Abstract
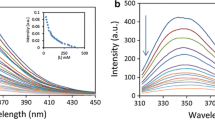
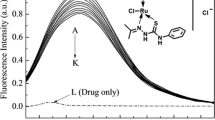
The interaction of ruthenium (II) complex [Ru(bpy)2(mal)]2+ (RBM) and [Ru(phen)2(mal)]2+ (RPM) (bpy = 2, 2-bipyridine, phen = 1,10-phenanthroline, mal = malonyl carboxylate) with human serum albumin (HSA) has been investigated by using fluorescence, UV absorption and circular dichroism (CD) spectroscopy approaches. A strong fluorescence quenching reaction of complexes to HSA was observed and the quenching mechanism was suggested as static quenching according to the Stern–Volmer (S–V) equation. The number of binding sites n and observed binding constant Kb was measured by fluorescence quenching method. The thermodynamic parameters ΔH, ΔS, and ΔG at different temperatures were calculated and the results indicate the binding reaction is mainly entropy-driven and Vander Waals force played a major role in the reaction. The result of CD showed that the secondary structure of HSA molecules was changed in the presence of the ruthenium (II) complexes. Furthermore, the cell viability of ruthenium (II) complexes was evaluated by MTT and complex RPM has shown significant higher anticancer potency than RBM against all the cell lines screened. RPM showed a significant antitumor activity through induction of apoptosis in A549 cells.








Similar content being viewed by others
References
Abuchowski A, Van Es T, Palczuk N, Davis F (1977) Alteration of immunological properties of bovine serum albumin by covalent attachment of polyethylene glycol. J Biol Chem 252:3578–3581
Yuan F, Chen X, Liu Y, Zhang T, Sun D, Liu J (2012) Chiral ruthenium complexes induce apoptosis of tumor cell and interact with bovine serum albumin. Chirality 24:174
Naik P, Chimatadar S, Nandibewoor S (2010) Interaction between a potent corticosteroid drug–dexamethasone with bovine serum albumin and human serum albumin: a fluorescence quenching and fourier transformation infrared spectroscopy study. J Photochem Photobiol B 100:147–159
He XM, Carter DC (1992) Atomic structure and chemistry of human serum albumin, Nature 209–215
Zsila F, Bikadi Z, Malik D, Hari P, Pechan I, Berces A, Hazai E (2011) Evaluation of drug–human serum albumin binding interactions with support vector machine aided online automated docking. Bioinformatics 27:1806–1813
Alessio E, Mestroni G, Bergamo A, Sava G (2004) Ruthenium antimetastatic agents. Curr Top Med Chem 4:1525–1535
Rademaker-Lakhai JM, van den Bongard D, Pluim D, Beijnen JH, Schellens JH (2004) A phase I and pharmacological study with imidazolium-trans-DMSO-imidazole-tetrachlororuthenate, a novel ruthenium anticancer agent. Clin Cancer Res 10:3717–3727
Levina A, Mitra A, Lay PA (2009) Recent developments in ruthenium anticancer drugs. Metallomics 1:458–470
Yu Q, Liu Y, Xu L, Zheng C, Le F, Qin X, Liu Y, Liu J (2014) Ruthenium (II) polypyridyl complexes: Cellular uptake, cell image and apoptosis of HeLa cancer cells induced by double targets. Eur J Med Chem 82:82–95
Zhang D, Huang J, Deng M, Zhang M, Zhou X. High fluorescence selectivity and visual detection of G-quadruplex structures by a novel dinuclear ruthenium complex. Chem Commun 46: 743–745
Gao H, Lei L, Liu J, Kong Q, Chen X, Hu Z (2004) The study on the interaction between human serum albumin and a new reagent with antitumour activity by spectrophotometric methods. J Photochem Photobiol A 167:213–221
Morgan O, Wang S, Bae SA, Morgan RJ, Baker AD, Strekas TC, Engel R (1997) Two complete stereochemical sets of dinuclear ruthenium complexes. J Chem Soc, Dalton Trans 3773–3776
Gil MA, Maside C, Cuello C, Parrilla I, Vazquez JM, Roca J, Martinez EA (2012) Effects of Hoechst 33342 staining and ultraviolet irradiation on mitochondrial distribution and DNA copy number in porcine oocytes and preimplantation embryos. Mol Reprod Dev 79:651–663
Huang H-L, Li Z-Z, Liang Z-H, Yao J-H, Liu Y-J (2011) Synthesis, cellular uptake, apopotosis, cytotoxicity, cell cycle arrest, interaction with DNA and antioxidant activity of ruthenium (II) complexes. Eur J Med Chem 46:3282–3290
Taghavi F, Moosavi-Movahedi A, Bohlooli M, Habibi-Rezaei M, Hadi Alijanvand H, Amanlou M, Sheibani N, Saboury A, Ahmad F (2014) Energetic domains and conformational analysis of human serum albumin upon co-incubation with sodium benzoate and glucose. J Biomol Struct Dyn 32:438–447
Wang Y, Wang X, Wang J, Zhao Y, He W, Guo Z (2011) Noncovalent interactions between a trinuclear monofunctional platinum complex and human serum albumin. Inorg Chem 50:12661–12668
Wang YQ, Zhang HM, Zhang GC (2006) Studies of the interaction between palmatine hydrochloride and human serum albumin by fluorescence quenching method. J Pharm Biomed 41:1041–1046
Zsila F, Bikdi Z, Simonyi M (2003) Probing the binding of the flavonoid, quercetin to human serum albumin by circular dichroism, electronic absorption spectroscopy and molecular modelling methods. Biochem Pharmacol 65:447–456
Sengupta B, Sengupta PK (2002) The interaction of quercetin with human serum albumin: a fluorescence spectroscopic study. Biochem Biophys Res Commun 299:400–403
Zhang YZ, Dai J, Zhang XP, Yang X, Liu Y (2008) Studies of the interaction between Sudan I and bovine serum albumin by spectroscopic methods. J Mol Struct 888:152–159
Lehrer S (1971) Solute perturbation of protein fluorescence. Quenching of the tryptophyl fluorescence of model compounds and of lysozyme by iodide ion. Biochemistry 10:3254–3263
Rubinszfein DC, DiFiglia M, Heintz N, Nixon RA, Qin Z-H, Ravikumar B, Stefanis L, Tolkovsy A (2005) Autophagy and its possible roles in nervous system diseases, damage and repair. Autophagy 1:11–22
Yan L, Wang X, Wang Y, Zhang Y, Li Y, Guo Z (2012) Cytotoxic palladium (II) complexes of 8-aminoquinoline derivatives and the interaction with human serum albumin. J Inorg Biochem 106(1):46–51
Porphyria, V. van’t Hoff Equation
Liu Y, Yu Q, Wang C, Sun D, Huang Y, Zhou Y, Liu J (2012) Ruthenium (II) complexes binding to human serum albumin and inducing apoptosis of tumor cells. Inorg Chem Commun 24:104–109
Sun D, Liu Y, Liu D, Zhang R, Yang X, Liu J (2012) Stabilization of Quadruplex DNA, Inhibition of Telomerase Activity and Live Cell Imaging Studies of Chiral Ruthenium (II) Complexes. Chem Eur J 18(14):4285–4295
Taylor RC, Cullen SP, Martin SJ (2008) Apoptosis: controlled demolition at the cellular level. Nat Rev Mol Cell Bio 9:231–241
Yang X, Chen L, Liu Y, Yang Y, Chen T, Zheng W, Liu J, He QY (2012) Ruthenium methylimidazole complexes induced apoptosis in lung cancer A549 cells through intrinsic mitochondrial pathway. Biochimie 94(2):345–353
Acknowledgments
This work was supported by the National Natural Science Foundation of China (21171070, 21371075), the Planned Item of Science and Technology of Guangdong Province (c1211220800571), and the Fundamental Research Funds for the Central Universities.
Conflict of Interest
There are no conflicts of interest to declare.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(DOC 429 kb)
Rights and permissions
About this article
Cite this article
Sun, J., Huang, Y., Zheng, C. et al. Ruthenium (II) Complexes Interact with Human Serum Albumin and Induce Apoptosis of Tumor Cells. Biol Trace Elem Res 163, 266–274 (2015). https://doi.org/10.1007/s12011-014-0165-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12011-014-0165-7