Abstract
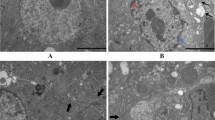
Excessive selenium (Se) intake is harmful for animals and humans. The aim of the present study was to examine the effect of long-term excessive Se supplementation on oxidative stress and endoplasmic reticulum (ER) stress-related injuries in chicken spleen. A total of 180 1-day-old chickens were randomly divided into four groups with different Se dietary contents (0.2 mg/kg Se, 5 mg/kg Se, 10 mg/kg Se, or 15 mg/kg Se) for 45 days. Then, the levels of antioxidative enzymes, GPx, SOD, and MDA as well as the expression levels of GRP78, ARF6, caspase 3, caspase 12, and Bcl 2 in the spleen were determined at days 15, 30, and 45, respectively. The results showed that excessive Se treatment decreased the activities of GPx and SOD (P < 0.05) but increased the levels of MDA (P < 0.05) in a dose- and time-dependent manner. In addition, the ER stress genes GRP78 and ATF6 were highly expressed (P < 0.05), and the apoptosis genes caspase 3 and caspase 12 were increased, but Bcl 2 was decreased by Se treatment (P < 0.05). Correlation analysis showed that there was a high correlation between these biomarkers, which indicated that ER stress and ER stress-related apoptosis were correlated with oxidative stress. These results showed the important role of oxidative stress and ER stress in Se-related immune injuries in chicken.




Similar content being viewed by others
References
Yao HD, Wu Q, Zhang ZW et al (2013) Gene expression of endoplasmic reticulum resident selenoproteins correlates with apoptosis in various muscles of se-deficient chicks. J Nutr 143:613–619
Yao H, Zhao W, Zhao X et al (2014) Selenium deficiency mainly influences the gene expressions of antioxidative selenoproteins in chicken muscles. Biol Trace Elem Res 161:318–327
Yao L, Du Q, Yao H et al. (2015) Roles of oxidative stress and endoplasmic reticulum stress in selenium deficiency-induced apoptosis in chicken liver. Biometals
Yu D, Li JL, Zhang JL et al (2011) Effects of dietary selenium on selenoprotein W gene expression in the chicken immune organs. Biol Trace Elem Res 144:678–687
Zwolak I, Zaporowska H (2012) Selenium interactions and toxicity: a review. Selenium interactions and toxicity. Cell Biol Toxicol 28:31–46
Zeng MS, Li X, Liu Y et al (2012) A high-selenium diet induces insulin resistance in gestating rats and their offspring. Free Radic Biol Med 52:1335–1342
Ayaz M, Ozdemir S, Yaras N et al (2005) Selenium-induced alterations in ionic currents of rat cardiomyocytes. Biochem Biophys Res Commun 327:163–173
Yao HD, Wu Q, Zhang ZW et al (2013) Selenoprotein W serves as an antioxidant in chicken myoblasts. Biochim Biophys Acta 1830:3112–3120
Yao HD, Liu W, Zhao WC et al (2014) Different responses of selenoproteins to the altered expression of selenoprotein W in chicken myoblasts. RSC Adv 4:64032
Huang JQ, Ren FZ, Jiang YY et al (2015) Selenoproteins protect against avian nutritional muscular dystrophy by metabolizing peroxides and regulating redox/apoptotic signaling. Free Radic Biol Med 83:129–138
Wu Q, Yao HD, Tan SR et al (2014) Possible correlation of selenoprotein w with inflammation factors in chicken skeletal muscles. Biol Trace Elem Res 161:167–172
Jiang ZH, Khoso PA, Yao HD et al (2015) SelW regulates inflammation-related cytokines in response to H2O2 in Se-deficient chicken liver. RSC Adv 5:37896–37905
Kitamura M, Hiramatsu N (2010) The oxidative stress: endoplasmic reticulum stress axis in cadmium toxicity. Biometals 23:941–950
Doyle KM, Kennedy D, Gorman AM et al (2011) Unfolded proteins and endoplasmic reticulum stress in neurodegenerative disorders. J Cell Mol Med 15:2025–2039
Srivastava RK, Li C, Chaudhary SC et al (2013) Unfolded protein response (UPR) signaling regulates arsenic trioxide-mediated macrophage innate immune function disruption. Toxicol Appl Pharmacol 272:879–887
Sano R, Reed JC (2013) ER stress-induced cell death mechanisms. Biochim Biophys Acta 1833:3460–3470
Liu L, Yang B, Cheng Y et al (2015) Ameliorative effects of selenium on cadmium-induced oxidative stress and endoplasmic reticulum stress in the chicken kidney. Biol Trace Elem Res 167:308–319
Zhao W, Liu W, Chen X et al (2014) Four endoplasmic reticulum resident selenoproteins may be related to the protection of selenium against cadmium toxicity in chicken lymphocytes. Biol Trace Elem Res 161:328–333
Yu J, Yao H, Gao X et al (2015) The role of nitric oxide and oxidative stress in intestinal damage induced by selenium deficiency in chickens. Biol Trace Elem Res 163:144–153
Esterbauer H, Cheeseman KH (1990) Determination of aldehydic lipid peroxidation products: malonaldehyde and 4-hydroxynonenal. Methods Enzymol 186:407–421
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254
Pfaffl MW (2001) A new mathematical model for relative quantification in real-time RT-PCR. Nucleic Acids Res 29, e45
Shao JJ, Yao HD, Zhang ZW et al (2012) The disruption of mitochondrial metabolism and ion homeostasis in chicken hearts exposed to manganese. Toxicol Lett 214:99–108
Zwolak I (2015) Increased cytotoxicity of vanadium to CHO-K1 cells in the presence of inorganic selenium. Bull Environ Contam Toxicol
Huang JQ, Li DL, Zhao H et al (2011) The selenium deficiency disease exudative diathesis in chicks is associated with downregulation of seven common selenoprotein genes in liver and muscle. J Nutr 141:1605–1610
Khoso PA, Yang Z, Liu C et al. (2015) Selenium deficiency downregulates selenoproteins and suppresses immune function in chicken thymus. Biol Trace Elem Res
Harisa GI, Abo-Salem OM, el El-sayed SM et al (2013) Effects of nutritional and excessive levels of selenium on red blood cells of rats fed a high cholesterol diet. Biol Trace Elem Res 152:41–49
Tsuji PA, Carlson BA, Anderson CB et al (2015) Dietary selenium levels affect selenoprotein expression and support the interferon-gamma and IL-6 immune response pathways in mice. Nutr 7:6529–6549
Li F, Hayashi T, Jin G et al (2005) The protective effect of dantrolene on ischemic neuronal cell death is associated with reduced expression of endoplasmic reticulum stress markers. Brain Res 1048:59–68
Kessel D, Castelli M, Reiners JJ (2005) Ruthenium red-mediated suppression of Bcl-2 loss and Ca(2+) release initiated by photodamage to the endoplasmic reticulum: scavenging of reactive oxygen species. Cell Death Differ 12:502–511
Masuda M, Miyazaki-Anzai S, Levi M et al (2013) PERK-eIF2alpha-ATF4-CHOP signaling contributes to TNFalpha-induced vascular calcification. J Am Heart Assoc 2, e000238
Wang SQ, Niu XL, Liu ZW et al (2013) Selenium deficiency is associated with endoplasmic reticulum stress in a rat model of cardiac malfunction. Biol Trace Elem Res 156:196–201
Csordas G, Hajnoczky G (2009) SR/ER-mitochondrial local communication: calcium and ROS. Biochim Biophys Acta 1787:1352–1362
Robert F. Feissner, Jolanta Skalska1, Gaum. WE et al. (2009) Crosstalk signaling between mitochondrial Ca2+ and ROS. Front Biosci 14: 1197–1218
Acknowledgments
The present work was financially supported by Southwest University of Science and Technology (15zx7121) and Mianyang Science and Technology Project (14 N043). The authors thank the Elsevier English Language Editing System to correct grammatical, spelling, and other common errors.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of Interest
The authors declare that they have no competing interests.
Additional information
Yachao Wang and Li Jiang should be considered as co-first authors
Yachao Wang and Li Jiang contributed equally to this work.
Rights and permissions
About this article
Cite this article
Wang, Y., Jiang, L., Li, Y. et al. Excessive Selenium Supplementation Induced Oxidative Stress and Endoplasmic Reticulum Stress in Chicken Spleen. Biol Trace Elem Res 172, 481–487 (2016). https://doi.org/10.1007/s12011-015-0596-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12011-015-0596-9