Abstract
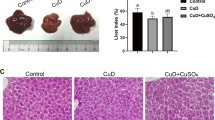
The aim of this study was to explore the effects of selenium addition on hepatic tissue ultrastructure and mitochondrial antioxidant capacity in copper-overloaded rats. Eighteen adult male Sprague-Dawley (SD) rats were randomly divided into three groups (n = 6 per group). Each group received 1 mL (intragastrically) of water (control, group I) or water containing copper chloride (CuCl2, 1 mol/L) (group II) or a mixture of CuCl2 (1 mol/L) with sodium selenite (Na2SeO3, 0.05 mol/L) (group III) once daily for 30 days. Histological examination revealed normal hepatocyte structure and no ultrastructural changes in mitochondria in controls. In contrast, group II exhibited severe ultrastructural alterations, fuzzy mitochondrial membranes, irregularly shaped and fragmented rough endoplasmic reticulum (RER), and the high melanin content; group III also exhibited larger amounts of engulfing vesicles (EV) in the cytoplasm. Compared to controls, the CuCl2 treatment lowered (P < 0.05) hepatocyte enzymatic activities and increased lipid peroxidation as indicated by an increase in malondialdehyde (MDA) levels. The effects of CuCl2 were attenuated by simultaneous administration with Na2SeO3. These results indicated that the adverse effects of copper toxicity can be partially attenuated by providing a source of selenium.

Similar content being viewed by others
References
Madsen E, Gitlin JD (2007) Copper and iron disorders of the brain. Annu Rev Neurosci 30:317–337
Gines S, Ivanova E, Seong IS, Saura CA, MacDonald ME (2003) Enhanced Akt signaling is an early pro-survival response that reflects N-methyl-D-aspartate receptor activation in Huntington’s disease knock-in striatal cells. J Biol Chem 278:50514–50522
Uriu-Adams JY, Keen CL (2005) Copper, oxidative stress, and human health. Mol Asp Med 26:268–298
Fernandes A, Mira ML, Azevedo MS, Manso C (1988) Mechanisms of hemolysis induced by copper. Free Radic Res Commun 4:291–298
Sardana MK, Sassa S, Kappas A (1982) Metal ion-mediated regulation of heme oxygenase induction in cultured avian liver cells. J Biol Chem 257:4806–4811
Ahasan HA, Chowdhury MA, Azhar MA (1994) Copper sulfate poisoning. Trop Dr 24:52–53
Britton RS, Bacon BR (1994) Role of free radicals in liver diseases and hepatic fibrosis. Hepatogastroenterology 41:343–348
Brewer GJ (2007) A brand new mechanism for copper toxicity. J Hepatol 47:621–622
Sokol RJ, Devereaux MW, Khandwala RA, Loehr JP (1993) Abnormal hepatic mitochondrial respiration and cytochrome C oxidase activity in rats with long-term copper overload. Gastroenterology 105:178–187
Al-Nasser IA (2000) Cadmium hepatotoxicity and alterations of the mitochondrial function. Clin Toxicol 38:407–413
Rauen U, Petrat F, Sustmann R, de Groot H (2004) Iron-induced mitochondrial permeabiltiy transition in cultured hepatocytes. J Hepatol 40:607–615
Xiang LX, Shao JZ (2003) Role of intracellular Ca2+, reactive oxygen species, mitochondria transmembrane potential, and antioxidant enzymes in heavy metal-induced apoptosis in fish cells. Bull Environ Contam Toxicol 71:114–122
Pourahmad J, O’ Brien PJ (2000) A comparison of hepatocyte cytotoxic mechanisms for Cu2+ and Cd2+. Toxicology 143:263–273
Zhao M, Antunes F, Eaton JW, Brunk UT (2003) Lysosomal enzymes promote mitochondrial oxidant production, cytochrome c release and apoptosis. Eur J Biochem 270:3778–3786
Krumschnabel G, Manzl C, Berger C, Hofer B (2005) Oxidative stress, mitochondrial permeability transition and cell death in Cu-exposed trout hepatocytes. Toxicol Appl Pharmacol 209:62–73
Rongsheng S, Wang R, Guo S, Cao H, Pan J, Li C, Shi D, Tang Z (2011) In vitro effect of copper chloride exposure on reactive oxygen species generation and respiratory chain complex activities of mitochondria isolated from broiler liver. Biol Trace Elem Res 144:668–677
Garcia N, Martinez-Abundis E, Pavon N, Correa F, Chavez E (2007) Copper induces permeability transition through its interaction with the adenine nucleotide translocase. Cell Biol Int 31:893–899
Sies H (1993) Strategies of antioxidant defence. Eur J Biochem 215:213–219
Monedero-Prieto MJ, González-Pérez JM, González-Reimers E, Hernández-Pérez O, Monereo-Muñoz M, Galindo-Martín L, Quintero-Platt G, Abreu-González P (2014) Effects of selenium on liver and muscle contents and urinary excretion of zinc, copper, iron and manganese. Biol Trace Elem Res 158:224–229
Humphrey C, Pittman G (1974) A simple methylene blue-azure 2 basic fuchsin for epoxy-embedded tissue sections. Stain Technol 49:9–14
Tang Z, Iqbal M, Cawthon D (2002) Heart and breast muscle mitochondrial dysfunction in pulmonary hypertension syndrome in broilers (Gallus domesticus). Comp Biochem Physiol A 3:527–540
Bradford HF, Dodd RR (1977) Convulsions and activation of epileptic foci induced by monosodium glutamate and related compounds. Biochem Pharmacol 26:253–254
Sokol RJ, Devereaux MW, Traber MG, Shikes RH (1989) Copper toxicity and lipid peroxidation in isolated rat hepatocytes: effect of vitamin E. Pediatr Res 25:55–62
Mudassar S, Andrabi KI, Khullar M, Ganguly NK, Walia BNS (1992) Effect of exogenous copper on lipid peroxidation in rat hepatocytes. Possible involvement of protein kinase C. J Pharm Pharmacol 44:609–611
Zhang SS, Noordin MM, Rahman SO, Haron J (2000) Effects of copper overload on hepatic lipid peroxidation and antioxidant defense in rats. Vet Hum Toxicol 42:261–264
Chen JJ, Yu BP (1994) Alterations in mitochondrial membrane fluidity by lipid peroxidation products. Free Radic Biol Med 17:411–418
Ferrante MC, Meli R, Mattace Raso G, Esposito E, Severino L, Di Carlo G, Lucisano A (2002) Effect of fumonisin B1 on structure and function of macrophage plasma membrane. Toxicol Lett 129:181–187
Halliwell B, Chirico S (1993) Lipid peroxidation: its mechanism, measurement, and significance. Am J Clin Nutr 57(5Suppl):715S–724S, discussion 724S-725S
Loumbourdis NS, Vogiatzis AK (2002) Impact of cadmium on liver pigmentary system of the frog Rana ridibunda. Ecotoxicol Environ Saf 53:52–58
Bustamante J, Bredeston L, Malanga G, Mordoh J (1993) Role of melanin as a scavenger ofactive oxygen species. Pigment Cell Res 6:348–353
Rozanowska M, Sarna T, Land EJ, Truscott TG (1999) Free radical scavenging properties of melanin interaction of eu- and pheomelanin models with reducing and oxidizing radicals. Free Radic Biol Med 26:518–525
Loumbourdis NS (2007) Liver histopathologic alterations in the frog Rana ridibunda from a small river of northern Greece. Arch Environ Contam Toxicol 53:418–425
Acknowledgments
This project was supported by foundation of Guangdong Provincial Key Laboratory of Prevention and Control for Severe Clinical Animal Diseases (Grant No. 2013A061401013).
Conflict of Interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Additional information
Rongsheng Su and Huabin Cao contributed equally to this work.
Rights and permissions
About this article
Cite this article
Su, R., Cao, H., Pan, J. et al. The Protective Roles of Selenium on Hepatic Tissue Ultrastructure and Mitochondrial Antioxidant Capacity in Copper-Overloaded Rats. Biol Trace Elem Res 167, 110–114 (2015). https://doi.org/10.1007/s12011-015-0293-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12011-015-0293-8