Abstract
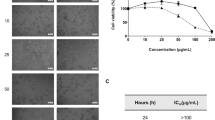
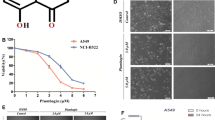
Lung cancer is one of the leading causes of death in the world, and non-small cell lung carcinoma (NSCLC) accounts for approximately 75–85% of all lung cancers. In the present work, we studied the cytotoxic activity, cell cycle arrest and induction apoptosis of the compound cis-(dichloro)tetramineruthenium(III) chloride {cis-[RuCl2(NH3)4]Cl} in human lung carcinoma tumor cell line A549. The results of MTT and trypan blue assays showed that cis-[RuCl2(NH3)4]Cl causes reduction in the viability of A549 cells when treating with 95 and 383 μM of the compound for 48 and 72 h. Lower concentrations of the compound (19, 3.8 and 0.38 μM), however, only slightly affected cell viability. The IC50 value for the compound was about 383 μM. Survival analysis of the A549 cells after treatment with ruthenium(III) compound using long term clonogenic assay showed that it reduced colony formation ability at concentrations of 0.38 and 3.8 μM, and at concentrations of 95 and 383 μM no colonies were observed. Cell cycle analysis showed that compound ruthenium led to an accumulation of A549 cells in S phase and increased in the sub-G1 peak. In addition, cis-(dichloro)tetramineruthenium(III) chloride treatment induced apoptosis, as observed by the increased numbers of annexin V-positive cells and increased messenger RNA expression of caspase-3.







Similar content being viewed by others
References
American Cancer Society (2009) Cancer facts & figures. American Cancer Society, Atlanta
Socinski MA (2004) Cytotoxic chemotherapy in advanced non-small cell lung cancer: a review of standard treatment paradigms. Clin Cancer Res 10:4210–4214
Rosell R, Reginald VNL, Taron M, Reguart N (2002) DNA repair and cisplatin resistance in non-small-cell lung cancer. Lung Cancer 38:217–227
Kelland L (2007) The resurgence of platinum-based cancer chemotherapy. Nature Reviews 7:573–584
Stinchcombe TE, Socinski MA (2009) Current treatments for advanced stage non-small cell lung cancer. Proc Am Thorac Soc 6:233–241
Otal E, Abel Y, Oshika Y et al (1995) Expression of the multidrug resistance-associated protein (MRP) gene in non-small-cell lung cancer. Br J Cancer 72:550–554
Clarke MJ (2003) Ruthenium metallopharmaceuticals. Coord Chem Rev 236:209–233
Kostova I (2006) Ruthenium complexes as anticancer agents. Curr Med Chem 13:1085–1107
Menezes CSR, Costa LCGP, de MR Ávila V et al (2007) Analysis in vivo of antitumor activity, cytotoxicity and interaction between plasmid DNA and the cis-dichlorotetraammineruthenium(III) chloride. Chem–Biol Interact 167:116–124
Allardyce CS, Dyson PJ (2001) Ruthenium in medicine: current clinical uses and future prospects. Platinum Metals Rev 45:62–69
Brabec V, Nováková O (2006) DNA binding mode of ruthenium complexes and relationship to tumor cell toxicity. Drug Resist Updat 9:111–122
Bergamo A, Gagliardi R, Scarcia V et al (1999) In vitro cell cycle arrest, in vivo action on solid metastasizing tumors and host toxicity of the antimetastatic drug NAMI-A and cisplatin. J Pharmacol Exp Ther 289:559–564
Scolaro C, Bergamo A, Brescacin L et al (2005) In vitro and in vivo evaluation of ruthenium (II)–arene PTA complexes. Journal Med Chem 48:4161–4171
Rademaker-Lakhai JM, van den Bongard D, Pluim D et al (2004) A phase I and pharmacological study with imidazolium-trans-DMSO-imidazole-tetrachlororuthenate, a novel ruthenium anticancer agent. Clin Cancer Res 10:3717–3727
Hartinger CG, Zorbas-Seifried S, Jakupec MA et al (2006) From bench to beside—preclinical and early clinical development of the anticancer agent indazolium trans-[tetrachlorobis (1H-indazazole)ruthenate (III)] KP1019 or FFC14A. J Inorg Biochem 100:891–904
Silveira-lacerda EP, Vilanova-costa CAST, Hamaguchi A et al (2009) The ruthenium complex cis-(dichloro)tetraammineruthenium(III) chloride presents selective cytotoxicity against murine B cell lymphoma (A-20), murine ascitic sarcoma 180 (S-180), human breast adenocarcinoma (SK-BR-3), and human T cell leukemia (Jurkat) tumor cell lines. Biol Trace Elem Res 135:98–111
Mosman T (1983) Rapid colorimetric assay for cellular growth an survival: application to proliferation an cytotoxicity assays. J Immunol Methods 16:55–63
Martins AKA (2005) Técnicas básicas em cultura de células. In: Peres MC, Curi R (eds) Como Cultivar Células, 1st edn. Guanabara Koogan, Rio de Janeiro, pp 10–24
Lima AP, Pereira FC, Vilanova-Costa CAST (2010) The compound cis-(dichloro)tetrammineruthenium(III) chloride induces caspase-mediated apoptosis in K562 cells. Toxicol Vitr 24:1562–1568
Lima AP, Pereira FC, Vilanova CAST (2010) The ruthenium complex cis-(dichloro)tetrammineruthenium(III) chloride induces apoptosis and damages DNA in murine sarcoma 180 cells. J Biosci 35:371–378
Capozzi I, Katiuscia C, Cocchietto M et al (1998) Modification of cell cycle and viability of TLX5 lymphoma in vitro by sulfoxide-ruthenium compounds and cisplatin detected by flow cytometry. Chemico-Biological Interactions 113:51–64
Kapitza S, Pongratz M, Jakupec MA et al (2005) Heterocyclic complexes of ruthenium (III) induce apoptosis in colorectal carcinoma cells. Journal Cancer Res Oncol 131:101–110
Krajewska M, Kim H, Shin E et al (2005) Tumor-associated alterations in caspase-14 expression in epithelial malignancies. Clin Cancer Res 11:5462–5471
Cohen GM (1997) Caspases: the executioners of apoptosis. Biochem J 326:1–16
Lowe SW, Lin AW (2000) Apoptosis in cancer. Carcinogenesis 21:485–495
Gallori E, Vettori C, Alessio E et al (2000) DNA as a possible target for antitumor ruthenium (III) complexes. Arch Biochem Biophys 376:156–162
Brabec V, Nováková O (2006) DNA binding mode of ruthenium complexes and relationship to tumor cell toxicity. Drug Resistance Updates 9:111–122
Acknowledgments
The authors gratefully acknowledge the financial support of Research and Projects Financing (FINEP) (grant No. 01.06.0941.00/CT-Saúde to Elisângela de Paula Silveira-Lacerda). Support from Coordination for the Advancement of Higher Education Staff (CAPES) was provided through a fellowship to Aliny Pereira de Lima and Cesar Augusto Sam Tiago Vilanova-Costa, and from the Brazilian National Counsel of Technological and Scientific Development (CNPq) through a fellowship to Flávia de Castro Pereira (grant No. 131960/2008-3).
Conflict of Interest
There are no financial or personal interests that might be viewed as inappropriate influences on the work presented herein.
Ethical Approval
No studies involving humans or experimental animals were conducted in this work. The human lung carcinoma A549 cells were purchased from the Rio de Janeiro Cell Bank (RJ, Brazil) and cultured in vitro.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
de Lima, A.P., Pereira, F.d.C., Vilanova-Costa, C.A.S.T. et al. Induction of Cell Cycle Arrest and Apoptosis by Ruthenium Complex cis-(Dichloro)tetramineruthenium(III) Chloride in Human Lung Carcinoma Cells A549. Biol Trace Elem Res 147, 8–15 (2012). https://doi.org/10.1007/s12011-011-9275-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12011-011-9275-7