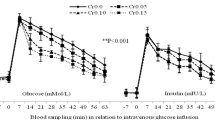
Dairy cows suffer insulin resistance following parturition and lactogenesis. Several researchers attempted to reduce insulin resistance via dietary and parenteral supplementations of different substances to promote metabolic performance of dairy cows. Due to mechanisms of actions of butaphosphan in combination with cyanocobalamin, we hypothesized that this compound may reduce insulin resistance of dairy cows following parturition; hence, the effects of the intravenous administration of butaphosphan and cyanocobalamin to prepartum dairy cows on their insulin resistance after calving were evaluated. Twenty-four multiparous Holstein dairy cows were enrolled 3 weeks prior to parturition and divided into four equal groups, including control (Ctrl) and butaphosphan and cyanocobalamin (B+C) 1, 2, and 3. Ctrl cows received 15 mL of 0.9% NaCl solution and B+C 1, 2, and 3 groups intravenously received 2, 4, and 6 mL/100 kg BW of 10% butaphosphan and 0.005% cyanocobalamin combination over three periods of 3 consecutive days, including 21-19, 12-10, and 3-1 days before calving, respectively. Intravenous glucose tolerance test was performed weekly 1, 2, and 3 weeks after parturition to evaluate the insulin resistance phenomenon. Circulating levels of glucose, insulin, non-esterified fatty acids (NEFA), and beta-hydroxybutyric acid (BHBA) were assessed 1, 2, and 3 weeks after calving. Ctrl cows were the most insulin-resistant group, and B+C1 group was the most insulin-sensitive, followed by B+C2 and B+C3 groups. The NEFA and BHBA levels in the B+C3 group were significantly lower than those in the other groups. In conclusion, intravenous administration of butaphosphan and cyanocobalamin to the late-pregnant dairy cows may reduce their insulin resistance after calving.

Similar content being viewed by others
References
Patton J, Kenny DA, Mcnamara S, Mee JF, O’Mara FP, Diskin MG, Murphy JJ (2007) Relationships among milk production, energy balance, plasma analytes, and reproduction in holstein-friesian cows. J Dairy Sci 90:649–658. https://doi.org/10.3168/jds.S0022-0302(07)71547-3
Castaneda-Gutierrez E, Pelton SH, Gilbert RO, Butler WR (2009) Effect of peripartum dietary energy supplementation of dairy cows on metabolites, liver function and reproductive variables. Anim Reprod Sci 112:301–315. https://doi.org/10.1016/j.anireprosci.2008.04.028
Mattmiller SA, Corl CM, Gandy JC, Loor JJ, Sordillo LM (2011) Glucose transporter and hypoxiaassociated gene expression in the mammary gland of transition dairy cattle. J Dairy Sci 94:2912–2922. https://doi.org/10.3168/jds.2010-3936
Chagas LM, Lucy MC, Back PJ, Blache D, Lee JM, Gore PJ, Sheahan AJ, Roche JR (2009) Insulin resistance in divergent strains of holstein-friesian dairy cows offered fresh pasture and increasing amounts of concentrate in early lactation. J Dairy Sci 92:216–222. https://doi.org/10.3168/jds.2008-1329
Zhang ZG, Wang JG, Gao RF, Zhang W, Li X, Liu G, Li X, Wang Z, Zhu X (2013) High-energy diet at antepartum decreases insulin receptor gene expression in adipose tissue of postpartum dairy cows. Bull Vet Inst Pulawy 57:203–207. https://doi.org/10.2478/bvip-2013-0037
Tsuruzoe K, Emkey R, Kriauciunas KM, Ueki K, Kahn CR (2001) Insulin receptor substrate 3 (IRS-3) and IRS-4 impair IRS-1- and IRS-2-mediated signaling. J Mol Cell Biol 21:26–38. https://doi.org/10.1128/MCB.21.1.26-38.2001
Ning J, Hong T, Yang X, Mei S, Liu Z, Liu H, Cao W (2011) Insulin and insulin signaling play a critical role in fat induction of insulin resistance in mouse. Am J Physiol Endocrinol Metab 301:391–401. https://doi.org/10.1152/ajpendo.00164.2011
Esposito G, Irons PC, Webb EC, Chapwanya A (2014) Interactions between negative energy balance, metabolic diseases, uterine health and immune response in transition dairy cows. Anim Reprod Sci 144:60–71. https://doi.org/10.1016/j.anireprosci.2013.11.007
Overton TR, Waldron MR (2004) Nutritional management of transition dairy cows: strategies to optimize metabolic health. J Dairy Sci 87:105–119. https://doi.org/10.3168/jds.S0022-0302(04)70066-1
Chibisa GE, Gozho GN, Van Kessel AG, Olkowski AA, Mutsvangwa T (2008) Effects of peripartum propylene glycol supplementation on nitrogen metabolism, body composition, and gene expression for the major protein degradation pathways in skeletal muscle in dairy cows. J Dairy Sci 91:3512–3527. https://doi.org/10.3168/jds.2007-0920
Chalmeh A, Hajimohammadi A, Nazifi S (2015) Endocrine and metabolic responses of high producing Holstein dairy cows to glucose tolerance test based on the stage of lactation. Livest Sci 181:179–186. https://doi.org/10.1016/j.livsci.2015.09.014
Kreipe L, Deniz A, Bruckmaier RM, Van Dorland HA (2011) First report about the mode of action of combined butafosfan and cyanocobalamin on hepatic metabolism in nonketotic early lactating cows. J Dairy Sci 94:4904–4914. https://doi.org/10.3168/jds.2010-4080
Pereira RA, Silveira PAS, Montagner P, Schneider A, Schmitt E, Rabassa VR, Pfeifer LF, Del Pino FA, Pulga ME, Corrêa MN (2013) Effect of butaphosphan and cyanocobalamin on postpartum metabolism and milk production in dairy cows. Animal 7:1143–1147. https://doi.org/10.1017/S1751731113000013
Furll M, Deniz A, Westphal B, Illing C, Constable PD (2010) Effect of multiple intravenous injections of butaphosphan and cyanocobalamin on the metabolism of periparturient dairy cows. J Dairy Sci 93:4155–4164. https://doi.org/10.3168/jds.2009-2914
Rollin E, Berghaus RD, Rapnicki P, Godden SM, Overton MW (2010) The effect of injectable butaphosphan and cyanocobalamin on postpartum serum beta-hydroxybutyrate, calcium, and phosphorus concentrations in dairy cattle. J Dairy Sci 93:978–987. https://doi.org/10.3168/jds.2009-2508
Kincaid RL, Socha MT (2007) Effect of cobalt supplementation during late gestation and early lactation on milk and serum measures. J Dairy Sci 90:1880–1886. https://doi.org/10.3168/jds.2006-296
Kennedy DG, Cannavan A, Molloy A, O’Harte F, Taylor SM, Kennedy S, Blanchflower WJ (1990) Methylmalonyl-CoA mutase (EC 5.4.99.2) and methionine synthetase (EC 2.1.1.13) in the tissues of cobalt-vitamin 12 deficient sheep. Br J Nutr 64:721–732. https://doi.org/10.1079/bjn19900074
Preynat A, Lapierre H, Thivierge MC, Palin MF, Matte JJ, Desrochers A, Girard CL (2009) Effects of supplements of folic acid, vitamin B12, and rumen-protected methionine on whole body metabolism of methionine and glucose in lactating dairy cows. J Dairy Sci 92:677–689. https://doi.org/10.3168/jds.2008-1525
Girard CL, Matte JJ (2005) Effects of intramuscular injections of vitamin B12 on lactation performance of dairy cows fed dietary supplements of folic acid and rumen-protected methionine. J Dairy Sci 88:671–676. https://doi.org/10.3168/jds.S0022-0302(05)72731-4
Akins MS, Bertics SJ, Socha MT, Shaver RD (2013) Effects of cobalt supplementation and vitamin B(12) injections on lactation performance and metabolism of Holstein dairy cows. J Dairy Sci 96:1755–1768. https://doi.org/10.3168/jds.2012-5979
Klopcic M, Hamoen A, Bewley J (2011) Body condition scoring of dairy cows. University of Ljubljana, Ljubljana
Itle AJ, Huzzey JM, Weary DM, von Keyserlingk MA (2015) Clinical ketosis and standing behavior in transition cows. J Dairy Sci 98:128–134. https://doi.org/10.3168/jds.2014-7932
De Koster JD, Opsomer G (2013) Insulin resistance in dairy cows. Vet Clin North Am Food Anim Pract 29:299–322. https://doi.org/10.1016/j.cvfa.2013.04.002
González-Grajales LA, Pieper L, Mengel S, Staufenbiel R (2018) Evaluation of glucose dose on intravenous glucose tolerance test traits in Holstein-Friesian heifers. J Dairy Sci 101:774–782. https://doi.org/10.3168/jds.2017-13215
Beever DE (2006) The impact of controlled nutrition during the dry period on dairy cow health, fertility and performance. Anim Reprod Sci 96:212–226. https://doi.org/10.1016/j.anireprosci.2006.08.002
Grundy SM, Abate N, Chandalia M (2002) Diet composition and the metabolic syndrome: what is the optimal fat intake? Am J Med Sci 30:25S–29S. https://doi.org/10.1016/s0002-9343(01)00988-3
Diamant M, Blaak EE, de Vos WM (2011) Do nutrient-gut-microbiota interactions play a role in human obesity, insulin resistance and type 2 diabetes? Obes Rev 12:272–281. https://doi.org/10.1111/j.1467-789X.2010.00797.x
Chalmeh A, Pourjafar M, Badiei K, Jalali M, Mazrouei Sebdani M (2020) The comparative effects of dietary monensin and propylene glycol on insulin resistance of transition dairy cows. Trop Anim Health Prod; In Press 52:1573–1582. https://doi.org/10.1007/s11250-019-02160-1
Weaver SR, Prichard AS, Maerz NL, Prichard AP, Endres EL, Hernández-Castellano LE, Akins MS, Bruckmaier RM, Hernandez LLL (2017) Elevating serotonin pre-partum alters the Holstein dairy cow hepatic adaptation to lactation. PLoS One 12:e0184939. https://doi.org/10.1371/journal.pone.0184939
Nuber U, van Dorland HA, Bruckmaier RM (2016) Effects of butafosfan with and without cyanocobalamin on the metabolism of early lactating cows with subclinical ketosis. Am J Physiol Endocrinol Metab 100:146–155. https://doi.org/10.1111/jpn.12332
Hayirli A (2006) The role of exogenous insulin in the complex of hepatic lipidosis and ketosis associated with insulin resistance phenomenon in postpartum dairy cattle. Vet Res Commun 30:749–774. https://doi.org/10.1007/s11259-006-3320-6
Berg JM, Tymoczko JL, Stryer L (2006) Glycolysis and gluconeogenesis. In: Berg JM, Tymoczko JL, Stryer L (eds) Biochemistry, 6th edn. W. H. Freeman and Co, New York, pp 433–474
Girard CL, Lapierre H, Matte JJ, Lobley GE (2005) Effects of dietary supplements of folic acid and rumen protected methionine on lactational performance and folate metabolism of dairy cows. J Dairy Sci 88:660–670. https://doi.org/10.3168/jds.S0022-0302(05)72730-2
Graulet B, Matte JJ, Desrochers A, Doepel L, Palin MF, Girard CL (2007) Effects of dietary supplements of folic acid and vitamin B12 on metabolism of dairy cows in early lactation. J Dairy Sci 90:3442–3455. https://doi.org/10.3168/jds.2006-718
Girard CL, Matte JJ (2005) Folic acid and vitamin B12 requirements of dairy cows: a concept to be revised. Livest Prod Sci 98:123–133. https://doi.org/10.1016/j.livprodsci.2005.10.009
Kennedy DG, Young PB, Blanchflower WJ, Scott JM, Weir DG, Molloy AM, Kennedy S (1994) Cobalt-vitamin B12 deficiency causes lipid accumulation, lipid peroxidation and decreased α-tocopherol concentrations in the liver of sheep. Int J Vitam Nutr Res 64:270–276
Longo N, Frigeni M, Pasquali M (2016) Carnitine transport and fatty acid oxidation. Biochim Biophys Acta 1863:2422–2435. https://doi.org/10.1016/j.bbamcr.2016.01.023
Takahashi-Iñiguez T, García-Hernandez E, Arreguín-Espinosa R, Flores ME (2012) Role of vitamin B12 on methylmalonyl-CoA mutase activity. J Zhejiang Univ Sci B 13:423–437. https://doi.org/10.1631/jzus.B1100329
Girard CL, Matte JJ (1999) Changes in serum concentrations of folates, pyridoxal, pyridoxal-5-phosphate and vitamin B12 during lactation of dairy cows fed dietary supplements of folic acid. Can J Anim Sci 79:107–113. https://doi.org/10.4141/A98-016
McDowell LR (2000) Vitamins in animal and human nutrition, 2nd edn. Iowa State University Press, Ames
Stangl GI, Schwarz FJ, Müller H, Kirchgessner M (2000) Evaluation of the cobalt requirement of beef cattle based on vitamin B12, folate, homocysteine and methylmalonic acid. Br J Nutr 84:645–653. https://doi.org/10.1017/s0007114500001987
Kincaid RL, Lefebvre LE, Cronrath JD, Socha MT, Johnson AB (2003) Effect of dietary cobalt supplementation on cobalt metabolism and performance of dairy cattle. J Dairy Sci 86:1405–1414. https://doi.org/10.3168/jds.S0022-0302(03)73724-2
Tiffany ME, Fellner V, Spears JW (2006) Influence of cobalt concentrations on vitamin B12 production and fermentation of mixed microorganisms grown in continuous culture flow-through fermentors. J Anim Sci 84:635–640. https://doi.org/10.2527/2006.843635x
Fiore E, Gianesella M, Arfuso F, Giudice E, Piccione G, Lora M, Stefani A, Morgante M (2014) Glucose infusion response on some metabolic parameters in dairy cows during transition period. Arch Tierzucht 57:1–9. https://doi.org/10.7482/0003-9438-57-003
Terao H, Fujita M, Tsumagari A, Suginob T, Bungo T (2010) Insulin dynamics in transition dairy cows as revealed by intravenous glucose tolerance testing. J Anim Vet Adv 9:2333–2337. https://doi.org/10.3923/javaa.2010.2333.2337
Gordon JL, Duffield TF, Herdt TH, Kelton DF, Neuder L, LeBlanc SJ (2017) Effects of a combination butaphosphan and cyanocobalamin product and insulin on ketosis resolution and milk production. J Dairy Sci 100:2954–2966. https://doi.org/10.3168/jds.2016-11925
Temizel EM, Batmaz H, Keskin A, Orman A, Gencoglu H, Catık S, Topal O (2015) Butaphosphan and cyanocobalamin treatment of pregnant ewes: metabolic effects and potential prophylactic effect for pregnancy toxaemia. Small Rumin Res 125:163–172. https://doi.org/10.1016/j.smallrumres.2015.02.016
Tabeleão VC, Schwegler E, Pereira RA, Krause ART, Montagner P, Feijó JO, Schneider A, Schmitt E, Brauner CC, Rabassa VR, Pino FD, Corrêa MN (2017) Combined of butaphosphan and cyanocobalamin on the glucose metabolism of dairy cows after calving. Arq Bras Med Vet Zootec 69:317–324. https://doi.org/10.1590/1678-4162-8453
Acknowledgments
We would like to thank Mr. Javad Chalmeh for providing the cows.
Funding
The authors received financial support from Shiraz University.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Chalmeh, A., Pourjafar, M., Badiei, K. et al. Intravenous Administration of Butaphosphan and Cyanocobalamin Combination to Late-Pregnant Dairy Cows Reduces Their Insulin Resistance After Calving. Biol Trace Elem Res 199, 2191–2200 (2021). https://doi.org/10.1007/s12011-020-02330-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12011-020-02330-5