Abstract
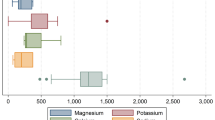
While general recommendations are for 300-mg magnesium intake a day, an advanced low-dose formulation of magnesium chloride, ChronoMag®, was designed to provide 100 mg of magnesium element, thus decreasing the risk of gastrointestinal side effects and allowing long-term supplementation in health conditions related to low magnesium levels. The present study aimed to compare magnesium release profile and bioavailability between this patented low-dose continuous-release magnesium chloride tablet (100 mg magnesium element) and a reference tablet at the usually prescribed dose (300 mg magnesium element). Magnesium release profile was determined by dissolving the tablets in solutions simulating the gastrointestinal tract environment. A randomized double-blind crossover controlled trial of ChronoMag® versus reference tablet (3 × 100 mg magnesium element tablets) in 12 normo-magnesemic healthy volunteers was conducted to evaluate the bioavailability of the patented magnesium chloride tablets (two 50 mg magnesium tablets, once-a-day intake). While the reference tablet released 100% of its magnesium within 1 h of dissolution, release from the magnesium chloride formulation was continuous for 6 h. Cumulative urinary magnesium levels compared to those with the reference tablet were 76% (0–5 h), 89% (0–10 h), and 87% (0–24 h). Elimination after 24 h was fairly similar with both supplements. Our results suggest that the new magnesium chloride formulation, providing continuous low-dose magnesium release throughout the gastrointestinal tract, improves absorption and bioavailability. This formulation conforms to the physiological mechanism of magnesium absorption throughout the digestive tract, allowing high absorption, and may improve gastrointestinal tolerance in long-term use.



Similar content being viewed by others
References
Saris NE, Mervaala E, Karppanen H et al (2000) Magnesium. An update on physiological, clinical and analytical aspects. Clin Chim Acta Int J Clin Chem 294:1–26
Gröber U, Schmidt J, Kisters K (2015) Magnesium in prevention and therapy. Nutrients 7:8199–8226. https://doi.org/10.3390/nu7095388
Rodríguez-Morán M, Simental Mendía LE, Zambrano Galván G, Guerrero-Romero F (2011) The role of magnesium in type 2 diabetes: a brief based-clinical review. Magnes Res Off Organ Int Soc Dev Res Magnes 24:156–162. https://doi.org/10.1684/mrh.2011.0299
Gobbo LCD, Imamura F, Wu JH, et al (2013) Circulating and dietary magnesium and risk of cardiovascular disease: a systematic review and meta-analysis of prospective studies. Am J Clin Nutr ajcn.053132. doi: https://doi.org/10.3945/ajcn.112.053132, 98, 160, 173
Sarrafzadegan N, Khosravi-Boroujeni H, Lotfizadeh M, Pourmogaddas A, Salehi-Abargouei A (2016) Magnesium status and the metabolic syndrome: a systematic review and meta-analysis. Nutr Burbank Los Angel Cty Calif 32:409–417. https://doi.org/10.1016/j.nut.2015.09.014
Rude RK, Gruber HE (2004) Magnesium deficiency and osteoporosis: animal and human observations. J Nutr Biochem 15:710–716. https://doi.org/10.1016/j.jnutbio.2004.08.001
Castiglioni S, Cazzaniga A, Albisetti W, Maier JAM (2013) Magnesium and osteoporosis: current state of knowledge and future research directions. Nutrients 5:3022–3033. https://doi.org/10.3390/nu5083022
Musso CG, Belloso WH, Glassock RJ (2016) Water, electrolytes, and acid-base alterations in human immunodeficiency virus infected patients. World J Nephrol 5:33–42. https://doi.org/10.5527/wjn.v5.i1.33
Musso CG (2009) Magnesium metabolism in health and disease. Int Urol Nephrol 41:357–362. https://doi.org/10.1007/s11255-009-9548-7
Del Gobbo LC, Elin RJ, Poirier P, Egeland GM (2012) Serum magnesium: a biomarker of cardiovascular risk revisited? Magnes Res 25:49–53. https://doi.org/10.1684/mrh.2012.0309
Del Gobbo LC, Song Y, Poirier P et al (2012) Low serum magnesium concentrations are associated with a high prevalence of premature ventricular complexes in obese adults with type 2 diabetes. Cardiovasc Diabetol 11:23. https://doi.org/10.1186/1475-2840-11-23
Simental-Mendía LE, Sahebkar A, Rodríguez-Morán M, Guerrero-Romero F (2016) A systematic review and meta-analysis of randomized controlled trials on the effects of magnesium supplementation on insulin sensitivity and glucose control. Pharmacol Res 111:272–282. https://doi.org/10.1016/j.phrs.2016.06.019
Verma H, Garg R (2017) Effect of magnesium supplementation on type 2 diabetes associated cardiovascular risk factors: a systematic review and meta-analysis. J Hum Nutr Diet n/a-n/a 30:621–633. https://doi.org/10.1111/jhn.12454
Joris PJ, Plat J, Bakker SJ, Mensink RP (2016) Long-term magnesium supplementation improves arterial stiffness in overweight and obese adults: results of a randomized, double-blind, placebo-controlled intervention trial. Am J Clin Nutr 103:1260–1266. https://doi.org/10.3945/ajcn.116.131466
Zhang X, Li Y, Del Gobbo LC et al (2016) Effects of magnesium supplementation on blood pressure: a meta-analysis of randomized double-blind placebo-controlled trials. Hypertens Dallas Tex 1979 68:324–333. https://doi.org/10.1161/HYPERTENSIONAHA.116.07664
Posadas-Sánchez R, Posadas-Romero C, Cardoso-Saldaña G, Vargas-Alarcón G, Villarreal-Molina MT, Pérez-Hernández N, Rodríguez-Pérez JM, Medina-Urrutia A, Jorge-Galarza E, Juárez-Rojas JG, Torres-Tamayo M (2016) Serum magnesium is inversely associated with coronary artery calcification in the genetics of atherosclerotic disease (GEA) study. Nutr J 15:22. https://doi.org/10.1186/s12937-016-0143-3
Aydin H, Deyneli O, Yavuz D et al (2010) Short-term oral magnesium supplementation suppresses bone turnover in postmenopausal osteoporotic women. Biol Trace Elem Res 133:136–143. https://doi.org/10.1007/s12011-009-8416-8
Sojka JE, Weaver CM (1995) Magnesium supplementation and osteoporosis. Nutr Rev 53:71–74
Veronese N, Stubbs B, Solmi M, Noale M, Vaona A, Demurtas J, Maggi S (2017) Dietary magnesium intake and fracture risk: data from a large prospective study. Br J Nutr 117:1–7. https://doi.org/10.1017/S0007114517001350
Abbasi B, Kimiagar M, Sadeghniiat K et al (2012) The effect of magnesium supplementation on primary insomnia in elderly: a double-blind placebo-controlled clinical trial. J Res Med Sci Off J Isfahan Univ Med Sci 17:1161
Killilea DW, Maier JAM (2008) A connection between magnesium deficiency and aging: new insights from cellular studies. Magnes Res 21:77–82
Veronese N, Zanforlini BM, Manzato E, Sergi G (2015) Magnesium and healthy aging. Magnes Res 28:112–115. https://doi.org/10.1684/mrh.2015.0387
Proctor ML, Murphy PA (2001) Herbal and dietary therapies for primary and secondary dysmenorrhoea. Cochrane Database Syst Rev CD002124. doi: https://doi.org/10.1002/14651858.CD002124
Fathizadeh N, Ebrahimi E, Valiani M, Tavakoli N, Yar MH (2010) Evaluating the effect of magnesium and magnesium plus vitamin B6 supplement on the severity of premenstrual syndrome. Iran J Nurs Midwifery Res 15:401–405
Ebrahimi E, Khayati Motlagh S, Nemati S, Tavakoli Z (2012) Effects of magnesium and vitamin b6 on the severity of premenstrual syndrome symptoms. J Caring Sci 1:183–189. https://doi.org/10.5681/jcs.2012.026
Stanisławska M, Szkup-Jabłońska M, Jurczak A, Wieder-Huszla S, Samochowiec A, Jasiewicz A, Noceń I, Augustyniuk K, Brodowska A, Karakiewicz B, Chlubek D, Grochans E (2014) The severity of depressive symptoms vs. serum Mg and Zn levels in postmenopausal women. Biol Trace Elem Res 157:30–35. https://doi.org/10.1007/s12011-013-9866-6
Park H, Parker GL, Boardman CH, Morris MM, Smith TJ (2011) A pilot phase II trial of magnesium supplements to reduce menopausal hot flashes in breast cancer patients. Support Care Cancer 19:859–863. https://doi.org/10.1007/s00520-011-1099-7
Fine KD, Santa Ana CA, Porter JL, Fordtran JS (1991) Intestinal absorption of magnesium from food and supplements. J Clin Invest 88:396–402. https://doi.org/10.1172/JCI115317
Institute of Medicine (1997) Food and nutrition board. Dietary reference intakes for calcium, phosphorus, magnesium, vitamin D, and fluoride. National Academy Press, Washington, DC
Graham LA, Caesar JJ, Burgen AS (1960) Gastrointestinal absorption and excretion of Mg 28 in man. Metabolism 9:646–659
Roth P, Werner E (1979) Intestinal absorption of magnesium in man. Int J Appl Radiat Isot 30:523–526
Coudray C, Rambeau M, Feillet-Coudray C et al (2005) Study of magnesium bioavailability from ten organic and inorganic Mg salts in Mg-depleted rats using a stable isotope approach. Magnes Res Off Organ Int Soc Dev Res Magnes 18:215–223
Firoz M, Graber M (2001) Bioavailability of US commercial magnesium preparations. Magnes Res Off Organ Int Soc Dev Res Magnes 14:257–262
Lindberg JS, Zobitz MM, Poindexter JR, Pak CY (1990) Magnesium bioavailability from magnesium citrate and magnesium oxide. J Am Coll Nutr 9:48–55
Bøhmer T, Røseth A, Holm H, Weberg-Teigen S, Wahl L (1990) Bioavailability of oral magnesium supplementation in female students evaluated from elimination of magnesium in 24-hour urine. Magnes Trace Elem 9:272–278
Joanny F (2011) Use of a matrix for orally administering sustained release magnesium, and composition containing said matrix
Dissolution test for solid dosage forms. In: Eur. Pharmacopoeia, 8.0. pp 288–295
Sabatier M, Arnaud MJ, Kastenmayer P, Rytz A, Barclay DV (2002) Meal effect on magnesium bioavailability from mineral water in healthy women. Am J Clin Nutr 75:65–71
Wilimzig C, Latz R, Vierling W, Mutschler E, Trnovec T, Nyulassy S (1996) Increase in magnesium plasma level after orally administered trimagnesium dicitrate. Eur J Clin Pharmacol 49:317–323
Kayne LH, Lee DB (1993) Intestinal magnesium absorption. Miner Electrolyte Metab 19:210–217
Pearnchob N, Siepmann J, Bodmeier R (2003) Pharmaceutical applications of shellac: moisture-protective and taste-masking coatings and extended-release matrix tablets. Drug Dev Ind Pharm 29:925–938. https://doi.org/10.1081/DDC-120024188
Sabatier M, Grandvuillemin A, Kastenmayer P, Aeschliman JM, Bouisset F, Arnaud MJ, Dumoulin G, Berthelot A (2011) Influence of the consumption pattern of magnesium from magnesium-rich mineral water on magnesium bioavailability. Br J Nutr 106:331–334. https://doi.org/10.1017/S0007114511001139
Acknowledgments
We would like to thank Pr. A. Berthelot, Dr. B. Shroot, Y. Marchenay, and Dr. Y. Juillet for their intellectual contribution to the study.
Funding
This study was sponsored by FJ LIFE SCIENCES.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The randomized, double-blind, crossover, controlled trial in healthy volunteers (registration number with the French health products safety agency, ANSM: 2013-001631-43; ClinicalTrials.gov trial number: NCT01935570) was conducted in the Clinical Pharmacology Center of the University Hospital of Clermont-Ferrand (France), in compliance with the principles of the Declaration of Helsinki and following Good Clinical Practices. Review board approval (CPP Sud-Est VI, Clermont-Ferrand, France) was obtained before the start of the study. Subjects were enrolled after giving written informed consent and fulfilling inclusion and exclusion criteria. Dr. Joanny declares she is the Founder and CEO of FJ LIFE SCIENCES.
Conflict of Interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Dualé, C., Cardot, JM., Joanny, F. et al. An Advanced Formulation of a Magnesium Dietary Supplement Adapted for a Long-Term Use Supplementation Improves Magnesium Bioavailability: In Vitro and Clinical Comparative Studies. Biol Trace Elem Res 186, 1–8 (2018). https://doi.org/10.1007/s12011-018-1277-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12011-018-1277-2