Abstract
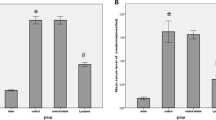
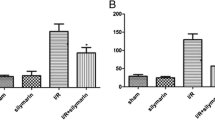
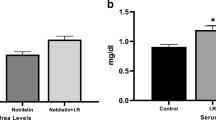
Ischemia/reperfusion (I/R) injury is associated with a strong inflammatory and oxidative stress response to hypoxia and reperfusion that impair organ function. We aimed to investigate the role of oxidative stress, renal inflammation, and apoptosis in the injury of the kidney tissue after ischemic reperfusion, and the protective effect of dose-dependent boric acid administration. For this purpose, 35 Sprague Dawley albino rats were divided into five groups of seven animals in each group: Sham, I/R and I/R + boric acid (BA) (i.p at doses of 50, 100, and 200 mg/kg). All animals underwent nephrectomy (the right kidney was removed) and were expected to recover for 15 days. After recovery, each animal received 45 min of ischemia. BA was injected intraperitoneally 10 min before reperfusion and a 24-h reperfusion procedure was performed. Sham group only underwent surgical stress procedure. In order to investigate the oxidative stress induced by I/R injury and antioxidant effects of different BA doses in the kidney tissue, TAS, TOS, MDA, SOD, CAT, and GSH levels were measured. DNA fragmentation, cytochrome C levels, caspase 3 activity were measured to determine apoptotic index in tissue. IL-6 and TNF-α levels were measured in the evaluation of inflammation. Hematoxylin-eosin and TUNEL staining was performed for histopathological examinations. As a result, increased oxidative stress, inflammation, and apoptosis after I/R were decreased with different doses of BA treatment. The application of high-dose BA was found to be lower in anti-apoptotic, anti-inflammatory, and antioxidant effects than in the low-dose groups.




Similar content being viewed by others
References
Jochmans I, Meurisse N, Neyrinck A, Verhaegen M, MonbaliuD PJ (2017) Hepatic ischemia/reperfusion injury associates with acute kidney injury in liver transplantation: prospective cohort study. Liver Transpl 23:634–644. https://doi.org/10.1002/lt.24728
Neri M, Riezzo I, Pascale N, Pomara C, Turillazzi E (2017) Ischemia/reperfusion injury following acute myocardial infarction: a critical issue for clinicians and forensic pathologists. Mediat Inflamm 2017:1–14. https://doi.org/10.1155/2017/7018393
Wu MY, Yiang GT, Liao WT, Tsai AP, Cheng YL, Cheng PW (2018) Current mechanistic concepts in ischemia and reperfusion injury. Cell Physiol Biochem 46:1650–1667. https://doi.org/10.1159/000489241
Uyanoğlu M, Canbek M, Şentürk H, Bayramoğlu G, Gündüz Ö, Özen A ve Turgak Ö (2011) Preventing organ injury with carvacrol after renal ischemia/reperfusion. 5:2–80
Kelly KJ, Plotkin Z, Dagher PC (2001) Guanosine supplementation reduces apoptosis and protects renal function in the setting of ischemic injury. J Clin Investig 108:1291–1298. https://doi.org/10.1172/JCI13018
Marko L, Vigolo E, Hinze C, Park JK, Roel G, Balogh A, Choi M, Wubken A, Cording J, Blasig IE, Luft FC, Scheidereit C, Schmidt-Ott KM, Schmidt-Ullrich R, Muller DN (2016) Tubular epithelial NF-kappaB activity regulates ischemic AKI. J Am Soc Nephrol 27:2658–2669. https://doi.org/10.1681/ASN.2015070748
Zhao L, Xu L, Tao X, Han X, Yin L, Qi Y, Peng J (2016) Protective effect of the total flavonoids from Rosa laevigata Michx fruit on renal ischemia-reperfusion injury through suppression of oxidative stress and inflammation. Molecules 21:952. https://doi.org/10.3390/molecules21070952
Weng XF, Li ST, Song Q, Zhu Q, Song DD, Qin ZH, Xie Y (2018) Protective effect of nicotinamide adenine dinucleotide phosphate on renal ischemia-reperfusion injury. Kidney Blood Press Res 43:651–663. https://doi.org/10.1159/000489620
Khaliq H, Juming Z, Ke-Mei P (2018) The physiological role of boron on health. Biol Trace Elem Res 186:31–51. https://doi.org/10.1007/s12011-018-1284-3
Ince S, Kucukkurt I, Cigerci IH, Fidan AF, Eryavuz A (2010) The effects of dietary boric acid and borax supplementation on lipid peroxidation, antioxidant activity, and DNA damage in rats. J Trace Elem Med Biol 24:161–164. https://doi.org/10.1016/j.jtemb.2010.01.003
Hunt CD (1994) The biochemical effects of physiologic amount of dietary boron in animal nutrition models. Environ Health Perspect 102:35–43. https://doi.org/10.1289/ehp.94102s735
Nielsen FH (1991) Nutritional requirements for boron, silicon, vanadium, nickel and arsenic: current knowledge and speculation. FASEB J 5:2661–2667. https://doi.org/10.1096/fasebj.5.12.1916090
Hunt CD (1998) Regulation of enzymatic activity: one possible role of dietary boron in higher animals and humans. Biol Trace Elem Res 66:205–225
Cao J, Jiang L, Zhang X, Yao X, Geng C, Xue X, Zhong L (2008) Boric acid inhibits LPS-induced TNF-alpha formation through a thiol-dependent mechanism in THP-1 cells. J Trace Elem Med Biol 22:189–195
Basbug M, Yildar M, Yaman I, Ozkan OF, Aksit H, Cavdar F et al (2015) Effects of boric acid in an experimental rat model of hepatic ischemia-reperfusion injury. Acta Medica Mediterr 31:1067–1073
Lévigne D, Tobalem M, Modarressi A, Pittet-Cuénod B (2013) Hyperglycemia increases susceptibility to ischemic necrosis. Biomed Res Int 2013:1–5.31
Geyikoglu F, Koc K, Colak S, Erol HS, Cerig S, Yardimci BK et al (2019) Propolis and its combination with boric acid protect against ischemia/reperfusion-induced acute kidney injury by inhibiting oxidative stress, inflammation, DNA damage, and apoptosis in rats. Biol Trace Elem Res 1–8
Ince S, Keles H, Erdogan M, Hazman O, Kucukkurt I (2012) Protective effect of boric acid against carbon tetrachloride-induced hepatotoxicity in mice. Drug Chem Toxicol 35(3):285–292
Sogut I, Oglakci A, Kartkaya K, Ol KK, Sogut MS, Kanbak G, Inal ME (2015) Effect of boric acid on oxidative stress in rats with fetal alcohol syndrome. Exp Ther Med 9(3):1023–1027
Ince S, Kücükkurt I, Demirel HH, Acaröz DA, Akbel E, Ciğerci IH (2014) Protective effects of boron on cyclophosphamide induced lipid peroxidation and genotoxicity in rats. Chemosphere 108:197–204
Turkez H, Geyikoglu F, Tatar A, Keles S, Ozkanc A (2007) Effects of some boron compounds on peripheral human blood. Z Naturforsch 62:889–896
Mohora M, Boghianu L, Muscurel C, Duta C, Dumitrache C (2002) Effects of boric acid on redox status in the rat liver. Romanian J Biophys 12:77–82
Saravan R, Pugalendi KV (2005) Assessment of the pharmacological effect of silymarin on ethanol induced DNA damage by sindle cell gelelectrophoresis. Indian J Pharmacol 37:261–262
Arumugam TV, Shiels IA, Woodruff TM, Granger DN, Taylor SM (2004) The role of the complement system in ischemia-reperfusion injury. Shock 21(5):401–409
Hazman Ö, Bozkurt MF, Fidan AF, Uysal FE, Çelik S (2018) The effect of boric acid and borax on oxidative stress, inflammation, ER stress and apoptosis in cisplatin toxication and nephrotoxicity developing as a result of toxication. Inflammation 41(3):1032–1048
Bahadoran H, Naghii MR, Mofid M, Asadi MH, Ahmadi K, Sarveazad A (2016) Protective effects of boron and vitamin E on ethylene glycol-induced renal crystal calcium deposition in rat. Endocr Regul 50:194–206
Ergul AB, Kara M, Karakukcu C, Tasdemir A, Aslaner H, Ergul MA, Muhtaroglu S, Zararsiz GE, Torun YA (2018) High doses of boron have no protective effect against nephrolithiasis or oxidative stress in a rat model. Biol Trace Elem Res 186(1):218–225
Okhawa H, Ohishi N, Yagi K (1979) Assay for lipid peroxides in animal tissues by thiobarbituric acid reaction. Anal Biochem 95:351–358
Beutler E, Duron O, Kelly BM (1963) Improved method for the determination of blood glutathione. J Lab Clin Med 61:882–890
Aebi H (1984) Catalase in vitro. Methods Enzymol 105:121–126
Winterbourn CC, Hawkins RE, Brian M, Carrell RW (1975) The estimation of red cell superoxide dismutase activity. J Lab Clin Med 85:337–341
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254. https://doi.org/10.1016/0003-2697(76)90527-3
Wyllie AH (1980) Glucocorticoid-induced thymocyte apoptosis is associated with endogenous endonuclease activation. Nature 284(5756):555–556
Şentürk H, Kar F, Hacıoğlu C, Kanbak G Renal İskemi Reperfüzyon ile İndüklenmiş Oksidatif Stres Hasarının Pankreas Üzerine Etkisi Doza Bağımlı Borik Asidin Rolü. Kahramanmaraş Sütçü İmam Üniversitesi Tarım ve Doğa Dergisi 21:944–949. https://doi.org/10.18016/ksutarimdoga.vi.430369
Friedewald JJ, Rabb H (2004) Inflammatory cells in ischemic acute renal failure. Kidney Int 66:486–491
Holderied A, Andersen NH (2014) Animal models of kidney inflammation in translational medicine. Drug Discov Today Dis Model 11:19–27.24
Bonventre JV, Zuk A (2004) Ischemic acute renal failure: an inflammatory disease? Kidney Int 66:480–485
Bonventre JV, Yang L (2011) Cellular pathophysiology of ischemic acute kidney injury. J Clin Invest 121:4210–21.26
Kar F, Hacıoğlu C, Kar E, Kara Y, Şentürk H, Kanbak G (2018) Renal ischemia reperfusion effect on spleen asremote tissue damage and role of boric acid. Presented At The Tbs International Biochemistry Congress
Hacıoğlu C, Kar F, Kar E, Kara Y, Şentürk H, Kanbak G (2018) Neuroprotectıve effects of boric acid on brain against renal ischemia reperfusion injury. Presented At The Tbs International Biochemistry Congress
Kara Y, Hacıoğlu C, Kar F, Kar E, Şentürk H, Kanbak G (2018) Effects of boric acid on heart tissue damage caused by renal ischemia/reperfusion. Presented At The Tbs International Biochemistry Congress
Hacıoğlu C, Kar F, Şentürk H, Kanbak G (2018) Effects of boric acid on electrolyte balance and lipid profile against renal ischemia reperfusion injury. Biol Divers Conserv 11:76–81 ISSN 1308-8084
Star RA (1998) Treatment of acute renal failure. Kidney Int 54:1817–31.27
Molina A, Ubeda M, Escribese MM, García-Bermejo L, Sancho D, Pérez de Lema G et al (2005) Renal ischemia/reperfusion injury: functional tissue preservation by anti-activated 1 integrin therapy. J Am Soc Nephrol 16:374–82.28
Visnagri A, Kandhare AD, Bodhankar SL (2015) Renoprotective effect of berberine via intonation on apoptosis and mitochondrial-dependent pathway in renal ischemia reperfusion-induced mutilation. Ren Fail 7:482–93.29
Kaya C, Karabulut R, Turkyilmaz Z, Sonmez K, Kulduk G, Gülbahar Ö, Köse F, Basaklar AC (2015) Lycopene has reduced renal damage histopathologically and biochemically in experimental renal ischemia–reperfusion injury. Ren Fail 37:1390–1395
Hoffmann D, Bijol V, Krishnamoorthy A, Gonzalez VR, Frendl G, Zhang Q et al (2012) Fibrinogen excretion in the urine and immunoreactivity in the kidney serves as a translational biomarker for acute kidney injury. AJP 181(3):818–828
Wang P, Zhu Q, Wu N, Siow YL, Aukema H, O K. (2013) Tyrosol attenuates ischemia-reperfusion-induced kidney injury via inhibition of induceble nitric oxide synthase. J Agric Food Chem 61:3669–3675
Mousavi G (2015) Study on the effect of black cumin (Nigella sativa Linn) on experimental renal ischemia-reperfusion injury in rats. Acta Cir Bras 30(18):542–550
Chen H, Xing B, Lui X, Zhan B, Zhou J, Zhu H et al (2008) Ozone oxidative preconditioning protects the rat kidney from reperfusion injury: the role of nitric oxide. J Surg Res 149:287–295
Guan Z, Gobè G, Willgoss D, Endre ZH (2006) Renal endothelial dysfunction and impaired autoregulation after ischemia-reperfusion injury result from excess nitric oxide. Am J Physiol Renal Physiol 291(3):F619–F628
Funding
This study was supported by the Eskişehir Osmangazi University Scientific Research Projects Commission (project no. 2018-2085).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The study was approved by the Committee of Local Ethics Committee of Eskişehir Osmangazi University Animal Experiments (ESOGU HADYEK) (decision no: 657 of 14.03.2018).
Conflict of Interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Kar, F., Hacioglu, C., Senturk, H. et al. The Role of Oxidative Stress, Renal Inflammation, and Apoptosis in Post Ischemic Reperfusion Injury of Kidney Tissue: the Protective Effect of Dose-Dependent Boric Acid Administration. Biol Trace Elem Res 195, 150–158 (2020). https://doi.org/10.1007/s12011-019-01824-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12011-019-01824-1