Abstract
Metabolic diseases or injuries damage bone structure and self-renewal capacity. Trace elements and hydroxyapatite crystals are important in the development of biomaterials to support the renewal of bone extracellular matrix. In this study, it was assumed that the boron-loaded nanometer-sized hydroxyapatite composite supports the construction of extracellular matrix by controlled boron release in order to prevent its toxic effect. In this context, boron release from nanometer-sized hydroxyapatite was calculated by ICP-MS as in large proportion within 1 h and continuing release was provided at a constant low dose. The effect of the boron-containing nanometer-sized hydroxyapatite composite on the proliferation of SaOS-2 osteoblasts and human bone marrow-derived mesenchymal stem cells was evaluated by WST-1 and compared with the effects of nano-hydroxyapatite and boric acid. Boron increased proliferation of mesenchymal stem cells at high doses and exhibited different effects on osteoblastic cell proliferation. Boron-containing nano-hydroxyapatite composites increased osteogenic differentiation of mesenchymal stem cells by increasing alkaline phosphatase activity, when compared to nano-hydroxyapatite composite and boric acid. The molecular mechanism of effective dose of boron-containing hydroxyapatite has been assessed by transcriptomic analysis and shown to affect genes involved in Wnt, TGF-β, and response to stress signaling pathways when compared to nano-hydroxyapatite composite and boric acid. Finally, a safe osteoconductive dose range of boron-containing nano-hydroxyapatite composites for local repair of bone injuries and the molecular effect profile in the effective dose should be determined by further studies to validation of the regenerative therapeutic effect window.





Similar content being viewed by others
References
Kankilic B, Kose S, Korkusuz P, Timucin M, Korkusuz F (2016) Mesenchymal stem cells and Nano-bioceramics for bone regeneration. Curr Stem Cell Res Ther 11(6):487–493
Kankilic B, Dede EC, Korkusuz P, Timuçin M, Korkusuz F (2017) Apatites for orthopedic applications. Clinical Applications of Biomaterials. Springer, In, pp 65–90
Pascaretti-Grizon F, Guillaume B, Terranova L, Arbez B, Libouban H, Chappard D (2017) Maxillary sinus lift with Beta-Tricalcium phosphate (beta-TCP) in edentulous patients: a Nanotomographic and Raman study. Calcif Tissue Int 101(3):280–290. https://doi.org/10.1007/s00223-017-0280-5
Ciftci E, Köse S, Korkusuz P, Timuçin M, Korkusuz F (2014) Boron containing Nano hydroxyapatites (Bn-HAp) stimulate mesenchymal stem cell adhesion, proliferation and differentiation. Key Eng Mater 631:373–378
Bi L, Rahaman MN, Day DE, Brown Z, Samujh C, Liu X, Mohammadkhah A, Dusevich V, Eick JD, Bonewald LF (2013) Effect of bioactive borate glass microstructure on bone regeneration, angiogenesis, and hydroxyapatite conversion in a rat calvarial defect model. Acta Biomater 9(8):8015–8026. https://doi.org/10.1016/j.actbio.2013.04.043
Korkusuz F, Timuçin M, Korkusuz P (2014) Nanocrystalline apatite-based biomaterials and stem cells in Orthopaedics. In: Ben-Nissan B (ed) Advances in calcium phosphate biomaterials. Springer, Berlin Heidelberg, Berlin, Heidelberg, pp 373–390. https://doi.org/10.1007/978-3-642-53980-0_12
Dessordi R, Spirlandeli AL, Zamarioli A, Volpon JB, Navarro AM (2017) Boron supplementation improves bone health of non-obese diabetic mice. J Trace Elem Med Biol 39:169–175. https://doi.org/10.1016/j.jtemb.2016.09.011
Cheng J, Peng K, Jin E, Zhang Y, Liu Y, Zhang N, Song H, Liu H, Tang Z (2011) Effect of additional boron on tibias of African ostrich chicks. Biol Trace Elem Res 144(1–3):538–549. https://doi.org/10.1007/s12011-011-9024-y
Hunt CD (2012) Dietary boron: progress in establishing essential roles in human physiology. J Trace Elem Med Biol 26(2–3):157–160. https://doi.org/10.1016/j.jtemb.2012.03.014
Kurtoğlu F, Kurtoğlu V, Celik I, Kececi T, Nizamlioğlu M (2005) Effects of dietary boron supplementation on some biochemical parameters, peripheral blood lymphocytes, splenic plasma cells and bone characteristics of broiler chicks given diets with adequate or inadequate cholecalciferol (vitamin D3) content. Br Poult Sci 46(1):87–96
Hakki SS, Malkoc S, Dundar N, Kayis SA, Hakki EE, Hamurcu M, Baspinar N, Basoglu A, Nielsen FH, Götz W (2015) Dietary boron does not affect tooth strength, micro-hardness, and density, but affects tooth mineral composition and alveolar bone mineral density in rabbits fed a high-energy diet. J Trace Elem Med Biol 29:208–215
Boyacioglu O, Orenay-Boyacioglu S, Yildirim H, Korkmaz M (2018) Boron intake, osteocalcin polymorphism and serum level in postmenopausal osteoporosis. J Trace Elem Med Biol 48:52–56. https://doi.org/10.1016/j.jtemb.2018.03.005
Calis M, Demirtas TT, Vatansever A, Irmak G, Sakarya AH, Atilla P, Ozgur F, Gumusderelioglu M (2017) A biomimetic alternative to synthetic hydroxyapatite: "boron-containing bone-like hydroxyapatite" precipitated from simulated body fluid. Ann Plast Surg 79(3):304–311. https://doi.org/10.1097/SAP.0000000000001072
Chen X, Zhao Y, Geng S, Miron RJ, Zhang Q, Wu C, Zhang Y (2015) In vivo experimental study on bone regeneration in critical bone defects using PIB nanogels/boron-containing mesoporous bioactive glass composite scaffold. Int J Nanomedicine 10:839
Dogan A, Demirci S, Bayir Y, Halici Z, Karakus E, Aydin A, Cadirci E, Albayrak A, Demirci E, Karaman A, Ayan AK, Gundogdu C, Sahin F (2014) Boron containing poly-(lactide-co-glycolide) (PLGA) scaffolds for bone tissue engineering. Mater Sci Eng C Mater Biol Appl 44:246–253. https://doi.org/10.1016/j.msec.2014.08.035
Gümüşderelioğlu M, Tunçay EÖ, Kaynak G, Demirtaş TT, Aydın ST, Hakkı SS (2015) Encapsulated boron as an osteoinductive agent for bone scaffolds. J Trace Elem Med Biol 31:120–128
Wu C, Miron R, Sculean A, Kaskel S, Doert T, Schulze R, Zhang Y (2011) Proliferation, differentiation and gene expression of osteoblasts in boron-containing associated with dexamethasone deliver from mesoporous bioactive glass scaffolds. Biomaterials 32(29):7068–7078. https://doi.org/10.1016/j.biomaterials.2011.06.009
Balasubramanian P, Grünewald A, Detsch R, Hupa L, Jokic B, Tallia F, Solanki AK, Jones JR, Boccaccini AR (2016) Ion release, hydroxyapatite conversion, and cytotoxicity of boron-containing bioactive glass scaffolds. Int J Appl Glas Sci 7(2):206–215
Li X, Wang X, Jiang X, Yamaguchi M, Ito A, Bando Y, Golberg D (2016) Boron nitride nanotube-enhanced osteogenic differentiation of mesenchymal stem cells. J Biomed Mater Res B Appl Biomater 104(2):323–329. https://doi.org/10.1002/jbm.b.33391
Lu X, Li K, Xie Y, Huang L, Zheng X (2016) Chemical stability and osteogenic activity of plasma-sprayed boron-modified calcium silicate-based coatings. J Mater Sci Mater Med 27(11):166. https://doi.org/10.1007/s10856-016-5781-7
Tuncay EO, Demirtas TT, Gumusderelioglu M (2017) Microwave-induced production of boron-doped HAp (B-HAp) and B-HAp coated composite scaffolds. J Trace Elem Med Biol 40:72–81. https://doi.org/10.1016/j.jtemb.2016.12.005
Arslan A, Cakmak S, Gumusderelioglu M (2018) Enhanced osteogenic activity with boron-doped nanohydroxyapatite-loaded poly (butylene adipate-co-terephthalate) fibrous 3D matrix. Artif Cells Nanomed Biotechnol:1–10. https://doi.org/10.1080/21691401.2018.1470522
Hakki SS, Bozkurt BS, Hakki EE (2010) Boron regulates mineralized tissue-associated proteins in osteoblasts (MC3T3-E1). J Trace Elem Med Biol 24(4):243–250. https://doi.org/10.1016/j.jtemb.2010.03.003
Ying X, Cheng S, Wang W, Lin Z, Chen Q, Zhang W, Kou D, Shen Y, Cheng X, Rompis FA, Peng L, Zhu Lu C (2011) Effect of boron on osteogenic differentiation of human bone marrow stromal cells. Biol Trace Elem Res 144(1–3):306–315. https://doi.org/10.1007/s12011-011-9094-x
Movahedi Najafabadi BA, Abnosi MH (2016) Boron induces early matrix mineralization via calcium deposition and elevation of alkaline phosphatase activity in differentiated rat bone marrow mesenchymal stem cells. Cell J 18(1):62–73
Yin C, Jia X, Miron RJ, Long Q, Xu H, Wei Y, Wu M, Zhang Y, Li Z (2018) Setd7 and its contribution to boron-induced bone regeneration in boron-mesoporous bioactive glass scaffolds. Acta Biomater 73:522–530. https://doi.org/10.1016/j.actbio.2018.04.033
Atila A, Halici Z, Cadirci E, Karakus E, Palabiyik SS, Ay N, Bakan F, Yilmaz SJMS, C E (2016) Study of the boron levels in serum after implantation of different ratios nano-hexagonal boron nitride–hydroxy apatite in rat femurs 58:1082–1089
Dominici M, Le Blanc K, Mueller I, Slaper-Cortenbach I, Marini F, Krause D, Deans R, Keating A, Prockop D, Horwitz E (2006) Minimal criteria for defining multipotent mesenchymal stromal cells. The International Society for Cellular Therapy position statement. Cytotherapy 8(4):315–317. https://doi.org/10.1080/14653240600855905
Goncu Y, Gecgin M, Bakan F, Ay N (2017) Electrophoretic deposition of hydroxyapatite-hexagonal boron nitride composite coatings on Ti substrate. Mater Sci Eng C Mater Biol Appl 79:343–353. https://doi.org/10.1016/j.msec.2017.05.023
Gautam C, Chakravarty D, Gautam A, Tiwary CS, Woellner CF, Mishra VK, Ahmad N, Ozden S, Jose S, Biradar S, Vajtai R, Trivedi R, Galvao DS, Ajayan PM (2018) Synthesis and 3D interconnected nanostructured h-BN-based biocomposites by low-temperature plasma sintering: bone regeneration applications. ACS Omega 3(6):6013–6021. https://doi.org/10.1021/acsomega.8b00707
Shuai C, Gao C, Feng P, Xiao T, Yu K, Deng Y, Peng S (2016) Boron nitride nanotubes reinforce Tricalcium phosphate scaffolds and promote the osteogenic differentiation of mesenchymal stem cells. J Biomed Nanotechnol 12(5):934–947
Nagarajan S, Belaid H, Pochat-Bohatier C, Teyssier C, Iatsunskyi I, Coy E, Balme S, Cornu D, Miele P, Kalkura NS, Cavailles V, Bechelany M (2017) Design of Boron Nitride/gelatin electrospun nanofibers for bone tissue engineering. ACS Appl Mater Interfaces 9(39):33695–33706. https://doi.org/10.1021/acsami.7b13199
Unal S, Ekren N, Sengil AZ, Oktar FN, Irmak S, Oral O, Sahin YM, Kilic O, Agathopoulos S, Gunduz O (2017) Synthesis, characterization, and biological properties of composites of hydroxyapatite and hexagonal boron nitride. J Biomed Mater Res B Appl Biomater 106:2384–2392. https://doi.org/10.1002/jbm.b.34046
Yang X, Li Y, He W, Huang Q, Zhang R, Feng Q (2018) Hydroxyapatite/collagen coating on PLGA electrospun fibers for osteogenic differentiation of bone marrow mesenchymal stem cells. 106 (11):2863–2870. https://doi.org/10.1002/jbm.a.36475
Cha M, Lee KM, Lee JHJTe, medicine r (2018) Positive Effects of Bisphosphonates on Osteogenic Differentiation in Patient-Derived Mesenchymal Stem Cells for the Treatment of Osteoporosis 15 (4):467–475
Takahara T, Arai Y, Kono Y, Shibata H, Maki M (2018) A microtubule-associated protein MAP1B binds to and regulates localization of a calcium-binding protein ALG-2. Biochem Biophys Res Commun 497(2):492–498
Bleicher F, Couble ML, Buchaille R, Farges JC, Magloire H (2001) New genes involved in odontoblast differentiation. Adv Dent Res 15:30–33. https://doi.org/10.1177/08959374010150010701
Deng X, Liang LN, Zhu D, Zheng LP, Yu JH, Meng XL, Zhao YN, Sun XX, Pan TW, Liu YQ (2018) Wedelolactone inhibits osteoclastogenesis but enhances osteoblastogenesis through altering different semaphorins production. Int Immunopharmacol 60:41–49. https://doi.org/10.1016/j.intimp.2018.04.037
Kang S, Kumanogoh A Semaphorins in bone development, homeostasis, and disease. In: Seminars in cell & developmental biology, 2013. vol 3. Elsevier, pp 163–171
Zhang Y, Wang N, Ma J, Chen XC, Li Z, Zhao W (2016) Expression profile analysis of new candidate genes for the therapy of primary osteoporosis. Eur Rev Med Pharmacol Sci 20(3):433–440
Zhang Y, Yang TL, Li X, Guo Y (2015) Functional analyses reveal the essential role of SOX6 and RUNX2 in the communication of chondrocyte and osteoblast. Osteoporos Int 26(2):553–561. https://doi.org/10.1007/s00198-014-2882-3
Lamora A, Talbot J, Mullard M, Brounais-Le Royer B, Redini F, Verrecchia F (2016) TGF-β signaling in bone remodeling and osteosarcoma progression. J Clin Med 5(11). https://doi.org/10.3390/jcm5110096
Yano M, Inoue Y, Tobimatsu T, Hendy G, Canaff L, Sugimoto T, Seino S, Kaji H (2012) Smad7 inhibits differentiation and mineralization of mouse osteoblastic cells. Endocr J 59(8):653–662
Kobylewski SE (2017) Activation of the EIF2α/ATF4 and ATF6 pathways in DU-145 cells by boric acid at the concentration reported in men at the US mean boron intake. Biol Trace Element Res 176(2):278–293
Polge C, Aniort J, Armani A, Claustre A, Coudy-Gandilhon C, Tournebize C, Deval C, Combaret L, Bechet D, Sandri M, Attaix D, Taillandier D (2018) Erratum: Polge, C., et al. UBE2E1 Is Preferentially Expressed in the Cytoplasm of Slow-Twitch Fibers and Protects Skeletal Muscles from Exacerbated Atrophy upon Dexamethasone Treatment. Cells 2018, 7, 214. Cells 7 (12). https://doi.org/10.3390/cells7120242
Kim JH, Mukherjee A, Madhavan SM, Konieczkowski M, Sedor JR (2012) WT1-interacting protein (Wtip) regulates podocyte phenotype by cell-cell and cell-matrix contact reorganization. Am J Physiol Renal Physiol 302(1):F103–F115. https://doi.org/10.1152/ajprenal.00419.2011
Henderson KA, Kobylewski SE, Yamada KE, Eckhert CD (2015) Boric acid induces cytoplasmic stress granule formation, eIF2α phosphorylation, and ATF4 in prostate DU-145 cells. Biometals 28(1):133–141
Czekanska EM, Stoddart MJ, Ralphs JR, Richards RG, Hayes JS (2014) A phenotypic comparison of osteoblast cell lines versus human primary osteoblasts for biomaterials testing. J Biomed Mater Res A 102(8):2636–2643. https://doi.org/10.1002/jbm.a.34937
Acknowledgments
Statistical analyses were done by Osman Dağ, MSc, research assistant at the Biostatistics Department Faculty of Medicine in Hacettepe University.
Funding
This study was supported by Hacettepe University Scientific Research Project Coordination Unit with project number TBB-2017-13-312.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interests
The authors declare that they have no conflict of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Sup. 1
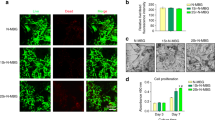
The mean-standard deviation graphics of the SaOS-2 cells and hBMCs proliferation by days. Optical density (OD) values of SaOS-2 cells proliferation a on day 1, b on day 3, c on day 5 (n = 14 for B-nHAp, nHAp composites and BA treated cells and n = 16 for untreated cells at each time point) and hBMSCs proliferation d on day 1, e on day 3 and f on day 5 are shown (n = 21 for B-nHAp, nHAp composites and BA treated cells and n = 24 for untreated cells at each time point). (a, b, c) p < 0.05, only the statistically significance of the relevant data is shown. (a) refers to the statistically significant difference comparing to untreated control, (b) and (c) refer to the statistically significant difference comparing to equivalent dose of nHAp and BA within the composite respectively. hBMSCs: Human bone marrow mesenchymal stem cells, B-nHAp: Boron containing nano-hydroxyapatite, nHAp: Nano-hydroxyapatite, BA: Boric acid. (PNG 336 kb)
Sup. 2
The mean-standard deviation graphics of the hBMSCs ALP activity by days. Optical density (OD) values of the ALP activity assay a on day 7, b on day 14 and c 21 are shown (n = 6 for all groups at each time point). (a, b, c) p < 0.05, Only the statistically significance of the relevant data is shown. (a) refers to the statistically significant difference comparing to untreated control, (b) and (c) refer to the statistically significant difference comparing to equivalent dose of nHAp and BA within the composite respectively. hBMSCs: Human bone marrow mesenchymal stem cells, ALP: Alkaline phosphatase, B-nHAp: Boron containing nano-hydroxyapatite, nHAp: Nano-hydroxyapatite, BA: Boric acid. (PNG 685 kb)
Rights and permissions
About this article
Cite this article
Gizer, M., Köse, S., Karaosmanoglu, B. et al. The Effect of Boron-Containing Nano-Hydroxyapatite on Bone Cells. Biol Trace Elem Res 193, 364–376 (2020). https://doi.org/10.1007/s12011-019-01710-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12011-019-01710-w