Abstract
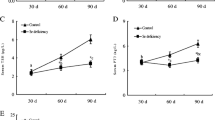
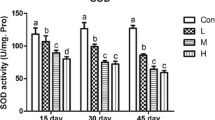
This study assessed the effects of different selenium (Se) supplementation levels on oxidative stress, cytokines, and immunotoxicity in chicken thymus. A total of 180 laying hens (1 day old; Mianyang, China) were randomly divided into 4 groups (n = 45). The chickens were maintained either on a basic diet (control group) containing 0.2 mg/kg Se, a low-supplemented diet containing 5 mg/kg Se, a medium-supplemented diet containing 10 mg/kg Se, or a high-supplemented diet containing 15 mg/kg Se for 15, 30, and 45 days, respectively. Over the entire experimental period, serum and thymus samples were collected and used for the detection of the experimental index. The results indicated that the antioxidative enzyme activities and messenger RNA (mRNA) levels of antioxidative enzymes, IFN-γ and IL-2 in the thymus, and the content of IFN-γ and IL-2 in the serum of excessive-Se-treated chickens at all time points (except for the 5 mg/kg Se supplement group at 15 days) were significantly decreased (P < 0.05) compared to the corresponding control groups. Interestingly, a significantly increase (P < 0.05) in the content of IFN-γ was observed in the serum and thymus in the 5 mg/kg Se supplement group at 15 and 30 days compared to the corresponding control groups. In histopathological examination, the thymus tissue from excessive-Se-treated chickens revealed different degrees of cortex drop, incrassation of the medulla, and degeneration of the reticular cells. These results suggested that the excessive Se could result in a decrease in immunity, an increase in oxidative damage, and a series of clinical pathology changes, such as cortex drop, incrassation of the medulla, and degeneration of the reticular cells.



Similar content being viewed by others
References
Shan H, Yan R, Diao J, Lin L, Wang S, Zhang M et al (2015) Involvement of caspases and their upstream regulators in myocardial apoptosis in a rat model of selenium deficiency-induced dilated cardiomyopathy. J Trace Elem Med Biol 31:85–91
Qin H, Zhu J, Liang L, Wang M, Su H (2013) The bioavailability of selenium and risk assessment for human selenium poisoning in high-Se areas, China. Environ Int 52:66–74
Combs GF Jr, Clark LC, Turnbull BW (2001) An analysis of cancer prevention by selenium. Biofactors 14:153–159
Li JL, Gao R, Li S, Wang JT et al (2010) Testicular toxicity induced by dietary cadmium in cocks and ameliorative effect by selenium. Biometals 23:695–705
Schweizer U, Schomburg L, Savaskan NE (2004) The neurobiology of selenium: lessons from transgenic mice. J Nutr 134:707–710
Martin-Romero FJ, Kryukov GV, Lobanov AV et al (2001) Selenium metabolism in Drosophila: selenoproteins, selenoprotein mRNA expression, fertility, and mortality. J Biol Chem 276:29798–29804
Rayman MP (2000) The importance of selenium to human health. Lancet 356:233–241
Hoffmann PR, Berry MJ (2008) The influence of selenium on immune responses. Mol Nutr Food Res 52:1273–1280
Chariot P, Bignani O (2003) Skeletal muscle disorders associated with selenium deficiency in humans. Muscle Nerve 27:662–668
Kaur P, Bansal MP (2005) Effect of selenium-induced oxidative stress on the cell kinetics in testis and reproductive ability of male mice. Nutrition 21:351–357
Brown KM, Arthur JR (2001) Selenium, selenoproteins and human health: a review. Public Health Nutr 4:593–599
Mahmoud KZ, Edens FW (2005) Influence of organic selenium on hsp70 response of heat-stressed and enteropathogenic Escherichia coli-challenged broiler chickens (Gallus gallus). Comp Biochem Physiol Part C Toxicol Pharmacol 141:69–75
Behne D, Kyriakopoulos A (2001) Mammalian seleniumcontaining proteins. Annu Rev Nutr 21:453–473
Clark LC, Combs GF Jr, Turnbull BW et al (1996) Effects of selenium supplementation for cancer prevention in patients with carcinoma of the skin. A randomized controlled trial. Nutritional Prevention of Cancer Study Group. JAMA 276:1957–1963
Rederstorff M, Krol A, Lescure A (2006) Understanding the importance of selenium and selenoproteins in muscle function. Cell Mol Life Sci 63:52–59
Zhang H, Feng X, Chan H−M, Larssen T (2014) New insights into traditional health risk assess-ments of mercury exposure: implications of selenium. Environ Sci Technol 48:1206–1212
Bajaj M, Eiche E, Neumann T, Winter J, Gallert C (2011) Hazardous concentrations of selenium in soil and groundwater in North-West India. J Hazard Mater 189:640–646
Lemire M, Philibert A, Fillion M, Passos CJ, Guimarães JR, Barbosa F Jr et al (2012) No evidence of selenosis from a selenium-rich diet in the Brazilian Amazon. Environ Int 40:128–136
Stadtman TC (2000) Selenium biochemistry. Mammalian selenoenzymes. Ann NY Acad Sci 899:399–402
Driscoll DM, Copeland PR (2003) Mechanism and regulation of selenoprotein synthesis. Annu Rev Nutr 23:17–40
Xu SW, Yao HD, Zhang J, Zhang ZW, Wang JT, Zhang JL, Jiang ZH (2013) The oxidative damage and disbalance of calcium homeostasis in brain of chicken induced by selenium deficiency. Biol Trace Elem Res 151:225–233
Ghazi Harsini S, Habibiyan M, Moeini MM, Abdolmohammadi AR (2012) Effects of dietary selenium, vitamin E, and their combination on growth, serum metabolites, and antioxidant defense system in skeletal muscle of broilers under heat stress. Biol Trace Elem Res 148:322–330
Rotruck JT, Pope AL, Ganther HE, Swanson AB, Hafeman DG, Hoekstra WG (1973) Selenium: biochemical role as a component of glutathione peroxi-dase. Science 179:588–590
Abbas HHH, Authman MMN (2009) Effects of accumulated selenium on some physiological parameters and oxidative stress indicators in tilapia fish (Oreochromis spp.). Am Eurasian J Agric Environ Sci 5:219–225
Miller LL, Wang F, Palace VP, Hontela A (2007) Effects of acute and subchronic exposures to waterborne selenite on the physiological stress response and oxidative stress indicators in juvenile rainbow trout. Aquat Toxicol 83:263–271
Xing H, Li S, Wang Z, Gao X, Xu S, Wang X (2012) Histopathological changes and antioxidant response in brain and kidney of common carp exposed to atrazine and chlorpyrifos. Chemosphere 88:377–383
Wang X, Xing H, Jiang Y, Wu H, Sun G, Xu Q, Xu S (2013) Accumulation, histopathological effects and response of biochemical markers in the spleens and head kidneys of common carp exposed to atrazine and chlorpyrifos. Food Chem Toxicol 62:148–158
Liu X, Li Z, Tie F, Liu N, Zhang Z, Xu S (2013) Effects of manganese-toxicity on immune-related organs of cocks. Chemosphere 90:2085–2100
Chen K, Shu G, Peng X, Fang J, Cui H, Chen J et al (2013) Protective role of sodium selenite on histopathological lesions, decreased T-cell subsets and increased apoptosis of thymus in broilers intoxicated with aflatoxin B1. Food Chem Toxicol 59:446–454
Wang D, Li X, Xu L, Hu Y, Zhang B, Liu J (2006) Immunologic synergism with IL-2 and effects of cCHMIs on mRNA expression of IL-2 and IFN-γ in chicken peripheral T lymphocyte. Vaccine 24:7109–7114
Shah MAA, Song X, Xu L, Yan R, Li X (2011) Construction of DNA vaccines encoding Eimeria acervulina cSZ-2 with chicken IL-2 and IFN-c and their efficacy against poultry coccidiosis. Res Vet Sci 90:72–77
Ren Z, Wang Y, Deng H, Deng Y, Deng J, Zuo Z et al (2015) Deoxynivalenol-induced cytokines and related genes in concanavalin A-stimulated primary chicken splenic lymphocytes. Toxicol in Vitro 29(2015):558–563
Chen K, Yuan S, Chen J, Peng X, Wang F, Cui H, Fang J (2013) Effects of sodium selenite on the decreased percentage of T cell subsets, contents of serum IL-2 and IFN-cinduced by aflatoxin B1in broilers. Res Vet Sci 95:143–145
Esterbauer H, Cheeseman KH (1990) Determination of aldehydic lipid peroxidation products: malonaldehyde and 4-hydroxynonenal. Methods Enzymol 186:407–421
Yao H, Liu W, Zhao W, Fan R, Zhao X, Khoso PA, Zhang Z, Xu S (2014) Different responses of selenoproteins to the altered expression of selenoprotein W in chicken myoblasts. RSC Adv 4:64032–64042
Yao H, Wu Q, Zhang Z, Li S, Wang X, Lei X, Xu S (2013) Selenoprotein W serves as an antioxidant in chicken myoblasts. Biochim Biophys Acta 1830:3112–3120
Yao HD, Wu Q, Zhang ZW, Zhang JL, Li S, Huang JQ, Ren FZ, Xu SW, Wang XL, Lei XG (2013) Gene expression of endoplasmic reticulum resident selenoproteins correlates with apoptosis in various muscles of Se-deficient chicks1–3. J Nutr 143:613–619
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254
Xing H, Li S, Wang X, Gao X, Xu S, Wang X (2013) Effects of atrazine and chlorpyrifos on the mRNA levels of HSP70 and HSC70 in the liver, brain, kidney and gill of common carp (Cyprinus carpio L.). Chemosphere 90:910–916
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2(−Delta Delta C(T)) method. Methods 25:402–408
Sun M, Liu G, Wu Q (2013) Speciation of organic and inorganic selenium in selenium-enriched rice by graphite furnace atomic absorption spectrometry after cloud point extraction. Food Chem 141:66–71
Tuzen M, Pekiner OZ (2015) Ultrasound-assisted ionic liquid dispersive liquid–liquid microextraction combined with graphite furnace atomic absorption spectrometric for selenium speciation in foods and beverages. Food Chem 188:619–624
Yurekli VA, Naziroglu M (2013) Selenium and topiramate attenuates blood oxidative toxicity in patients with epilepsy: a clinical pilot study. Biol Trace Elem Res 152:180–186
Lemly AD (2014) Teratogenic effects and monetary cost of selenium poisoning of fish in Lake Sutton, North Carolina. Ecotoxicol Environ Saf 104:160–167
Zeng J, Zhou J, Huang K (2009) Effect of selenium on pancreatic proinflammatory cytokines in streptozotocin-induced diabetic mice. J Nutr Biochem 20:530–536
Liu J, Zhao H, Liu Y, Liu Y, Wang X (2012) Effect of two selenium sources on hepatocarcinogenesis and several angiogenic cytokines in diethylnitrosamine-induced hepatocarcinoma rats. J Trace Elem Med Biol 26:255–261
Wu R, Lian X, Tang X, Fu Y, He C (2007) Study on the effects of cytokine level in ducklings with selenium poisoning. Agric Sci China 6:375–380
Hauser-Davis RA, Silva JAN, Rocha RCC, Pierre TS, Ziollib RL, Arruda MAZ (2016) Acute selenium selenite exposure effects on oxidative stress biomarkers and essential metals and trace-elements in the modelorganism zebrafish (Danio rerio). J Trace Elem Med Biol 33:68–72
Acknowledgments
Southwest University of Science and Technology Project (No. 15zx7121) and Mianyang Science and Technology Project (No. 14 N043) supported this study.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of Interest
None of the authors have any financial or personal relationships that could inappropriately influence or bias the content of the paper.
Additional information
Yachao Wang and Li Jiang contributed equally to this work.
Rights and permissions
About this article
Cite this article
Wang, Y., Jiang, L., Li, Y. et al. Effect of Different Selenium Supplementation Levels on Oxidative Stress, Cytokines, and Immunotoxicity in Chicken Thymus. Biol Trace Elem Res 172, 488–495 (2016). https://doi.org/10.1007/s12011-015-0598-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12011-015-0598-7