Abstract
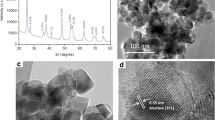
Nanoparticles have been widely used in various fields due to the superior physicochemical properties and functions. As a result, human exposure to nanoparticles increases dramatically. Previous researches have shown that nanoparticles could travel through the respiratory, digestive system, or skin into the blood and then to the secondary organs such as the brain, heart, and liver. Besides, the nanoparticle toxicity is controversial and dependent on the sensitivity of the cell type, route of exposure, and condition, as well as their characteristics. Similarly, cobalt ferrite nanoparticles (CoFe2O4-NPs) have been used in different industrial fields, and have also various application possibilities in medical and biomedical fields. CoFe2O4-NPs induce toxic responses in various organisms such as human, mice, and algae. However, there is a serious deficit of information concerning their effects on human health and the environment. We aimed to investigate the toxic effects of CoFe2O4-NPs on liver (HepG2), colon (Caco-2), lung (A549), and neuron (SH-SY5Y) cells, which reflect different exposure routes in vitro, by using various toxicological endpoints. The cytotoxicity, genotoxicity, oxidative damage, and apoptosis induction of CoFe2O4-NPs (39 ± 17 nm) were evaluated. After 24 h, the nanoparticles decreased cell viability at ≤100 μg/mL, while increasing viability at >100 μg/mL. CoFe2O4-NPs induced DNA and oxidative damage with increased malondialdehyde (MDA) and 8-hydroxy deoxyguanosine (8-OHdG) levels and decreased glutathione (GSH) levels with no change in protein carbonyl (PC) levels. CoFe2O4-NPs had apoptotic effect in HepG2 and Caco-2 cells in a concentration-dependent manner and necrotic effects on SH-SY5Y and A549 cells. Consequently, the adverse effects of CoFe2O4-NPs should raise concern about their safety in consumer products.




Similar content being viewed by others
Abbreviations
- 8-OHdG:
-
8-Hydroxy deoxyguanosine
- ATCC:
-
American type culture collection
- AV:
-
Annexin V–FITC
- CoFe2O4-NPs:
-
Cobalt ferrite nanoparticles
- DMEM:
-
Dulbecco’s modified eagle medium
- DNA:
-
Deoxyribonucleic acid
- EDTA:
-
Ethylene diamine tetraacetic acid
- ELISA:
-
Enzyme-linked immune sorbent assay
- EMEM:
-
Eagle’s minimum essential medium
- FBS:
-
Fetal bovine serum
- GSH:
-
Glutathione
- H2O2 :
-
Hydrogen peroxide
- IARC:
-
International Agency for Research on Cancer
- ICP-MS:
-
Inductively coupled plasma-mass spectrometry
- MDA:
-
Malondialdehyde
- MTT:
-
3-[4,5-dimethylthiazol-2-yl]-2,5-diphenyl-tetrazolium bromide
- NRU:
-
Neutral red uptake
- OD:
-
Optical density
- PBS:
-
Phosphate buffered saline
- PC:
-
Protein carbonyl
- PI:
-
Propidium iodide
- ROS:
-
Reactive oxygen species
- SD:
-
Standard deviation
- TEM:
-
Transmission electron microscopy
References
Dhawan A, Sharma V (2010) Toxicity assessment of nanomaterials: methods and challenges. Anal Bioanal Chem 398:589–605
Kim YJ, Yu M, Park HO, Yang SI (2010) Comparative study of cytotoxicity, oxidative stress and genotoxicity induced by silica nanomaterials in human neuronal cell line. Mol Cell Toxicol 6:337–344
Arora S, Rajwade JM, Paknikar KM (2012) Nanotoxicology and in vitro studies: the need of the hour. Toxicol Appl Pharmacol 258:151–165
Brooking J, Davis SS, Illum L (2001) Transport of nanoparticles across the rat nasal mucosa. J Drug Target 9:267–279
Alarifi S, Ali D, Verma A, Alakhtani S, et al. (2013) Cytotoxicity and genotoxicity of copper oxide nanoparticles in human skin keratinocytes cells. Int J Toxicol 32:296–307
Alinovi R, Goldoni M, Pinelli S, Campanini M, et al. (2015) Oxidative and pro-inflammatory effects of cobalt and titanium oxide nanoparticles on aortic and venous endothelial cells. Toxicol in Vitro 29:426–437
Maaz K, Mumtaz A, Hasanain SK, Ceylan A (2007) Synthesis and magnetic properties of cobalt ferrite (CoFe2O4) nanoparticles prepared by wet chemical route. J Magn Magn Mater 308:289–295
Di Guglielmo C, López DR, De Lapuente J, Mallafre JM, et al. (2010) Embryotoxicity of cobalt ferrite and gold nanoparticles: a first in vitro approach. Reprod Toxicol 30:271–276
Amiri S, Shokrollahi H (2013) The role of cobalt ferrite magnetic nanoparticles in medical science. Mater Sci Eng C 33:1–8
Ahmad F, Yao H, Ying Z, Xiaoyi L (2015) Toxicity of cobalt ferrite (CoFe2O4) nanobeads in Chlorella vulgaris: interaction, adaptation and oxidative stress. Chemosphere 139:479–485
Gianoncelli A, Marmorato P, Ponti J, Pascolo L, et al. (2013) Interaction of magnetic nanoparticles with 87MG cells studied by synchrotron radiation X-ray fluorescence techniques. X-Ray Spectrom 42:316–320
Matsuda S, Nakanishi T, Kaneko K, Osaka T (2015) Synthesis of cobalt ferrite nanoparticles using spermine and their effect on death in human breast cancer cells under an alternating magnetic field. Electrochim Acta 183:153–159
Horev-Azaria L, Baldi G, Beno D, Bonacchi D, et al. (2013) Predictive toxicology of cobalt ferrite nanoparticles: comparative in-vitro study of different cellular models using methods of knowledge discovery from data. Part Fibre Toxicol:10–32
Hwang DW, Lee DS, Kim S (2012) Gene expression profiles for genotoxic effects of silica-free and silica-coated cobalt ferrite nanoparticles. J Nucl Med 53(1):106–112
Chattopadhyay S, Dash SK, Tripathy S, Das B, et al. (2015) Toxicity of cobalt oxide nanoparticles to normal cells: an in vitro and in vivo study. Chem Biol Interact 226:58–71
Repetto G, del Peso A, Zurita JL (2008) Neutral red uptake assay for the estimation of cell viability/cytotoxicity. Nat Protoc 3:1125–1131
Van Meerloo J, Kaspers GJ, Cloos J (2011) Cell sensitivity assays: the MTT assay. Methods Mol Biol 731:237–245
Collins AR (2004) The comet assay for DNA damage and repair principles, applications, and limitations. Mol Biotechnol 26:249–261
Speit G, Hartmann A (1999) The comet assay (single-cell gel test): a sensitive genotoxicity test for the detection of DNA damage and repair. DNA Repair Protocols 113:203–212
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 7:248–254
Ahmad F, Liu X, Zhou Y, Yao H (2015) An in vivo evaluation of acute toxicity of cobalt ferrite (CoFe2O4) nanoparticles in larval-embryo zebrafish (Danio rerio). Aquat Toxicol 166:21–28
Mahmoudi M, Hofmann H, Rutishauser BR, Fink AP (2012) Assessing the in vitro and in vivo toxicity of superparamagnetic iron oxide nanoparticles. Chem Rev 112:2323–2338
Marmorato P, Ceccone G, Gianoncelli A, Pascolo L, et al. (2011) Cellular distribution and degradation of cobalt ferrite nanoparticles in Balb/3 T3 mouse fibroblasts. Toxicol Lett 207:128–136
Mariani V, Ponti J, Giudetti G, Broggi F, et al. (2012) Online monitoring of cell metabolism to assess the toxicity of nanoparticles: the case of cobalt ferrite. Nanotoxicology 6(3):272–287
Pershina AG, Sazonov AE, Novikov DV, et al. (2011) Study of DNA interaction with cobalt ferrite nanoparticles. J Nanosci Nanotechnol 11:2673–2677
Ahmad F, Liu X, Zhou Y, Yao H, et al. (2015) Assessment of thyroid endocrine system impairment and oxidative stress mediated by cobalt ferrite (CoFe2O4) nanoparticles in zebrafish larvae. Environ Toxicol. doi:10.1002/tox.
Kapilevich LV, D’yakova EY, Nosarev AV, Zaitseva TN, et al. (2010) Effect of nanodisperse ferrite cobalt (CoFe2O4) particles on contractile reactions in Guinea pigs airways. Bull Exp Biol Med 149(1):70–72
Acknowledgment
This work was supported by the Research Fund of Istanbul University (Project No: 40441).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Abudayyak, M., Altincekic Gurkaynak, T. & Özhan, G. In Vitro Toxicological Assessment of Cobalt Ferrite Nanoparticles in Several Mammalian Cell Types. Biol Trace Elem Res 175, 458–465 (2017). https://doi.org/10.1007/s12011-016-0803-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12011-016-0803-3