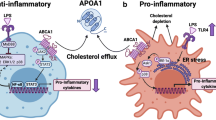
Abstract
For several strategies formulated to prevent atherosclerosis, Apolipoprotein A1 Milano (ApoA1M) remains a prime target. ApoA1M has been reported to have greater efficiency in reducing the incidence of coronary artery diseases. Furthermore, recombinant ApoA1M based mimetic peptide exhibits comparatively greater atheroprotective potential, offers a hope in reducing the burden of atherosclerosis in in vivo model system. The aim of this review is to emphasize on some of the observed ApoA1M structural and functional effects that are clinically and therapeutically meaningful that might converge on the basic role of ApoA1M in reducing the chances of glycation assisted ailments in diabetes. We also hypothesize that the nonenzymatic glycation prone arginine amino acid of ApoA1 gets replaced with cysteine residue and the rate of ApoA1 glycation may decrease due to change substitution of amino acid. Therefore, to circumvent the effect of ApoA1M glycation, the related mechanism should be explored at the cellular and functional levels, especially in respective experimental disease model in vivo.

Similar content being viewed by others
Data Availability
The data that support this study are available from the corresponding author, upon reasonable request.
References
Ahmad, A. (2017). A cure for coronary artery disease. J. Diabetes Metab, 8, 769.
Ahmad, S., Alouffi, S., Khan, S., Khan, M., Akasha, R., Ashraf, J. M. Khan M. Y. (2022). Physicochemical characterization of in vitro LDL glycation and its inhibition by ellagic acid (EA) An in vivo approach to inhibit diabetes in experimental animals. BioMed Research International 2022.
Ahmad, S., Khan, M. Y., Rafi, Z., Khan, H., Siddiqui, Z., Rehman, S. and Khan, M. S. (2018, April). Oxidation, glycation and glycoxidation—The vicious cycle and lung cancer. In Seminars in cancer biology (Vol. 49, pp. 29-36). Academic Press.
Alexander, E. T., Tanaka, M., Kono, M., Saito, H., Rader, D. J., & Phillips, M. C. (2009a). Structural and functional consequences of the Milano mutation (R173C) in human apolipoprotein AI. Journal of Lipid Research, 50(7), 1409–1419.
Alexander, E. T., Weibel, G. L., Joshi, M. R., Vedhachalam, C., de la Llera-Moya, M., Rothblat, G. H., & Rader, D. J. (2009b). Macrophage reverse cholesterol transport in mice expressing ApoA-I Milano. Arteriosclerosis, Thrombosis, and Vascular Biology, 29(10), 1496–1501.
Baidanoff, F. M., Trebucq, L. L., Plano, S. A., Eaton, P., Golombek, D. A., & andamp; Chiesa, J. J. (2022). Cysteine oxidation promotes dimerization/oligomerization of circadian protein period2. Biomolecules, 12(7), 892.
Bain, C. C., & Mowat, A. M. (2014). Macrophages in intestinal homeostasis and inflammation. Immunological reviews, 260(1), 102–117.
Baker, J. V., Neuhaus, J., Duprez, D., Cooper, D. A., Hoy, J., Kuller, L., & Neaton, J. D. (2011). Inflammation predicts changes in high-density lipoprotein particles and apolipoprotein A1 following initiation of antiretroviral therapy. AIDS (London, England), 25(17), 2133.
Bedi, S., Morris, J., Shah, A., Hart, R. C., Jerome, W. G., Aller, S. G Davidson, W. S. (2022).Conformational flexibility of apolipoprotein AI amino-and carboxy-termini is necessary for lipid bindingbut not cholesterol efflux. Journal of Lipid Research 63(3).
Bhale, A. S., & Venkataraman, K. (2022). Leveraging knowledge of HDLs major protein ApoA1 Structure, function mutations, and potential therapeutics. Biomedicine and Pharmacotherapy, 154, 113634.
Bielicki, J. K., & Oda, M. N. (2002). Apolipoprotein A-IMilano and apolipoprotein A-IParis exhibit an antioxidant activity distinct from that of wild-type apolipoprotein AI. Biochemistry, 41(6), 2089–2096.
Borthwick, F., Warnakula, S., Mangat, R., Uwiera, R. R., Russell, J. C., Kelly, S. E., & Proctor, S. D. (2012). ApoA-1 infusion reduces arterial cholesterol and myocardial lesions in a rat model of cardiac dysfunction and insulin resistance. Atherosclerosis, 222(2), 402–408.
Brewer, H. B., Jr. (2004). High-density lipoproteins: a new potential therapeutic target for the prevention of cardiovascular disease. Arteriosclerosis, Thrombosis, and Vascular Biology, 24(3), 387–391.
Brown, B. E., Nobecourt, E., Zeng, J., Jenkins, A. J., Rye, K. A., & Davies, M. J. (2013). Apolipoprotein AI glycation by glucose and reactive aldehydes alters phospholipid affinity but not cholesterol export from lipid-laden macrophages. PLoS One, 8(5), e65430.
Cedó, L., García-León, A., Baila-Rueda, L., Santos, D., Grijalva, V., Martínez-Cignoni, M. R., & Blanco-Vaca, F. (2016). ApoA-I mimetic administration, but not increased apoA-I-containing HDL, inhibits tumour growth in a mouse model of inherited breast cancer. Scientific reports, 6(1), 1–11.
Chien, K. L., Chen, W. J., Hsu, H. C., Su, T. C., Chen, M. F., & Lee, Y. T. (2006). Segregation analysis of apolipoprotein A1 levels in families of adolescents: A community-based study in Taiwan. BMC genetics, 7(1), 1–9.
Chiesa, G., & Sirtori, C. R. (2003). Apolipoprotein A-IMilano: Current perspectives. Current opinion in lipidology, 14(2), 159–163.
Cho, K. H., Durbin, D. M., & Jonas, A. (2001). Role of individual amino acids of apolipoprotein AI in the activation of lecithin: Cholesterol acyltransferase and in HDL rearrangements. Journal of Lipid Research, 42(3), 379–389.
Chyu, K.Y., & Shah, P. K. (2015). HDL/ApoA-1 infusion and ApoA-1 gene therapy inatherosclerosis. Frontiers In Pharmacology, 6, 187.
Ciesielska, A., Matyjek, M., & Kwiatkowska, K. (2021). TLR4 and CD14 trafficking and its influence onLPS-induced pro-inflammatory signaling. Cellular and Molecular Life Sciences, 78(4), 1233–1261.
Cochran, B. J., Ong, K. L., Manandhar, B., & Rye, K. A. (2021). APOA1: a protein with multiple therapeutic functions. Current Atherosclerosis Reports, 23(3), 1–10.
Das, M., & Gursky, O. (2015). Amyloid-forming properties of human apolipoproteins Sequence analyses and structural insights. Lipids in Protein Misfolding, 175–211.
De Nardo, D., Labzin, L. I., Kono, H., Seki, R., Schmidt, S. V., Beyer, M., & Latz, E. (2014). High-density lipoprotein mediates anti-inflammatory reprogramming of macrophages via the transcriptional regulator ATF3. Nature Immunology, 15(2), 152–160.
Dong, H., Zhang, Y., Hu, P., Wang, J., & Lu, N. (2022). Serum apolipoprotein A1 rather than apolipoprotein B is associated with hypertension prevalence in Chinese people with coronary artery disease. Blood Pressure Monitoring, 27(2), 121–127.
Ducroux, C., Desilles, J. P., Mawhin, M. A., Delbosc, S., Ho-Tin-Noé, B., Ollivier, V., & Amarenco, P. (2020). Protective effect of ApoA1 (apolipoprotein A1)-Milano in a rat model of large vesselocclusion stroke. Stroke, 51(6), 1886–1890.
Ducroux, C., Desilles, J. P., Mawhin, M. A., Delbosc, S., Ho-Tin-Noé, B., Ollivier, V., & Amarenco, P. (2020). Protective effect of ApoA1 (apolipoprotein A1)-Milano in a rat model of large vessel occlusion stroke. Stroke, 51(6), 1886–1890.
Eren, E., Yilmaz, N., & Aydin, O. (2012). High density lipoprotein and it’s dysfunction. The Open Biochemistry Journal, 6, 78.
Favari, E., Gomaraschi, M., Zanotti, I., Bernini, F., Lee-Rueckert, M., Kovanen, P. T., & Calabresi, L. (2007). A unique protease-sensitive high density lipoprotein particle containing the apolipoprotein A-IMilano dimer effectively promotes ATP-binding cassette A1-mediated cell cholesterol efflux. Journal of Biological Chemistry, 282(8), 5125–5132.
Fernández-de Retana, S., Montañola, A., Marazuela, P., De La Cuesta, M., Batlle, A., Fatar, M., & Hernández-Guillamon, M. (2017). Intravenous treatment with human recombinant ApoA-I Milano reduces beta amyloid cerebral deposition in the APP23-transgenic mouse model of Alzheimer’s disease. Neurobiology of Aging, 60, 116–128.
Franceschini, G., Sirtori, C. R., Capurso, A. 2., Weisgraber, K. H., & Mahley, R. W. (1980). A-IMilano apoprotein. Decreased high density lipoprotein cholesterol levels with significant lipoprotein modifications and without clinical atherosclerosis in an Italian family. The Journal of clinical investigation, 66(5), 892–900.
Frondelius, K., Borg, M., Ericson, U., Borné, Y., Melander, O., & Sonestedt, E. (2017). Lifestyle and dietary determinants of serum apolipoprotein A1 and apolipoprotein B concentrations: cross-sectional analyses within a Swedish cohort of 24,984 individuals. Nutrients, 9(3), 211.
Gilchrist, M., Thorsson, V., Li, B., Rust, A. G., Korb, M., Kennedy, K., & Aderem, A. (2006). Systems biology approaches identify ATF3 as a negative regulator of Toll-like receptor 4. Nature, 441(7090), 173–178.
Giudice, R., Nilsson, O., Domingo-Espín, J., & Lagerstedt, J. O. (2017). Synchrotron radiation circular dichroism spectroscopy reveals structural divergences in HDL-bound apoA-I variants. Scientific Reports, 7(1), 1–9.
Godfrey, L., Yamada-Fowler, N., Smith, J., Thornalley, P. J., & Rabbani, N. (2014). Arginine-directed glycation and decreased HDL plasma concentration and functionality. Nutrition and diabetes, 4(9), e134–e134.
Gol, S., Pena, R. N., Rothschild, M. F., & TorEstany, J., M. (2018). A polymorphism in the fatty acid desaturase-2 gene is associated with the arachidonic acid metabolism in pigs. Scientific Reports, 8(1), 1–9.
Gursky, O., Jones, M. K., Mei, X., Segrest, J. P., & Atkinson, D. (2013). Structural basis for distinctfunctions of the naturally occurring Cys mutants of human apolipoprotein AI [S]. Journal of Lipid Research, 54(12), 3244–3257.
Hai, T., Wolford, C. C., & Chang, Y. S. (2010). ATF3, a hub of the cellular adaptive-response network, in the pathogenesis of diseases: Is modulation of inflammation a unifying component? Gene Expression The Journal of Liver Research, 15(1), 1–11.
Kajani, S., Curley, S., & McGillicuddy, F. C. (2018). Unravelling HDL—looking beyond the cholesterolsurface to the quality within. International Journal of Molecular Sciences, 19(7), 1971.
Kallend, D. G., Reijers, J. A. A., Bellibas, S. E., Bobillier, A., Kempen, H., Burggraaf, J., & Wijngaard, P. L. J. (2016). A single infusion of MDCO-216 (ApoA-1 Milano/POPC) increases ABCA1-mediated cholesterol efflux and pre-beta 1 HDL in healthy volunteers and patients with stable coronary artery disease. European Heart Journal-Cardiovascular Pharmacotherapy, 2(1), 23–29.
Kashyap, S. R., Osme, A., Ilchenko, S., Golizeh, M., Lee, K., Wang, S., & Kasumov, T. (2018). Glycation reduces the stability of ApoAI and increases HDL dysfunction in diet-controlled type 2 diabetes. The Journal of Clinical Endocrinology and Metabolism, 103(2), 388–396.
Khaidakov, M., and Mehta, J. L. (2012). Oxidized LDL triggers pro-oncogenic signaling in human breast mammary epithelial cells partly via stimulation of MiR-21.
Khan, M. Y., Alouffi, S., & Ahmad, S. (2018). Immunochemical studies on native and glycated LDL–An approach to uncover the structural perturbations. International Journal of Biological Macromolecules, 115, 287–299.
Klancic, T., Woodward, L., Hofmann, S. M., & Fisher, E. A. (2016). High density lipoprotein and metabolic disease: Potential benefits of restoring its functional properties. Molecular Metabolism, 5(5), 321–327.
Kosmas, C. E., Martinez, I., Sourlas, A., Bouza, K. V., Campos, F. N., Torres, V. Guzman, E.(2018). High-density lipoprotein (HDL) functionality and its relevance to atherosclerotic cardiovasculardisease. Drugs in context, 7.
Kunnen, S., & Van Eck, M. (2012). Lecithin: Cholesterol acyltransferase: old friend or foe in atherosclerosis? Journal of Lipid Research, 53(9), 1783–1799.
Lai, P. F., Cheng, C. F., Lin, H., Tseng, T. L., Chen, H. H., and Chen, S. H. (2013). ATF3 protects against LPS-induced inflammation in mice via inhibiting HMGB1 expression. Evidence-Based Complementary and Alternative Medicine, 2013.
Ma’arfi, F., Khan, M. Y., Fatima, J. E., Fatima, U., Mir, S. S. Yusuf, M. A. (2019). Structural insights into human apolipoprotein A1 variants and their correlation with amyloidogenic propensities: an in silico approach. Biochemical and Cellular Archives, 19(2), 3539–3548.
Ma’arfi, F., Prasad, S. C., Fatima, J., Khan, M. Y., Mir, S. S., & Yusuf, M. A. (2020). Probing the structure–function relationship and amyloidogenic propensities in natural variants of apolipoprotein AI. Biochemistry and Biophysics Reports, 24, 100815.
Madahian, S., Daniel Navab, K., Pourtabatabaei, N., Seyedali, S., Safar, S., Vazirian, S., & Hough, G. (2014). Inflammation, high density lipoprotein and endothelium. Current Medicinal Chemistry, 21(25), 2902–2909.
Matsunaga, A. (2014). Apolipoprotein AI mutations and clinical evaluation. In The HDL Handbook (pp. 9-35). Academic Press.
Mei, X., Liu, M., Herscovitz, H., & Atkinson, D. (2016). Probing the C-terminal domain of lipid-free apoA-I demonstrates the vital role of the H10B sequence repeat in HDL formation. Journal of Lipid Research, 57(8), 1507–1517.
Mineo, C., & Shaul, P. W. (2012). Novel biological functions of high-density lipoprotein cholesterol. Circulation Research, 111(8), 1079–1090.
Mineo, C., Deguchi, H., Griffin, J. H., & Shaul, P. W. (2006). Endothelial and antithrombotic actions of HDL. Circulation research, 98(11), 1352–1364.
Morgantini, C., Imaizumi, S., Grijalva, V., Navab, M., Fogelman, A. M., & Reddy, S. T. (2010). Apolipoprotein AI mimetic peptides prevent atherosclerosis development and reduce plaque inflammation in a murine model of diabetes. Diabetes, 59(12), 3223–3228.
Musgrave, I., Pirie-Shepherd, S., andamp; Theobald, D. (2003). Apolipoprotein AI mutations and information. http://www.talkorigins.org/faqs/information/apolipoprotein.html
Natarajan, P., Ray, K. K., & Cannon, C. P. (2010). High-density lipoprotein and coronary heart disease: Current and future therapies. Journal of the American College of Cardiology, 55(13), 1283–1299.
Nguyen, C. T., Kim, E. H., Luong, T. T., Pyo, S., & Rhee, D. K. (2015). TLR4 mediates pneumolysin-induced ATF3 expression through the JNK/p38 pathway in Streptococcus pneumoniae-infected RAW 264.7 cells. Molecules and Cells, 38(1), 58.
Nissen, S. E., Tsunoda, T., Tuzcu, E. M., Schoenhagen, P., Cooper, C. J., Yasin, M., & Kerensky, R. (2003). Effect of recombinant ApoA-I Milano on coronary atherosclerosis in patients with acute coronary syndromes: A randomized controlled trial. Jama, 290(17), 2292–2300.
Oda, MN. (2017). Lipid-free apoA-I structure-Origins of model diversity. Biochimica et Biophysica Acta (BBA)-Molecular and Cell Biology of Lipids, 1862(2), 221-233.
O’neill, L. A., Sheedy, F. J. McCoy, C. E. (2011). MicroRNAs: The fine-tuners of Toll-like receptor signalling. Nature Reviews Immunology, 11(3), 163–175.
Petrlova, J., Dalla-Riva, J., Mörgelin, M., Lindahl, M., Krupinska, E., Stenkula, K. G., & Lagerstedt, J. O. (2014). Secondary structure changes in ApoA-I Milano (R173C) are not accompanied by a decrease in protein stability or solubility. PloS one, 9(4), e96150.
Pollard, R. D., Fulp, B., Sorci-Thomas, M. G., & Thomas, M. J. (2016). High-density lipoprotein biogenesis: defining the domains involved in human apolipoprotein AI lipidation. Biochemistry, 55(35), 4971–4981.
Purushothaman, M., Puru, P., Cimmino, G., & Badiom, J. J. (2010). Antioxidant and anti-inflammatory properties of apo A-I Milano. The FASEB Journal, 24, 589–18.
Ramella, N. A., Rimoldi, O. J., Prieto, E. D., Schinella, G. R., Sanchez, S. A., Jaureguiberry, M. S., & Tricerri, M. A. (2011). Human apolipoprotein AI-derived amyloid: its association with atherosclerosis. PLoS One, 6(7), e22532.
Romano, G., Reggi, S., Kutryb-Zajac, B., Facoetti, A., Chisci, E., Pettinato, M., & Giovannoni, R. (2018). APOA-1Milano muteins, orally delivered via genetically modified rice, show anti-atherogenicand anti-inflammatory properties in vitro and in Apoe−/− atherosclerotic mice. International Journal of Cardiology, 271, 233–239.
Shah, P. K., Yano, J., Reyes, O., Chyu, K. Y., Kaul, S., Bisgaier, C. L., ... & Cercek, B. (2001). High-dose recombinant apolipoprotein A-IMilano mobilizes tissue cholesterol and rapidly reduces plaque lipid and macrophage content in apolipoprotein E–deficient mice: potential implications for acute plaque stabilization. Circulation, 103(25), 3047–3050.
Sherman, C. B., Peterson, S. J., & Frishman, W. H. (2010). Apolipoprotein AI mimetic peptides: A potential new therapy for the prevention of atherosclerosis. Cardiology in Review, 18(3), 141–147.
Sirtori, C. R., Calabresi, L., Franceschini, G., Baldassarre, D., Amato, M., Johansson, J., & Agabiti-Rosei, E. (2001). Cardiovascular status of carriers of the apolipoprotein A-IMilano mutant: The Limonesul Garda study. Circulation, 103(15), 1949–1954.
Sirtori, C. R., Muiesan, M. L., Salvetti, M., Agabiti-Rosei, E., Calabresi, L., Zulli, R., …, & Johansson, J. (2012). Cardiovascular status of carriers of the apolipoprotein A-I Milano mutant. Circulation, 103(15), 1949–1954.
Smith, J. D. (2010). Apolipoprotein AI and its mimetics for the treatment of atherosclerosis. Currentopinion in investigational drugs (London, England: 2000), 11(9), 989.
Stephaen Nicholls. (2016). MILANO-PILOT brings ApoA-1 Milano hopes to an end. Once-promising HDL-targeted therapy fails to show plaque regression. Heart andamp; Vascular Research..
Strang, A. C., Knetsch, M. L., Koole, L. H., de Winter, R. J., van der Wal, A. C., de Vries, C. J., & Rotmans, J. I. (2015). Effect of anti-ApoA-I antibody-coating of stents on neointima formation in a rabbit balloon-injury model. PloS one, 10(3), e0122836.
Su, F., Kozak, K. R., Imaizumi, S., Gao, F., Amneus, M. W., Grijalva, V., & Farias-Eisner, R. (2010). Apolipoprotein AI (apoA-I) and apoA-I mimetic peptides inhibit tumor development in a mouse model of ovarian cancer. Proceedings of the National Academy of Sciences, 107(46), 19997–20002.
Trakaki, A., & Marsche, G. (2021). Current understanding of the immunomodulatory activities of high-density lipoproteins. Biomedicines, 9(6), 58.
Vallejo-Vaz, AJ. Ray, KK. (2016). Promoting high-density lipoprotein function via intravenousinfusion: the rebirth of apoA-I Milano?. European Heart Journal-CardiovascularPharmacotherapy 2(1) 30-31
Vitali, C., & Cuchel, M. (2017). Therapies targeting HDLc levels and HDL function. In The HDL handbook (pp. 257–300). Academic Press.
Wang, L., Tian, F., Arias, A., Yang, M., Sharifi, B. G., & Shah, P. K. (2016). Comparative effects of diet-induced lipid lowering versus lipid lowering along with apo AI Milano gene therapy on regression of atherosclerosis. Journal of Cardiovascular Pharmacology and Therapeutics, 21(3), 320–328.
Wang, Y., Wang, Y., Jia, S., Dong, Q., Chen, Y., Lu, S. Hou, L. (2017). Effect of lipid-bound apolipoprotein AI cysteine mutant on ATF3 in RAW264 7 cells. Bioscience Reports, 37(1).
Wang, Y., Zhu, X., Wu, G., Shen, L., & Chen, B. (2008). Effect of lipid-bound apoA-I cysteine mutants on lipopolysaccharide-induced endotoxemia in mices. Journal of Lipid Research, 49(8), 1640–1645.
Witkowski, A., Chan, G. K., Boatz, J. C., Li, N. J., Inoue, A. P., Wong, J. C., & Cavigiolio, G. (2018). Methionine oxidized apolipoprotein AI at the crossroads of HDL biogenesis and amyloid formation. The FASEB Journal, 32(6), 3149.
Witkowski, A., Chan, G. K., Boatz, J. C., Li, N. J., Inoue, A. P., Wong, J. C., & Cavigiolio, G. (2018). Methionine oxidized apolipoprotein AI at the crossroads of HDL biogenesis and amyloid formation. The FASEB Journal, 32(6), 3149.
Zhang, X., Wang, L., & Chen, B. (2015). Recombinant HDL (Milano) protects endotoxin-challenged rats from multiple organ injury and dysfunction. Biological Chemistry, 396(1), 53–60.
Zhao, X., Yu, Y., Xu, W., Dong, L., Wang, Y., Gao, B., …, & Zhang, W. (2016). Apolipoprotein A1-unique peptide as a diagnostic biomarker for acute ischemic stroke. International Journal of Molecular Sciences, 17(4), 458.
Funding
This study is supported by the intramural fund in the form of seed money project (Ref: UU/DRI/SM/2021/08) of Uttaranchal University.
Author information
Authors and Affiliations
Contributions
M.Y. Khan and F.M. contributed hand to hand in this work. Writing initial idea, original draft, and editing by M.Y. Khan F. Maarfi and S. Rehman. A.S. Abdullah organized the structure of the review. S. Aswal and M.F.A. Ahmad done figure and content preparation. Review and editing were performed M.A. Yusuf and A.S., and overall supervision was performed by M.Y.K. There were no disagreements between authors.
Corresponding author
Ethics declarations
Ethical Approval
Not applicable
Consent to Participate
Not applicable.
Consent to Publish
Not applicable.
Conflict of Interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Maarfi, F., Yusuf, M.A., Ahmad, M.F.A. et al. A Study on Multiple Facets of Apolipoprotein A1 Milano. Appl Biochem Biotechnol 195, 4653–4672 (2023). https://doi.org/10.1007/s12010-023-04330-2
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12010-023-04330-2