Abstract
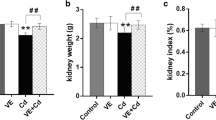
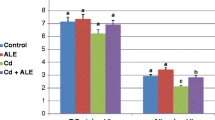
Cadmium (Cd) exposure has been recognized to result in a wide variety of cellular responses, including oxidative stress and body weight loss. The aim of the present study was to examine the effect of lycopene supplementation on the antioxidant defense system, lipid peroxidation (LPO) level, nitric oxide (NO), tumor necrosis factor alpha (TNF-α) production, and body weight in Cd-exposed rats. Animals were divided into four groups (n = 7): control, Cd-treated, Cd plus lycopene-treated, and lycopene-treated. Cadmium (as CdCl2) was administrated orally for 20 days (6.6 mg kg−1 day−1), and lycopene (10 mg kg−1 day−1) was similarly administered. Lycopene administration significantly suppressed Cd-induced LPO in plasma and kidney homogenates. Lycopene also reversed Cd-decreased body weight compared to the control. Cadmium treatment had diverse effects on the antioxidant enzyme activities. Although antioxidant superoxide dismutase activity was unchanged, glutathione peroxidase activity was decreased, and catalase activity was elevated in kidney homogenates of Cd-administrated group. However, lycopene treatment reversed Cd-changed enzyme activities to the control level. Xanthine oxidase activity and TNF-α concentration were not altered by Cd administration, indicating that superoxide anion production and inflammation were not stimulated. Cadmium did not change NO levels in kidney homogenates but decreased those in plasma, and this effect was not prevented by lycopene supplementation. The result suggests that consumption of adequate levels of lycopene may be useful to prevent heavy-metal-induced LPO and body weight loss.


Similar content being viewed by others
References
Waalkes MP (2000) Cadmium carcinogenesis. J Inorg Biochem 79:241–244
Rikans LE, Yamano T (2000) Mechanisms of cadmium mediated acute hepatotoxicity. J Biochem Mol Toxicol 14:110–117
Galan A, Garcia-Bermejo L, Troyano A, Vilaboa NE, Fernandez C, de Blas E, Aller P (2001) The role of intracellular oxidation in death induction (apoptosis and necrosis) in human promonocytic cells treated with stress inducers (cadmium, heat, X-rays). Eur J Cell Biol 80:312–320
Watanabe M, Henmi K, Ogawa K, Suzuki T (2003) Cadmium dependent generation of reactive oxygen species and mitochondrial DNA breaks in photosynthetic and non-photosynthetic strains of Euglena gracilis. Comp Biochem Physiol C Toxicol Pharmacol 134:227–234
Amoruso MA, Witz G, Goldstein BD (1982) Enhancement of rat and human phagocyte superoxide anion radical production by cadmium in vitro. Toxicol Lett 10:133–138
Ochi T, Otsuka F, Takahashi K, Oshawa M (1988) Glutathione and metallothioneins as cellular defense against cadmium toxicity in culture Chinese hamster cells. Chem Biol Interact 65:1–14
Zhong Z, Trol W, Koenig KL, Frenkel K (1990) Carcinogenic sulfide salts of nickel and cadmium induce H2O2 formation by human polymorphonuclear leukocytes. Cancer Res 20:7564–7570
Sugiyama M (1994) Role of cellular antioxidants in metal-induced damage. Cell Biol Toxicol 10:1–22
Stohs SJ, Bagchi D (1995) Oxidative mechanisms in the toxicity of metal-ions. Free Radic Biol Med 18:321–336
Schwartz GG, Reis I. M (2000) Is cadmium a cause of human pancreatic cancer? Cancer Epidemiol Biomarkers Prev 9:139–145
Cronin JR (2000) Lycopene: the powerful antioxidant makes tomatoes red. Altern Complement Ther 6:92–94
Nguyen ML, Schwartz SJ (1999) Lycopene: chemical and biological properties. Food Technol 53:38–45
Bramley PM (2000) Is lycopene beneficial to human health? Phytochemistry 54:233–296
Sahin K, Ozercan R, Onderci M, Sahin N, Gursu MF, Khachik F, Sarkar FH, Munkarah A, Ali-Fehmi R, Kmak D, Kucuk O (2004) Lycopene supplementation prevents the development of spontaneous smooth muscle tumors of the oviduct in Japanese quail. Nutr Cancer 50:181–189
Di Mascio P, Kaiser S, Sies H (1989) Lycopene as the most efficient biological carotenoid singlet oxygen quencher. Arch Biochem Biophys 274:532–538
Conn PF, Schalch W, Truscott TG, Photochem J (1991) The singlet oxygen and carotenoid interaction. Photobiol Bull 11:41–47
Cantrell A, McGarvey DJ, Truscott TG, Rancan F, Bohm F (2003) Singlet oxygen quenching by dietary carotenoids in a model membrane environment. Arch Biochem Biophys 412:47–54
Pannala AS, Rice-Evans C, Sampson J, Singh S (1998) Interaction of peroxynitrite with carotenoids and tocopherols within low density lipoprotein. FEBS Lett 423:297–301
Woodall AA, Britton G, Jackson MJ (1997) Carotenoids and protection of phospholipids in solution or in liposomes against oxidation by peroxyl radicals: relationship between carotenoid structure and protective ability. Biochim Biophys Acta 1336:575–586
Agarwal S, Rao AV (1998) Tomato lycopene and low-density lipoprotein oxidation: a human dietary intervention study. Lipids 33:981–984
USAF (1990) Cadmium. In: Harry G (ed) Installation restoration program toxicology guide, vol. 5. Armstrong Aerospace Medical Research Laboratory, Wright Patterson AFB, OH, USA
Bradford MM (1976) A rapid and sensitive method fort ha quantization of microgram quantities of protein utilizing the principle of protein-dye binding. Ann Biochem 72:248–254
Yoshoiko T, Kawada K, Shimada T (1979) Lipid peroxidation in maternal and cord blood and protective mechanism against active-oxygen toxicity in the blood. Am J Obstet Gynecol 135:372–376
Cortas NK, Wakid NW (1990) Determination of inorganic nitrate in serum and urine by a kinetic cadmium-reduction method. Clin Chem 36:1440–1443
Sun Y, Oberley LW, Li Y (1988) A simple method for clinical assay of superoxide dismutase. Clin Chem 34:497–500
Aebi HE (1984) Catalase in vitro. Methods Enzymol 105:121
Prajda N, Weber G (1975) Malign transformation-linked imbalance: decreased XO activity in hepatomas. FEBS Lett 59:245–249
Rahel-Khazen R, Henriksen H, Bolann BJ, Ulvik RJ (2000) Validation of inductively coupled plasma atomic emission spectrometry technique (ICP-AES) for multi-element analysis of trace elements in human serum. Scand J Clin Lab Invest 38:765–772
Erdogan Z, Erdogan S, Celik S, Unlu A (2005) Effects of ascorbic acid on cadmium-induced oxidative stress and performance of broilers. Biol Trace Elem Res 104:19–32
Han XY, Xu ZR, Wang YZ, Huang QC (2006) Effect of cadmium on lipid peroxidation and activities of antioxidant enzymes in growing pigs. Biol Trace Elem Res 110:251–263
Waisberg M, Joseph P, Hale B, Beyersmann D (2003) Molecular and cellular mechanisms of cadmium carcinogenesis. Toxicology 192:95–117
Shen Y, Sangiah S (1995) Na, K-ATPase, glutathione, and hydroxyl free radicals in cadmium chloride-induced testicular toxicity in mice. Arch Environ Contam Toxicol 29:174–179
Sarkar S, Yadav P, Bhatnagar D (1997) Cadmium-induced lipid peroxidation and the antioxidant system in rat erythrocytes: the role of antioxidants. J Trace Elem Med Biol 11:8–13
Alvarez SM, Gomez NN, Scardapane L, Zirulnik F, Martinez D, Gimenez MS (2004) Morphological changes and oxidative stress in rat prostate exposed to a non-carcinogenic dose of cadmium. Toxicol Lett 153:365–376
Karahan I, Atessahin A, Yilmaz S, Ceribasi AO. Sakin F (2005) Protective effect of lycopene on gentamicin-induced oxidative stress and nephrotoxicity in rats. Toxicology 215:198–204
Bansal P, Gupta SK, Ojha SK, Nandave M, Mittal R, Kumari S, Arya DS (2006) Cardioprotective effect of lycopene in the experimental model of myocardial ischemia-reperfusion injury. Mol Cell Biochem 289:1–9
Schaeffer F, Stainer RY (1978) Glucose-6-phosphate dehydrogenase, kinetics and molecular properties. Arch Microbiol 116–119
Ursini F, Mairorino M, Valente M, Ferri L, Gregolin C (1982) Purification from pig liver of a protein which protects liposomes and biomembranes from peroxidative degradation and exhibits glutathione peroxidase activity on phophatidylcholine hydroperoxides. Biochim Biophys Acta 710:197
Stajn A, Zikic RV, Ognjanovic B, Saicic ZS, Pavlovic SZ, Kostic MM, Petrovic VM (1997) Effect of cadmium and selenium on the antioxidant defense system in rat kidneys. Comp Biochem Physiol 117C:167–172
Magos L, Webb M (1980) The interactions of selenium with cadmium and mercury. CRC Crit Rev Toxicol 8:1–42
Tian L, Lawrence DA (1996) Metal-induced modulation of nitric oxide production in vitro by murine macrophages: lead, nickel, and cobalt utilize different mechanisms. Toxicol Appl Pharmacol 141:540–547
Ramirez DC, Martinez LD, Marchevsky E, Gimenez MS (1999) Biphasic effect of cadmium in non-cytotoxic conditions on the secretion of nitric oxide from peritoneal macrophages. Toxicology 139:167–177
Hsiao G, Fong TH, Tzu NH, Lin KH, Chou DS, Sheu JR (2004) A potent antioxidant, lycopene, affords neuroprotection against microglia activation and focal cerebral ischemia in rats. In Vivo 18:351–356
Harstad EB, Klaassen CD (2002) Gadolinium chloride pre-treatment prevents cadmium chloride-induced liver damage in both wild-type and MT-null mice. Toxicol Appl Pharmacol 180:178–185
Chandra RK, McBean LD (1994) Zinc and immunity. Nutrition 10:79–80
Swiergosz-Kowalewska R, Holewa I (2007) Cadmium, zinc and iron interactions in the tissues of bank vole Clethrionomys glarelous after exposure to low and high doses of cadmium chloride. Biometals (in press)
Acknowledgement
This study was supported by the Commission of Scientific Research Projects of Mustafa Kemal University (project no: 05 L 0301).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Rencuzogullari, N., Erdogan, S. Oral Administration of Lycopene Reverses Cadmium-suppressed Body Weight Loss and Lipid Peroxidation in Rats. Biol Trace Elem Res 118, 175–183 (2007). https://doi.org/10.1007/s12011-007-0027-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12011-007-0027-7