Abstract
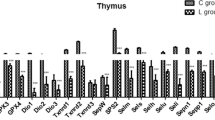
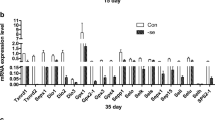
This study describes the effects of selenium (Se) deficiency on the messenger ribonucleic acid (mRNA) expression of 25 selenoproteins (Sels) (including glutathione peroxidases (GPx1–GPx4), thioredoxin reductases (TrxR1–TrxR3), iodothyronine deiodinases (ID1–ID3), selenophosphate synthetase 2 (SPS2), 15-kDa Sel (Sel15), SelH, SelI, SelK, SelM, Sepn1, SelO, Sepx, Selpb, SelS, SelT, SelW, Sepp1, and SelU in the adipose tissues (subcutaneous adipose, visceral adipose, and articular adipose) of chickens. One hundred and fifty 1-day-old chickens were randomly assigned to two groups of 75 each and were fed a low-Se diet (0.032 mg/kg Se) or a control diet (0.282 mg/kg Se). The expression levels of 25 Sel mRNAs were determined on days 35, 45, and 55 from three parts (subcutaneous adipose, visceral adipose, and articular adipose) of the chicken adipose tissues. The results showed that the expression levels of the 25 Sel mRNAs were significantly lower (P < 0.05) in the low-selenium group than in the control group. In addition, the Sel mRNA expression levels in the three adipose tissues were observed to decrease in a time-dependent manner with increasing feeding time.



Similar content being viewed by others
References
Foreman JH, Potter KA, Bayly WM et al (1986) Generalized steatitis associated with selenium deficiency and normal vitamin E status in a foal. J Am Vet Med Assoc 189:83–86
Geloen A, Arthur JR, Beckett GJ et al (1990) Effect of selenium and iodine deficiency on the level of uncoupling protein in brown adipose tissue of rats. Biochem Soc Trans 18:1269–1270
Fytianou A, Koutinas AF, Saridomichelakis MN et al (2006) Blood alpha-Tocopherol, selenium, and glutathione peroxidase changes and adipose tissue fatty acid changes in kittens with experimental steatitis (yellow fat disease): a comparative study between the domestic shorthaired and Siamese breed. Biol Trace Elem Res 112:131–143
Rasmussen LB, Hollenbach B, Laurberg P et al (2009) Serum selenium and selenoprotein P status in adult Danes—8-year followup. J Trace Elem Med Biol 23:265–271
Burk RF, Hill KE, Motley AK et al (2006) Deletion of selenoprotein P upregulates urinary selenium excretion and depresses whole-body selenium content. Biochim Biophys Acta 1760:1789–1793
Mariotti M, Ridge PG, Zhang Y et al (2012) Composition and evolution of the vertebrate and mammalian selenoproteomes. PLoS ONE 7:e33066
Aachmann FL, Fomenko DE, Soragni A et al (2007) Solution structure of selenoprotein W and NMR analysis of its interaction with 14-3-3 proteins. J Biol Chem 282:37036–37044
Novoselov SV, Kryukov GV, Xu XM et al (2007) Selenoprotein H is a nucleolar thioredoxin-like protein with a unique expression pattern. J Biol Chem 282:11960–11968
Li JL, Li HX, Li S et al (2012) Effects of Selenoprotein W gene expression by selenium involves regulation of mRNA stability in chicken embryos neurons. Biometals 25:459–468
Gao X, Xing H, Li S et al (2012) Selenium regulates gene expression of selenoprotein W in chicken gastrointestinal tract. Biol Trace Elem Res 145:181–188
Zhang JL, Li JL, Huang XD et al (2012) Dietary selenium regulation of transcript abundance of selenoprotein N and selenoprotein W in chicken muscle tissues. Biometals 25:297–307
Sun B, Wang R, Li J et al (2011) Dietary selenium affects selenoprotein W gene expression in the liver of chicken. Biol Trace Elem Res 143:1516–1523
Wang R, Sun B, Zhang Z et al (2011) Dietary selenium influences pancreatic tissue levels of selenoprotein W in chickens. J Inorg Biochem 105:1156–1160
Yu D, Li JL, Zhang JL et al (2011) Effects of dietary selenium on selenoprotein W gene expression in the chicken immune organs. Biol Trace Elem Res 144:678–687
Sheng PF, Jiang Y, Zhang ZW et al (2014) The effect of Se-deficient diet on gene expression of inflammatory cytokines in chicken brain. Biometals 27:33–43
Li JL, Li HX, Gao XJ et al (2012) Priority in selenium homeostasis involves regulation of SepSecS transcription in the chicken brain. PLoS ONE 7:e35761
Yao HD, Wu Q, Zhang ZW et al (2013) Gene expression of endoplasmic reticulum resident selenoproteins correlates with apoptosis in various muscles of se-deficient chicks. J Nutr 143:613–619
Peirson SN, Butler JN, Foster RG (2003) Experimental validation of novel and conventional approaches to quantitative real-time PCR data analysis. Nucleic Acids Res 31:e73
Pfaffl MW (2001) A new mathematical model for relative quantification in real-time RT-PCR. Nucleic Acids Res 29:e45
Jotty K, Ojeda ML, Nogales F et al (2013) Selenium dietary supplementation as a mechanism to restore hepatic selenoprotein regulation in rat pups exposed to alcohol. Alcohol 47:545–552
Kershaw EE, Flier JS (2004) Adipose tissue as an endocrine organ. J Clin Endocrinol Metab 89:2548–2556
Peinado JR, Jimenez-Gomez Y, Pulido MR et al (2010) The stromal-vascular fraction of adipose tissue contributes to major differences between subcutaneous and visceral fat depots. Proteomics 10:3356–3366
Aribi M, Merzouk H, Haddouche M et al (2010) Clinical evaluation of lipids, lipoproteins and red blood cells sodium and potassium in patients with different grades of hypertension. Clin Biochem 43:942–947
Jayaprakash V, Marshall JR (2011) Selenium and other antioxidants for chemoprevention of gastrointestinal cancers. Best Pract Res Clin Gastroenterol 25:507–518
Keshavarzi B, Moore F, Najmeddin A et al (2012) The role of selenium and selected trace elements in the etiology of esophageal cancer in high risk Golestan province of Iran. Sci Total Environ 433:89–97
Wang X, Zhang W, Chen H et al (2014) High selenium impairs hepatic insulin sensitivity through opposite regulation of ROS. Toxicol Lett 224:16–23
Rayman MP, Stranges S (2013) Epidemiology of selenium and type 2 diabetes: can we make sense of it. Free Radic Biol Med 65:1557–1564
Reeves MA, Hoffmann PR (2009) The human selenoproteome: recent insights into functions and regulation. Cell Mol Life Sci 66:2457–2478
Utomo A, Jiang X, Furuta S et al (2004) Identification of a novel putative non-selenocysteine containing phospholipid hydroperoxide glutathione peroxidase (NPGPx) essential for alleviating oxidative stress generated from polyunsaturated fatty acids in breast cancer cells. J Biol Chem 279:43522–43529
Combs GF Jr, Gray WP (1998) Chemopreventive agents: selenium. Pharmacol Ther 79:179–192
Zoidis E, Pappas AC, Georgiou CA et al (2010) Selenium affects the expression of GPx4 and catalase in the liver of chicken. Comp Biochem Physiol B Biochem Mol Biol 155:294–300
Bianco AC, Salvatore D, Gereben B et al (2002) Biochemistry, cellular and molecular biology, and physiological roles of the iodothyronine selenodeiodinases. Endocr Rev 23:38–89
Kohrle J, Gartner R (2009) Selenium and thyroid. Best Pract Res Clin Endocrinol Metab 23:815–827
Holmgren A (1989) Thioredoxin and glutaredoxin systems. J Biol Chem 264:13963–13966
Lu J, Holmgren A (2009) Selenoproteins. J Biol Chem 284:723–727
Geisberger R, Kiermayer C, Homig C et al (2007) B- and T-cell-specific inactivation of thioredoxin reductase 2 does not impair lymphocyte development and maintenance. Biol Chem 388:1083–1090
Sun Y, Rigas B (2008) The thioredoxin system mediates redox-induced cell death in human colon cancer cells: implications for the mechanism of action of anticancer agents. Cancer Res 68:8269–8277
Sunde RA, Raines AM, Barnes KM et al (2009) Selenium status highly regulates selenoprotein mRNA levels for only a subset of the selenoproteins in the selenoproteome. Biosci Rep 29:329–338
Panee J, Stoytcheva ZR, Liu W et al (2007) Selenoprotein H is a redox-sensing high mobility group family DNA-binding protein that up-regulates genes involved in glutathione synthesis and phase II detoxification. J Biol Chem 282:23759–23765
Kipp AP, Banning A, van Schothorst EM et al (2012) Marginal selenium deficiency down-regulates inflammation-related genes in splenic leukocytes of the mouse. J Nutr Biochem 23:1170–1177
Ye Y, Shibata Y, Yun C et al (2004) A membrane protein complex mediates retro-translocation from the ER lumen into the cytosol. Nature 429:841–847
Curran JE, Jowett JB, Elliott KS et al (2005) Genetic variation in selenoprotein S influences inflammatory response. Nat Genet 37:1234–1241
Kim KH, Gao Y, Walder K et al (2007) SEPS1 protects RAW264.7 cells from pharmacological ER stress agent-induced apoptosis. Biochem Biophys Res Commun 354:127–132
Pappas AC, Zoidis E, Surai PF et al (2008) Selenoproteins and maternal nutrition. Comp Biochem Physiol B Biochem Mol Biol 151:361–372
Loflin J, Lopez N, Whanger PD et al (2006) Selenoprotein W during development and oxidative stress. J Inorg Biochem 100:1679–1684
Korotkov KV, Kumaraswamy E, Zhou Y et al (2001) Association between the 15-kDa selenoprotein and UDP-glucose:glycoprotein glucosyltransferase in the endoplasmic reticulum of mammalian cells. J Biol Chem 276:15330–15336
Kryukov GV, Castellano S, Novoselov SV et al (2003) Characterization of mammalian selenoproteomes. Science 300:1439–1443
Petit N, Lescure A, Rederstorff M et al (2003) Selenoprotein N: an endoplasmic reticulum glycoprotein with an early developmental expression pattern. Hum Mol Genet 12:1045–1053
Acknowledgments
This study was supported by the Major Projects of International Cooperation and Exchanges NSFC (31320103920), the National Natural Science Foundation of China (31272626), the Doctoral Fund of the Ministry of Education of China (20122325110018), and the Heilongjiang Postdoctoral Fund (LBH-Z13028). The authors thank Elsevier English Language Editing System in correcting grammatical, spelling, and other common errors.
Author information
Authors and Affiliations
Corresponding authors
Additional information
All authors have read the manuscript and agreed to submit it in its current form for consideration for publication in the journal
Rights and permissions
About this article
Cite this article
Liang, Y., Lin, Sl., Wang, Cw. et al. Effect of Selenium on Selenoprotein Expression in the Adipose Tissue of Chickens. Biol Trace Elem Res 160, 41–48 (2014). https://doi.org/10.1007/s12011-014-0024-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12011-014-0024-6