Abstract
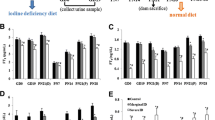
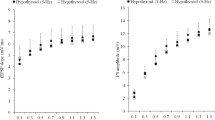
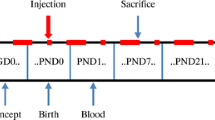
Iodine is essential for the synthesis of triiodothyronine (T3) and thyroxine (T4). Iodine deficiency leads to inadequate thyroid hormone. Hypothyroidism induced by iodine deficiency during gestation and postnatal period leads to cognitive deficits in learning and memory. However, the mechanism underlying these deficits is unclear. Calcium-dependent calmodulin kinase II (CaMKII) known as a potential memory molecule regulates important neuronal functions including learning and memory. Recent studies have shown that hypothyroidism alters phosphorylation of CaMKII in hippocampus or even in sympathetic ganglia of rats. Though the entorhinal cortex (EC) is an important functional structure within the neuronal network responsible for learning and memory, little is known about the effect of hypothyroidism on phosphorylation of CaMKII in the EC. Here, we report that iodine deficiency and propylthiouracil treatment through gestation and lactation reduce phosphorylation of CaMKII in the EC of pups. The increase of calcineurin, as well as reduction of neurogranin and calmodulin, may account for the reduced phosphorylation of CaMKII induced by developmental iodine deficiency and hypothyroidism. These findings in the EC may contribute to understanding the mechanisms that underlie impairment of learning and memory induced by developmental iodine deficiency and hypothyroidism.




Similar content being viewed by others
References
Martinez-Galan JR, Pedraza P, Santacana M, Escobar del Rey F, Morreale de Escobar G, Ruiz-Marcos A (1997) Myelin basic protein immunoreactivity in the internal capsule of neonates from rats on a low iodine intake or on methylmercaptoimidazole (MMI). Brain Res Dev Brain Res 101:249–256
Sui L, Gilbert ME (2003) Pre- and postnatal propylthiouracil-induced hypothyroidism impairs synaptic transmission and plasticity in area CA1 of the neonatal rat hippocampus. Endocrinology 144:4195–4203
Kibirige MS, Hutchison S, Owen CJ, Delces HT (2004) Prevalence of maternal dietary iodine insufficiency in the north east of England: implications for the fetus. Arch Dis Child Fetal Neonatal Ed 89:F436–F439
Berbel P, Obregon MJ, Bernal J, Escobar del Rey F, Morreale de Escobar G (2007) Iodine supplementation during pregnancy: a public health challenge. Trends Endocrinol Metab 18:338–343
Tang Z, Liu W, Yin H, Wang P, Dong J, Wang Y, Chen J (2007) Investigation of intelligence quotient and psychomotor development in schoolchildren in areas with different degrees of iodine deficiency. Asia Pac J Clin Nutr 16:731–737
Dussault JH, Ruel J (1987) Thyroid hormones and brain development. Annu Rev Physiol 49:321–334
Yasuda M, Mayford MR (2006) CaMKII activation in the entorhinal cortex disrupts previously encoded spatial memory. Neuron 50:309–318
Majchrzak M, Ferry B, Marchand AR, Herbeaux K, Seillier A, Barbelivien A (2006) Entorhinal cortex lesions disrupt fear conditioning to background context but spare fear conditioning to a tone in the rat. Hippocampus 16:114–124
Harich S, Kinfe T, Koch M, Schwabe K (2008) Neonatal lesions of the entorhinal cortex induce long-term changes of limbic brain regions and maze learning deficits in adult rats. Neuroscience 153:918–928
Gomez-Isla T, Price JL, McKeel DW Jr, Morris JC, Growdon JH, Hyman BT (1996) Profound loss of layer II entorhinal cortex neurons occurs in very mild Alzheimer's disease. J Neurosci 16:4491–4500
Diekmann S, Nitsch R, Ohm TG (1994) The organotypic entorhinal-hippocampal complex slice culture of adolescent rats. A model to study transcellular changes in a circuit particularly vulnerable in neurodegenerative disorders. J Neural Transm Suppl 44:61–71
Lippa CF, Hamos JE, Pulaski-Salo D, DeGennaro LJ, Drachman DA (1992) Alzheimer's disease and aging: effects on perforant pathway perikarya and synapses. Neurobiol Aging 13:405–411
Zhou W, Jiang D, Raisman G, Zhou C (1998) Embryonic entorhinal transplants partially ameliorate the deficits in spatial memory in adult rats with entorhinal cortex lesions. Brain Res 792:97–104
Yamauchi T (2005) Neuronal Ca2+/calmodulin-dependent protein kinase II—discovery, progress in a quarter of a century, and perspective: implication for learning and memory. Biol Pharm Bull 28:1342–1354
Limback-Stokin K, Korzus E, Nagaoka-Yasuda R, Mayford M (2004) Nuclear calcium/calmodulin regulates memory consolidation. J Neurosci 24:10858–10867
Ran X, Miao HH, Sheu FS, Yang D (2003) Structural and dynamic characterization of a neuron-specific protein kinase C substrate, neurogranin. Biochemistry 42:5143–5150
Wu J, Huang KP, Huang FL (2003) Participation of NMDA-mediate phosphorylation and oxidation of neurogranin in the regulation of Ca2+ and Ca2+/calmodulin-dependent neuronal signaling in the hippocampus. J Neurochem 86:1524–1533
Mansuy IM (2003) Calcineurin in memory and bidirectional plasticity. Biochem Biophys Res Commun 311:1195–1208
Gerges NZ, Alzoubi KH, Alkadhi KA (2005) Role of phosphorylated CaMKII and calcineurin in the differential effect of hypothyroidism on LTP of CA1 and dentate gyrus. Hippocampus 15:480–490
Alzoubi KH, Bedawi AS, Aleisa AM, Alkadhi KA (2004) Hypothyroidism impairs long-term potentiation in sympathetic ganglia: electrophysiologic and molecular studies. J Neurosci Res 78:393–402
Alzoubi KH, Gerges NZ, Alkadhi KA (2005) Levothyroxin restores hypothyroidism-induced impairment of LTP of hippocampal CA1: electrophysiological and molecular studies. Exp Neurol 195:330–341
Alzoubi KH, Aleisa AM, Alkadhi KA (2006) Molecular studies on the protective effect of nicotine in adult-onset hypothyroidism-induced impairment of long-term potentiation. Hippocampus 16:861–874
Dong J, Yin H, Liu W, Wang P, Jiang Y, Chen J (2005) Congenital iodine deficiency and hypothyroidism impair LTP and decrease c-fos and c-jun expression in rat hippocampus. Neurotoxicology 26:417–426
Bradford M (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254
Wong CC, Leung MS (2001) Effects of neonatal hypothyroidism on the expressions of growth cone proteins and axon guidance molecules related genes in the hippocampus. Mol Cell Endocrinol 184:143–150
Li J, Yang C, Han S, Zu P, Wu J, Xu Q, Fang L (2006) Increased phosphorylation of neurogranin in the brain of hypoxic preconditioned mice. Neurosci Lett 391:150–153
Colbran RJ (1992) Regulation and role of brain calcium/calmodulin-dependent protein kinase II. Neurochem Int 21:469–497
D'Alcantara P, Schiffmann SN, Swillens S (2003) Bidirectional synaptic plasticity as a consequence of interdependent Ca2+-controlled phosphorylation and dephosphorylation pathways. Eur J NeuroSci 17:2521–2528
Hudmon A, Schulman H (2002) Neuronal CA2+/calmodulin-dependent protein kinase II: the role of structure and autoregulation in cellular function. Annu Rev Biochem 71:473–510
Sethi V, Kapil U (2004) Iodine deficiency and development of brain. Indian J Pediatr 71:325–329
Hetzel BS (2000) Iodine and neuropsychological development. J Nutr 130:493S–495S
Zoeller RT, Rover J (2004) Timing of thyroid hormone action in the developing brain: clinical observations and experimental findings. J Neuroendocrinol 16:809–818
Kobayashi K, Tsuji R, Yoshioka T, Kushida M, Yabushita S, Sasaki M, Mino T, Seki T (2005) Effects of hypothyroidism induced by perinatal exposure to PTU on rat behavior and synaptic gene expression. Toxicology 212:135–147
Calloni GW, Penno CA, Cordova FM, Trentin AG, Neto VM, Leal RB (2005) Congenital hypothyroidism alters the phosphorylation of ERK1/2 and p38MAPK in the hippocampus of neonatal rats. Brain Res Dev Brain Res 154:141–145
Sui L, Anderson WL, Gilbert ME (2005) Impairment in short-term but enhanced long-term synaptic potentiation and ERK activation in adult hippocampal area CA1 following developmental thyroid hormone insufficiency. Toxicol Sci 85:647–656
Lisman J, Schulman H, Cline H (2002) The molecular basis of CaMKII function in synaptic and behavioural memory. Nat Rev Neurosci 3:175–190
Cammarota M, Bevilaqua LR, Viola H, Kerr DS, Reichmann B, Teixeira V, Bulla M, Izquierdo I, Medina JH (2002) Participation of CaMKII in neuronal plasticity and memory formation. Cell Mol Neurobiol 22:259–267
Miyamoto E (2006) Molecular mechanism of neuronal plasticity: induction and maintenance of long-term potentiation in the hippocampus. J Pharmacol Sci 100:433–442
Iniguez MA, De Lecea L, Guadano-Ferraz A, Morte B, Gerendasy D, Sutcliffe JG, Bernal J (1996) Cell-specific effects of thyroid hormone on RC3/neurogranin expression in rat brain. Endocrinology 137:1032–1041
Iniguez MA, Rodriguez-Pena A, Ibarrola N, Aguilera M, Munoz A, Bernal J (1993) Thyroid hormone regulation of RC3, a brain-specific gene encoding a protein kinase-C substrate. Endocrinology 133:467–473
Martinez de Arrieta C, Morte B, Coloma A, Bernal J (1999) The human RC3 gene homolog, NRGN contains a thyroid hormone-responsive element located in the first intron. Endocrinology 140:335–343
Dineley KT, Hogan D, Zhang WR, Taglialatela G (2007) Acute inhibition of calcineurin restores associative learning and memory in Tg2576 APP transgenic mice. Neurobiol Learn Mem 88:217–224
Mansuy IM, Mayford M, Jacob B, Kandel ER, Bach ME (1998) Restricted and regulated overexpression reveals calcineurin as a key component in the transition from short-term to long-term memory. Cell 92:39–49
Mansuy IM, Winder DG, Moallem TM, Osman M, Mayford M, Hawkins RD, Kandel ER (1998) Inducible and reversible gene expression with the rtTA system for the study of memory. Neuron 21:257–265
Malleret G, Haditsch U, Genoux D, Jones MW, Bliss TV, Vanhoose AM, Weitlauf C, Kandel ER, Winder DG, Mansuy IM (2001) Inducible and reversible enhancement of learning, memory, and long-term potentiation by genetic inhibition of calcineurin. Cell 104:675–686
Wen Z, Guirland C, Ming GL, Zheng JQ (2004) A CaMKII/calcineurin switch controls the direction of Ca(2+)-dependent growth cone guidance. Neuron 43:835–846
Kazi TG, Kandhro GA, Afridi HI, Kazi N, Baig JA, Arain MB, Shah AQ, Syed N, Kumar S, Kolachi NF, Khan S (2009) Interaction of copper with iron, iodine, and thyroid hormone status in goitrous patients. Biol Trace Elem Res. doi:10.1007/s12011-009-8478-7
Shrader RE, Keen CL, Hurley LS, Zeman FJ (1982) Hematologic and trace element alterations following chronic maternal ingestion of propylthiourea. Exp Hematol 10:44–55
Gonzalez-Reimers E, Santolaria-Fernandez F, Perez-Labajos J, Rodriguez-Moreno F, Martinez-Riera A, Hernandez-Torres O, Valladares-Parrilla F, Molina-Perez M (1996) Relative and combined effects of propylthiouracil, ethanol and protein deficiency on liver histology and hepatic iron, zinc, manganese and copper contents. Alcohol Alcohol 31:535–545
Acknowledgments
This work was supported by the National Natural Science Foundation of China [grant number 30500400 and 30800896] and also supported by Natural Science Foundation of Liaoning Province, China for PhD holders [grant number 20081053]. We thank Prof. Hong Lai from the Department of Anatomy College of Basic Medical Sciences at China Medical University for her excellent technical assistance with the experiments of rat brain anatomy.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Wang, Y., Hou, Y., Dong, J. et al. Developmental Iodine Deficiency and Hypothyroidism Reduce Phosphorylation of Calcium/Calmodulin-Dependent Kinase II in the Rat Entorhinal Cortex. Biol Trace Elem Res 137, 353–363 (2010). https://doi.org/10.1007/s12011-009-8591-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12011-009-8591-7