Abstract
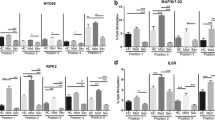
The pathogenesis of recurrent oral ulcers (ROU) is complex, with a long duration of illness and challenging to cure. According to traditional Chinese medicine (TCM),”heat accumulation in the heart-spleen” is one of the main causative factors. Jiaweidaochi powder (JWDCP) is based on the ancient Chinese medicine formula JWDCS, with the addition of Tongcao and gypsum and the removal of Mu Tong. It is generally used to treat “heat accumulation in the heart-spleen.” Previous studies have demonstrated that it effectively reduces recurrence rates and is anti-inflammatory in modulating immunity. The ROU rats’ model for JWDCP intervention treatment had been established, and histological tests revealed that JWDCP has a therapeutic effect on the pathological changes in the oral mucosa. In addition, the methylation levels of peripheral blood IFNG gene were detected by bisulfite sequencing PCR (BSP), and the methylation levels of the IFNG promoter region in the model group and each dose group were lower than those in the control group. However, no significant methylation differences were observed. Furthermore, the results of enzyme-linked immunosorbent assay (ELISA) and RNA quantitative polymerase chain reaction showed that JWDCP could reduce IFN-γ and IL-4 protein concentrations, with high GATA-3 mRNA production, T-bet mRNAproduction was upgraded, elevated IL-4 mRNA levels, and reduced IFN-γ mRNA levels after treatment (P < 0.001). The expression of transcription factor T-betmRNA and GATA-3 gene mRNA was accompanied by changes in IFN-γmRNA and IL-4mRNA, demonstrating that Th2 type differentiation in RAS suppresses the body’s immunity and that the imbalance of transcription factor expression further leads to Th1/Th2 drift. JWDCP is likely to reduce the protein concentration by regulating the imbalance of transcription factors and enhancing antioxidant capacity, thus achieving therapeutic effects. Treatment of recurrent oral ulcer models is not sufficient to reset IFNG methylation levels, correlating with the refractoriness of ROU, further confirming the complexity of epigenetic mechanisms and that epigenetic alterations in specific mediators may persist locally.






Similar content being viewed by others
Data Availability
Not applicable.
References
Toche, P., Salinas, L., Guzmán, M. M., Afani, S., & Jadue, A. (2007). Recurrent oral ulcer: Clinical characteristic and differential diagnosis. Revista Chilena de Infectologia: Organo Oficial de la Sociedad Chilena de Infectologia, 24(3), 215–219.
Sánchez, J., Conejero, C., & Conejero, R. (2020). Recurrent aphthous stomatitis. Actas Dermo-Sifiliográficas (English Edition), 111(6), 471–480.
Altenburg, A., El-Haj, N., Micheli, C., Puttkammer, M., Abdel-Naser, M. B., & Zouboulis, C. C. (2014). The treatment of chronic recurrent oral aphthous ulcers. Deutsches Ärzteblatt International, 111(40), 665.
Queiroz, S. I. M. L., Silva, M. VAd., Medeiros, A. MCd., Oliveira, PTd., Gurgel, BCd. V., & Silveira, É. JDd. (2018). Recurrent aphthous ulceration: an epidemiological study of etiological factors, treatment and differential diagnosis. Anais brasileiros de dermatologia, 93, 341–346.
Saikaly, S. K., Saikaly, T. S., & Saikaly, L. E. (2018). Recurrent aphthous ulceration: A review of potential causes and novel treatments. Journal of Dermatological Treatment, 29(6), 542–552.
Lewkowicz, N., Lewkowicz, P., Dzitko, K., Kur, B., Tarkowski, M., Kurnatowska, A., & Tchorzewski, H. (2008). Dysfunction of CD4+ CD25high T regulatory cells in patients with recurrent aphthous stomatitis. Journal of oral pathology & medicine, 37(8), 454–461.
Lewkowicz, N., Kur, B., Kurnatowska, A., Tchorzewski, H., & Lewkowicz, P. (2011). Expression of Th1/Th2/Th3/Th17-related genes in recurrent aphthous ulcers. Archivum immunologiae et therapiae experimentalis, 59(5), 399–406.
Guan, C., Wu, Y., Guo, H., & Li, J. (2014). Experimental study of the therapeutic effects of Dao chi San plus gypsum decoction with vitamin B12 on oral ulcer in rats. Journal of Clinical Stomatology, 30(07), 404–406.
Guo, H., & Guan, C. (2015). Modulation of Th1/Th2 balance in recurrent oral ulcer patients by adding the flavour of Jia Wei Dao Chi San. Chinese Remedies & Clinics, 15(12), 1745–1747.
Zhang, S., Crivello, A., Offenbacher, S., Moretti, A., Paquette, D. W., & Barros, S. P. (2010). Interferon-gamma promoter hypomethylation and increased expression in chronic periodontitis. Journal of clinical periodontology, 37(11), 953–961.
Szabo, S. J., Kim, S. T., Costa, G. L., Zhang, X., Fathman, C. G., & Glimcher, L. H. (2000). A novel transcription factor, T-bet, directs Th1 lineage commitment. Cell, 100(6), 655–669.
Skapenko, A., Leipe, J., Niesner, U., Devriendt, K., Beetz, R., Radbruch, A., . . . Schulze-Koops, H. (2004). GATA-3 in human T cell helper type 2 development. The Journal of experimental medicine, 199(3), 423–428.
Guan, C., Wu, Y., Guo, H., & Zhao, W. (2014). Effects of folic acid and jiaweidaochi powder on recurrent oral ulcer. Chinese Remedies & Clinics, 14(04), 426–428.
Kobayashi, T., Ishida, K., & Yoshie, H. (2016). Increased expression of interleukin-6 (IL-6) gene transcript in relation to IL-6 promoter hypomethylation in gingival tissue from patients with chronic periodontitis. Archives of oral biology, 69, 89–94.
Maggi, E., Biswas, P., Del Prete, G., Parronchi, P., Macchia, D., Simonelli, C., . . . Ricci, M. (1991). Accumulation of Th-2-like helper T cells in the conjunctiva of patients with vernal conjunctivitis. The Journal of immunology, 146(4), 1169–1174.
Ruterbusch, M., Pruner, K. B., Shehata, L., & Pepper, M. (2020). In vivo CD4+ T cell differentiation and function: Revisiting the Th1/Th2 paradigm. Annual review of immunology, 38, 705–725.
Gibney, E., & Nolan, C. (2010). Epigenetics and gene expression. Heredity, 105(1), 4–13.
Bird, A. (2002). DNA methylation patterns and epigenetic memory. Genes & development, 16(1), 6–21.
Saha, A., Dhir, A., Ranjan, A., Gupta, V., Bairwa, N., & Bamezai, R. (2005). Functional IFNG polymorphism in intron 1 in association with an increased risk to promote sporadic breast cancer. Immunogenetics, 57(3), 165–171.
Zheng, W.-P., & Flavell, R. A. (1997). The transcription factor GATA-3 is necessary and sufficient for Th2 cytokine gene expression in CD4 T cells. Cell, 89(4), 587–596.
Chang, S., Collins, P. L., & Aune, T. M. (2008). T-bet dependent removal of Sin3A-histone deacetylase complexes at the Ifng locus drives Th1 differentiation. The Journal of immunology, 181(12), 8372–8381.
Miller, S. A., Huang, A. C., Miazgowicz, M. M., Brassil, M. M., & Weinmann, A. S. (2008). Coordinated but physically separable interaction with H3K27-demethylase and H3K4-methyltransferase activities are required for T-box protein-mediated activation of developmental gene expression. Genes & development, 22(21), 2980–2993.
Jones, B., & Chen, J. (2006). Inhibition of IFN-γ transcription by site-specific methylation during T helper cell development. The EMBO journal, 25(11), 2443–2452.
Chang, S., & Aune, T. M. (2007). Dynamic changes in histone-methylation “marks” across the locus encoding interferon-γ during the differentiation of T helper type 2 cells. Nature immunology, 8(7), 723–731.
Romagnani, S. (1991). Human TH1 and TH2 subsets: Doubt no more. Immunology today, 12(8), 256–257.
Demeure, C. E., Yang, L. P., Byun, D. G., Ishihara, H., Vezzio, N., & Delespesse, G. (1995). Human naive CD4 T cells produce interleukin-4 at priming and acquire a Th2 phenotype upon repetitive stimulations in neutral conditions. European journal of immunology, 25(9), 2722–2725.
Esnault, S., Benbernou, N., Lavaud, F., Shin, H., Potron, G., Guenounou, M. J. C., & Immunology, E. (1996). Differential spontaneous expression of mRNA for IL-4, IL-10, IL-13, IL-2 and interferon-gamma (IFN-γ) in peripheral blood mononuclear cells (PBMC) from atopic patients. 103(1), 111–118.
Lighvani, A. A., Frucht, D. M., Jankovic, D., Yamane, H., Aliberti, J., Hissong, B. D., . . . Paul, W. E. J. P. o. t. N. A. o. S. (2001). T-bet is rapidly induced by interferon-γ in lymphoid and myeloid cells. 98(26), 15137–15142.
Ślebioda, Z., Szponar, E., Kowalska, A. J. A. i. d., & Alergologii, a. P. D. I. (2013). Recurrent aphthous stomatitis: Genetic aspects of etiology. 30(2), 96.
Williams, C. L., Schilling, M. M., Cho, S. H., Lee, K., Wei, M., & Boothby, M. J. T. J. O. I. (2013). STAT4 and T-bet are required for the plasticity of IFN-γ expression across Th2 ontogeny and influence changes in Ifng promoter DNA methylation. The Journal of Immunology, 191(2), 678–687.
Nares, S., Moutsopoulos, N. M., Angelov, N., Rangel, Z. G., Munson, P. J., Sinha, N., & Wahl, S. M. J. T. A. j. o. p. (2009). Rapid myeloid cell transcriptional and proteomic responses to periodontopathogenic Porphyromonas gingivalis. 174(4), 1400–1414.
Viana, M. B., Cardoso, F. P., Diniz, M. G., Costa, F. O., da Costa, J. E., Gomez, R. S., & Moreira, P. R. J. I. (2011). Methylation pattern of IFN-γ and IL-10 genes in periodontal tissues. Immunobiology, 216(8), 936–941.
Stefani, F. A., Viana, M. B., Dupim, A. C., Brito, J. A. R., Gomez, R. S., da Costa, J. E., & Moreira, P. R. J. I. (2013). Expression, polymorphism and methylation pattern of interleukin-6 in periodontal tissues. Immunobiology, 218(7), 1012–1017.
Asa'ad, F., Bollati, V., Pagni, G., Castilho, R. M., Rossi, E., Pomingi, F., . . . Rasperini, G. J. J. o. C. P. (2017). Evaluation of DNA methylation of inflammatory genes following treatment of chronic periodontitis: A pilot case–control study. 44(9), 905–914.
Funding
This work was supported by General Project of Shanxi Province Key R&D Program (No.201603D321044) and Postgraduate Innovation and Entrepreneurship Program of Shanxi University of Traditional Chinese Medicine (No.2021CX042).
Author information
Authors and Affiliations
Contributions
The authors contributed equally.
Corresponding author
Ethics declarations
Ethics Approval
All work has been done under the guidelines of Institutional Ethics Committee.
Competing Interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Hu, Y., Guo, H., He, L. et al. The Correlation Between IFNG Gene Methylation and Th1|Th2 Cell Balance in ROU and the Interventional Study of Jiaweidaochi Powder. Appl Biochem Biotechnol 195, 6737–6751 (2023). https://doi.org/10.1007/s12010-023-04417-w
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12010-023-04417-w