Abstract
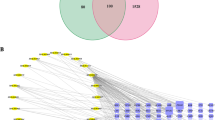
In China, a traditional Chinese medicine formulation called astragalus membranaceus (AM) has been utilised for more than 20 years to treat tumors with extraordinary effectiveness. The fundamental mechanisms, nevertheless, are still not well understood. The aim of this study is identifying its possible therapeutic targets and to evaluate the effects of AM in combination with a PARP inhibitor (olaparib) in the treatment of BRCA wild-type ovarian cancer. Significant genes were collected from Therapeutic Target Database and Database of Gene-Disease Associations. The components of AM were analyzed using the Traditional Chinese Medicine System Pharmacology (TCMSP) database to screen the active ingredients of AM based on their oral bioavailability and drug similarity index. In order to find intersection targets, Venn diagrams and STRING website diagrams were employed. STRING was also used to create a protein-protein interaction network. In order to create the ingredient-target network, Cytoscape 3.8.0 was used. DAVID database was utilized to carry out enrichment and pathway analyses. The binding ability of the active compounds of AM to the core targets of AM-OC was verified with molecular docking using AutoDock software. Experimental validations, including cell scratch, cell transwell, cloning experiment, were conducted to verify the effects of AM on OC cells. A total of 14 active ingredients of AM and 28 AM-OC-related targets were screened by network pharmacology analysis. The ten most significant Gene Ontology (GO) biological function analyses, as well as the 20 foremost Kyoto Encyclopedia of Genes and Genomes (KEGG) enrichment pathways were selected. Moreover, molecular docking results showed that bioactive compound (quercetin) demonstrated a good binding ability with tumor protein p53 (TP53), MYC, vascular endothelial growth factor A (VEGFA), phosphatase and tensin homolog (PTEN), AKT serine/threonine kinase 1 (AKT1) and cyclin D1 (CCND1) oncogenes. According to experimental methods, in vitro OC cell proliferation and migration appeared to be inhibited by quercetin, which also increased apoptosis. In addition, the combination with olaparib further enhanced the effect of quercetin on OC. Based on network pharmacology, molecular docking, and experimental validation, the combination of PARP inhibitor and quercetin enhanced the anti-proliferative activity in BRCA wild-type ovarian cancer cells, which supplies the theoretical groundwork for additional pharmacological investigation.


















Similar content being viewed by others
Data Availability
Not applicable.
References
Doubeni, C. A., Doubeni, A. R., & Myers, A. E. (2016). Diagnosis and management of ovarian cancer. American Family Physician, 93(11), 937–944.
Bonadio, R. C., & Estevez-Diz, M. (2021). Perspectives on PARP inhibitor combinations for ovarian cancer. Frontiers in Oncology, 11, 754524. https://doi.org/10.3389/fonc.2021.754524
Curtin, N. J., & Szabo, C. (2020). Poly(ADP-ribose) polymerase inhibition: Past, present and future. Nature Reviews Drug Discovery, 19(10), 711–736. https://doi.org/10.1038/s41573-020-0076-6
Javle, M., & Curtin, N. J. (2011). The role of PARP in DNA repair and its therapeutic exploitation. British Journal of Cancer, 105(8), 1114–1122. https://doi.org/10.1038/bjc.2011.382
Hodgson, D. R., Dougherty, B. A., Lai, Z., Fielding, A., Grinsted, L., Spencer, S., O’Connor, M. J., Ho, T. W., Robertson, J. D., Lanchbury, J. S., Timms, K. M., Gutin, A., Orr, M., Jones, H., Gilks, B., Womack, C., Gourley, C., Ledermann, J., & Barrett, J. C. (2018). Candidate biomarkers of PARP inhibitor sensitivity in ovarian cancer beyond the BRCA genes. British Journal of Cancer, 119(11), 1401–1409. https://doi.org/10.1038/s41416-018-0274-8
Rose, M., Burgess, J. T., O’Byrne, K., Richard, D. J., & Bolderson, E. (2020). PARP inhibitors: Clinical relevance, mechanisms of action and tumor resistance. Frontiers in Cell and Developmental Biology, 8, 564601. https://doi.org/10.3389/fcell.2020.564601
Pujade-Lauraine, E., Ledermann, J. A., Selle, F., Gebski, V., Penson, R. T., Oza, A. M., Korach, J., Huzarski, T., Poveda, A., Pignata, S., Friedlander, M., Colombo, N., Harter, P., Fujiwara, K., Ray-Coquard, I., Banerjee, S., Liu, J., Lowe, E. S., Bloomfield, R., & Pautier, P. (2017). Olaparib tablets as maintenance therapy in patients with platinum-sensitive, relapsed ovarian cancer and a BRCA1/2 mutation (SOLO2/ENGOT-Ov21): A double-blind, randomised, placebo-controlled, phase 3 trial. Lancet Oncology, 18(9), 1274–1284. https://doi.org/10.1016/S1470-2045(17)30469-2
Moore, K., Colombo, N., Scambia, G., Kim, B. G., Oaknin, A., Friedlander, M., Lisyanskaya, A., Floquet, A., Leary, A., Sonke, G. S., Gourley, C., Banerjee, S., Oza, A., González-Martín, A., Aghajanian, C., Bradley, W., Mathews, C., Liu, J., Lowe, E. S., … DiSilvestro, P. (2018). Maintenance olaparib in patients with newly diagnosed advanced ovarian cancer. New England Journal of Medicine, 379(26), 2495–2505. https://doi.org/10.1056/NEJMoa1810858
Xiang, Y., Guo, Z., Zhu, P., Chen, J., & Huang, Y. (2019). Traditional chinese medicine as a cancer treatment: Modern perspectives of ancient but advanced science. Cancer Medicine, 8(5), 1958–1975. https://doi.org/10.1002/cam4.2108
Liu, Y., Yang, S., Wang, K., Lu, J., Bao, X., Wang, R., Qiu, Y., Wang, T., & Yu, H. (2020). Cellular senescence and cancer: Focusing on traditional chinese medicine and natural products. Cell Proliferation, 53(10), e12894. https://doi.org/10.1111/cpr.12894
Wang, K., Chen, Q., Shao, Y., Yin, S., Liu, C., Liu, Y., Wang, R., Wang, T., Qiu, Y., & Yu, H. (2021). Anticancer activities of TCM and their active components against tumor metastasis. Biomedicine & Pharmacotherapy, 133, 111044. https://doi.org/10.1016/j.biopha.2020.111044
Ge, S., Xing, Q., Zhang, A., & Wang, Y. (2021). Effect of traditional chinese medicine (TCM) on survival, quality of life, and immune function in patients with ovarian carcinoma: A protocol for systematic review and meta analysis. Medicine, 100(2), e23904. https://doi.org/10.1097/MD.0000000000023904
Wang, R., Sun, Q., Wang, F., Liu, Y., Li, X., Chen, T., Wu, X., Tang, H., Zhou, M., Zhang, S., Xiao, Y., Huang, W., Wang, C. C., & Li, L. (2019). Efficacy and safety of Chinese herbal medicine on ovarian cancer after reduction surgery and adjuvant chemotherapy: A systematic review and Meta-analysis. Frontiers in Oncology, 9, 730. https://doi.org/10.3389/fonc.2019.00730
Zhang, Y. M., Zhang, Z. Y., & Wang, R. X. (2020). Protective mechanisms of quercetin against myocardial ischemia reperfusion injury. Frontiers in Physiology, 11, 956. https://doi.org/10.3389/fphys.2020.00956
Li, Y., Chen, M., Xu, Y., Yu, X., Xiong, T., Du, M., Sun, J., Liu, L., Tang, Y., & Yao, P. (2016). Iron-mediated lysosomal membrane permeabilization in ethanol-induced hepatic oxidative damage and apoptosis: protective effects of quercetin. Oxidative Medicine and Cellular Longevity, 2016, 4147610. https://doi.org/10.1155/2016/4147610
Ferreira, P. E., Lopes, C. R., Alves, A. M., Alves, ÉP., Linden, D. R., Zanoni, J. N., & Buttow, N. C. (2013). Diabetic neuropathy: An evaluation of the use of quercetin in the cecum of rats. World Journal of Gastroenterology, 19(38), 6416–6426. https://doi.org/10.3748/wjg.v19.i38.6416
Wang, R. E., Hunt, C. R., Chen, J., & Taylor, J. S. (2011). Biotinylated quercetin as an intrinsic photoaffinity proteomics probe for the identification of quercetin target proteins. Bioorganic & Medicinal Chemistry, 19(16), 4710–4720. https://doi.org/10.1016/j.bmc.2011.07.005
Wang, M., Chen, X., Yu, F., Zhang, L., Zhang, Y., & Chang, W. (2022). The targeting of noncoding RNAs by Quercetin in Cancer Prevention and Therapy. Oxidative Medicine and Cellular Longevity, 2022, 4330681. https://doi.org/10.1155/2022/4330681
Ru, J., Li, P., Wang, J., Zhou, W., Li, B., Huang, C., Li, P., Guo, Z., Tao, W., Yang, Y., Xu, X., Li, Y., Wang, Y., & Yang, L. (2014). TCMSP: A database of systems pharmacology for drug discovery from herbal medicines. Journal of Cheminformatics, 6, 13. https://doi.org/10.1186/1758-2946-6-13
Zhang, W., Chen, Y., Jiang, H., Yang, J., Wang, Q., Du, Y., & Xu, H. (2020). Integrated strategy for accurately screening biomarkers based on metabolomics coupled with network pharmacology. Talanta, 211, 120710. https://doi.org/10.1016/j.talanta.2020.120710
Pundir, S., Martin, M. J., & O’Donovan, C. (2017). UniProt protein knowledgebase. Methods in Molecular Biology (Clifton N J), 1558, 41–55. https://doi.org/10.1007/978-1-4939-6783-4_2
Rebhan, M., Chalifa-Caspi, V., Prilusky, J., & Lancet, D. (1997). GeneCards: Integrating information about genes, proteins and diseases. Trends in Genetics: TIG, 13(4), 163. https://doi.org/10.1016/s0168-9525(97)01103-7
Piñero, J., Bravo, À, Queralt-Rosinach, N., Gutiérrez-Sacristán, A., Deu-Pons, J., Centeno, E., García-García, J., Sanz, F., & Furlong, L. I. (2017). DisGeNET: A comprehensive platform integrating information on human disease-associated genes and variants. Nucleic Acids Research, 45(D1), D833–D839. https://doi.org/10.1093/nar/gkw943
Koscielny, G., An, P., Carvalho-Silva, D., Cham, J. A., Fumis, L., Gasparyan, R.,Hasan, S., Karamanis, N., Maguire, M., Papa, E., Pierleoni, A., Pignatelli, M., Platt,T., Rowland, F., Wankar, P., Bento, A. P., Burdett, T., Fabregat, A., Forbes, S.,Gaulton, A., … Dunham, I. (2017). Open Targets: a platform for therapeutic target identification and validation. Nucleic Acids Research, 45(D1), D985–D994. https://doi.org/10.1093/nar/gkw1055
Jia, A., Xu, L., & Wang, Y. (2021). Venn diagrams in bioinformatics. Briefings in Bioinformatics, 22(5), bbab108. https://doi.org/10.1093/bib/bbab108
Shannon, P., Markiel, A., Ozier, O., Baliga, N. S., Wang, J. T., Ramage, D., Amin, N., Schwikowski, B., & Ideker, T. (2003). Cytoscape: A software environment for integrated models of biomolecular interaction networks. Genome Research, 13(11), 2498–2504. https://doi.org/10.1101/gr.1239303
von Mering, C., Huynen, M., Jaeggi, D., Schmidt, S., Bork, P., & Snel, B. (2003). STRING: A database of predicted functional associations between proteins. Nucleic Acids Research, 31(1), 258–261. https://doi.org/10.1093/nar/gkg034
Dennis, G., Jr, Sherman, B. T., Hosack, D. A., Yang, J., Gao, W., Lane, H. C., & Lempicki, R. A. (2003). DAVID: Database for annotation, visualization, and integrated discovery. Genome Biology, 4(5), P3.
Kanehisa, M., Goto, S., Sato, Y., Furumichi, M., & Tanabe, M. (2012). KEGG for integration and interpretation of large-scale molecular data sets. Nucleic Acids Research, 40(Database issue), D109–D114. https://doi.org/10.1093/nar/gkr988
Pilipović, A., Mitrović, D., Obradović, S., & Poša, M. (2021). Docking-based analysis and modeling of the activity of bile acids and their synthetic analogues on large conductance Ca2 + activated K channels in smooth muscle cells. European Journal of Gynaecological Oncology, 25(23), 7501–7507. https://doi.org/10.26355/eurrev_202112_27449
Dong, Y., Zhao, Q., & Wang, Y. (2021). Network pharmacology-based investigation of potential targets of astragalus membranaceous-angelica sinensis compound acting on diabetic nephropathy. Scientific Reports, 11(1), 19496. https://doi.org/10.1038/s41598-021-98925-6
Yan, C., & Zou, X. (2015). Predicting peptide binding sites on protein surfaces by clustering chemical interactions. Journal of Computational Chemistry, 36(1), 49–61. https://doi.org/10.1002/jcc.23771
Pereira, J. C., Caffarena, E. R., & Dos Santos, C. N. (2016). Boosting docking-based virtual screening with deep learning. Journal of Chemical Information and Modeling, 56(12), 2495–2506. https://doi.org/10.1021/acs.jcim.6b00355
Xiao, S. M., Bai, R., & Zhang, X. Y. (2016). [Genomic research of traditional chinese medicines in vivo metabolism]. Zhongguo Zhong Yao Za Zhi, 41(22), 4103–4111. https://doi.org/10.4268/cjcmm20162204
Yang, Y., Zhang, Z., Li, S., Ye, X., Li, X., & He, K. (2014). Synergy effects of herb extracts: Pharmacokinetics and pharmacodynamic basis. Fitoterapia, 92, 133–147. https://doi.org/10.1016/j.fitote.2013.10.010
Wang, Z., Wan, H., Tong, X., He, Y., Yang, J., Zhang, L., Shao, C., Ding, Z., Wan, H., & Li, C. (2021). An integrative strategy for discovery of functional compound combination from traditional chinese medicine: Danhong Injection as a model. Biomedicine & Pharmacotherapy, 138, 111451. https://doi.org/10.1016/j.biopha.2021.111451
Hu, X. Q., Sun, Y., Lau, E., Zhao, M., & Su, S. B. (2016). Advances in synergistic combinations of chinese Herbal Medicine for the treatment of Cancer. Current Cancer Drug Targets, 16(4), 346–356. https://doi.org/10.2174/1568009616666151207105851
Qi, F., Zhao, L., Zhou, A., Zhang, B., Li, A., Wang, Z., & Han, J. (2015). The advantages of using traditional chinese medicine as an adjunctive therapy in the whole course of cancer treatment instead of only terminal stage of cancer. Bioscience Trends, 9(1), 16–34. https://doi.org/10.5582/bst.2015.01019
Zhu, X., Wu, Z., Cao, Y., Gao, R., Zhang, X., & Li, J. (2021). Efficacy and safety of TCM therapies combined with hyperthermic intraperitoneal chemotherapy for peritoneal metastasis of gastric cancer: A protocol for systematic review and meta-analysis. Medicine, 100(4), e24337. https://doi.org/10.1097/MD.0000000000024337
Xiao, Z., Chen, Z., Han, R., Lu, L., Li, Z., Lin, J., Hu, L., Huang, X., & Lin, L. (2021). Comprehensive TCM treatments combined with chemotherapy for advanced non-small cell lung cancer: A randomized, controlled trial. Medicine, 100(18), https://doi.org/10.1097/MD.0000000000025690
Zhang, G. L., Pan, M., Wang, Y. Z., Huang, J. X., Gu, G. S., Wang, Y., Wu, Q., Yao, L. T., Xie, H. R., & Hu, X. J. (2021). [Regulation effect of myeloid leukemia No.1 chinese Herb Medicine prescription combined with chemotherapy on Th17 cells in bone marrow of patients with Acute myeloid leukemia]. Zhongguo Shi Yan Xue Ye Xue Za Zhi, 29(2), 328–332. https://doi.org/10.19746/j.cnki.issn.1009-2137.2021.02.004
Li, S., Sun, Y., Huang, J., Wang, B., Gong, Y., Fang, Y., Liu, Y., Wang, S., Guo, Y., Wang, H., Xu, Z., & Guo, Y. (2020). Anti-tumor effects and mechanisms of Astragalus membranaceus (AM) and its specific immunopotentiation: Status and prospect. Journal of Ethnopharmacology, 258, 112797. https://doi.org/10.1016/j.jep.2020.112797
Tang, S. M., Deng, X. T., Zhou, J., Li, Q. P., Ge, X. X., & Miao, L. (2020). Pharmacological basis and new insights of quercetin action in respect to its anti-cancer effects. Biomedicine & Pharmacotherapy, 121, 109604. https://doi.org/10.1016/j.biopha.2019.109604
Vafadar, A., Shabaninejad, Z., Movahedpour, A., Fallahi, F., Taghavipour, M., Ghasemi, Y., Akbari, M., Shafiee, A., Hajighadimi, S., Moradizarmehri, S., Razi, E., Savardashtaki, A., & Mirzaei, H. (2020). Quercetin and cancer: New insights into its therapeutic effects on ovarian cancer cells. Cell & Bioscience, 10, 32. https://doi.org/10.1186/s13578-020-00397-0
Shafabakhsh, R., & Asemi, Z. (2019). Quercetin: A natural compound for ovarian cancer treatment. Journal of Ovarian Research, 12(1), 55. https://doi.org/10.1186/s13048-019-0530-4
Ren, M. X., Deng, X. H., Ai, F., Yuan, G. Y., & Song, H. Y. (2015). Effect of quercetin on the proliferation of the human ovarian cancer cell line SKOV-3 in vitro Experimental and Therapeutic Medicine, 10(2), 579–583. https://doi.org/10.3892/etm.2015.2536
Domcke, S., Sinha, R., Levine, D. A., Sander, C., & Schultz, N. (2013). Evaluating cell lines as tumour models by comparison of genomic profiles. Nature Communications, 4, 2126. https://doi.org/10.1038/ncomms3126
Zhong, Q., Hu, Z., Li, Q., Yi, T., Li, J., & Yang, H. (2019). Cyclin D1 silencing impairs DNA double strand break repair, sensitizes BRCA1 wildtype ovarian cancer cells to olaparib. Gynecologic Oncology, 152(1), 157–165. https://doi.org/10.1016/j.ygyno.2018.10.027
Li, H., Liu, Z. Y., Wu, N., Chen, Y. C., Cheng, Q., & Wang, J. (2020). PARP inhibitor resistance: The underlying mechanisms and clinical implications. Molecular Cancer, 19(1), 107. https://doi.org/10.1186/s12943-020-01227-0
Pillay, N., Tighe, A., Nelson, L., Littler, S., Coulson-Gilmer, C., Bah, N., Golder, A., Bakker, B., Spierings, D., James, D. I., Smith, K. M., Jordan, A. M., Morgan, R. D., Ogilvie, D. J., Foijer, F., Jackson, D. A., & Taylor, S. S. (2019). DNA replication vulnerabilities render ovarian Cancer cells sensitive to poly(ADP-Ribose) glycohydrolase inhibitors. Cancer Cell, 35(3), 519-533e8. https://doi.org/10.1016/j.ccell.2019.02.004
Lee, E. K., & Matulonis, U. A. (2020). Emerging drugs for the treatment of ovarian cancer: A focused review of PARP inhibitors. Expert Opinion on Emerging Drugs, 25(2), 165–188. https://doi.org/10.1080/14728214.2020.1773791
Boussios, S., Karihtala, P., Moschetta, M., Karathanasi, A., Sadauskaite, A., Rassy, E., & Pavlidis, N. (2019). Combined strategies with poly (ADP-Ribose) polymerase (PARP) inhibitors for the treatment of ovarian cancer: a literature review. Diagnostics (Basel Switzerland), 9(3), 87. https://doi.org/10.3390/diagnostics9030087
Slade, D. (2020). PARP and PARG inhibitors in cancer treatment. Genes & Development, 34(5–6), 360–394. https://doi.org/10.1101/gad.334516.119
Su, C., Haskins, A. H., Omata, C., Aizawa, Y., & Kato, T. A. (2017). PARP inhibition by Flavonoids Induced selective cell killing to BRCA2-Deficient cells. Pharmaceuticals (Basel Switzerland), 10(4), 80. https://doi.org/10.3390/ph10040080
Rather, R. A., & Bhagat, M. (2020). Quercetin as an innovative therapeutic tool for cancer chemoprevention: Molecular mechanisms and implications in human health. Cancer Medicine, 9(24), 9181–9192. https://doi.org/10.1002/cam4.1411
Reyes-Farias, M., & Carrasco-Pozo, C. (2019). The anti-cancer effect of quercetin: molecular implications in cancer metabolism. International Journal of Molecular Sciences, 20(13), 3177. https://doi.org/10.3390/ijms20133177
Author information
Authors and Affiliations
Contributions
The authors contributed equally.
Corresponding authors
Ethics declarations
Ethics Approval
All work has been done under the guidelines of Institutional Ethics Committee.
Consent to Participate
All authors have their consent to participate.
Consent for Publication
All authors have their consent to publish their work.
Competing Interests
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Liu, Y., Guo, Z., Lang, F. et al. Anticancer Effect of Active Component of Astragalus Membranaceus Combined with Olaparib on Ovarian Cancer Predicted by Network-Based Pharmacology. Appl Biochem Biotechnol 195, 6994–7020 (2023). https://doi.org/10.1007/s12010-023-04462-5
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12010-023-04462-5