Abstract
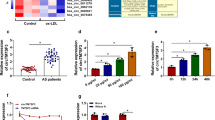
Atherosclerosis (AS) is one of the most common and important vascular diseases. It is believed that the abnormal expression of circular RNAs (circRNAs) plays an important role in AS. Hence, we investigate the function and mechanism of circ-C16orf62 in AS development.
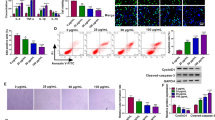
In this study, oxidized low-density lipoprotein (ox-LDL)-treated human macrophages (THP-1) were used as pathological conditions of AS in vitro. The expression of circ-C16orf62, miR-377 and Ras-related protein (RAB22A) mRNA was detected by real-time quantitative polymerase chain reaction (RT-qPCR) or western blot. Cell viability or cell apoptosis was assessed by cell counting kit-8 (CCK-8) assay or flow cytometry assay. The releases of proinflammatory factors were investigated using enzyme-linked immunosorbent assay (ELISA). The production of malondialdehyde (MDA) and superoxide dismutase (SOD) was examined to assess oxidative stress. Total cholesterol (T-CHO) level was detected, and cholesterol efflux level was tested using a liquid scintillation counter. The putative relationship between miR-377 and circ-C16orf62 or RAB22A was verified by dual-luciferase reporter assay and RNA immunoprecipitation (RIP) assay.
circ-C16orf62 expression was elevated in AS serum samples and ox-LDL-treated THP-1 cells. Apoptosis, inflammation, oxidative stress and cholesterol accumulation induced by ox-LDL were suppressed by circ-C16orf62 knockdown. Circ-C16orf62 could bind to miR-377 and thus increased the expression level of RAB22A. Rescued experiments showed that circ-C16orf62 knockdown alleviated ox-LDL-induced THP-1 cell injuries by increasing miR-377 expression, and miR-377 overexpression lessened ox-LDL-induced THP-1 cell injuries by degrading RAB22A level.
In conclusion, circ-C16orf62 played a crucial role in the regulation of apoptosis, inflammation, oxidative stress and cholesterol accumulation in ox-LDL-treated human macrophages via mediating the miR-377/RAB22A axis, hinting that circ-C16orf62 might be involved in AS progression.









Similar content being viewed by others
Data Availability
The datasets used and analyzed during the current study are available from the corresponding author on reasonable request.
References
Frohlich, J., & Al-Sarraf, A. (2013). Cardiovascular risk and atherosclerosis prevention. Cardiovascular Pathology : The Official Journal Of The Society For Cardiovascular Pathology, 22, 16–18.
Chistiakov, D. A., Bobryshev, Y. V., & Orekhov, A. N. (2016). Macrophage-mediated cholesterol handling in atherosclerosis. Journal Of Cellular And Molecular Medicine, 20, 17–28.
Shah, P., Bajaj, S., Virk, H., Bikkina, M., & Shamoon, F. (2015). Rapid Progression of Coronary Atherosclerosis: A Review. Thrombosis 2015, 634983
Linton, M. F., Babaev, V. R., Huang, J., Linton, E. F., Tao, H., & Yancey, P. G. (2016). Macrophage apoptosis and efferocytosis in the pathogenesis of atherosclerosis. Circulation Journal, 80, 2259–2268.
Wang, G., Li, Y., Liu, Z., Ma, X., Li, M., Lu, Q., et al. (2020). Circular RNA circ_0124644 exacerbates the ox-LDL-induced endothelial injury in human vascular endothelial cells through regulating PAPP-A by acting as a sponge of miR-149-5p. Molecular And Cellular Biochemistry, 471, 51–61.
Yang, L., Yang, F., Zhao, H., Wang, M., & Zhang, Y. (2019). Circular RNA circCHFR facilitates the Proliferation and Migration of Vascular smooth muscle via miR-370/FOXO1/Cyclin D1 pathway. Mol Ther Nucleic Acids, 16, 434–441.
Sun, C., Fu, Y., Gu, X., Xi, X., Peng, X., Wang, C., et al. (2020). Macrophage-enriched lncRNA RAPIA: a novel therapeutic target for atherosclerosis. Arteriosclerosis, Thrombosis, And Vascular Biology, 40, 1464–1478.
Yan, L., Liu, Z., Yin, H., Guo, Z., & Luo, Q. (2019). Silencing of MEG3 inhibited ox-LDL-induced inflammation and apoptosis in macrophages via modulation of the MEG3/miR-204/CDKN2A regulatory axis. Cell Biology International, 43, 409–420.
Zhen, Z., Ren, S., Ji, H., Ding, X., Zou, P., & Lu, J. (2019). The lncRNA DAPK-IT1 regulates cholesterol metabolism and inflammatory response in macrophages and promotes atherogenesis. Biochemical And Biophysical Research Communications, 516, 1234–1241.
Qu, S., Zhong, Y., Shang, R., Zhang, X., Song, W., Kjems, J., et al. (2017). The emerging landscape of circular RNA in life processes. Rna Biology, 14, 992–999.
Li, M., Ding, W., Sun, T., Tariq, M. A., Xu, T., Li, P., et al. (2018). Biogenesis of circular RNAs and their roles in cardiovascular development and pathology. Febs Journal, 285, 220–232.
Kattoor, A. J., Kanuri, S. H., & Mehta, J. L. (2019). Role of Ox-LDL and LOX-1 in atherogenesis. Current Medicinal Chemistry, 26, 1693–1700.
Wang, L., Zheng, Z., Feng, X., Zang, X., Ding, W., Wu, F., et al. (2019). circRNA/lncRNA-miRNA-mRNA network in oxidized, Low-Density, Lipoprotein-Induced Foam cells. Dna And Cell Biology, 38, 1499–1511.
Xiong, D. D., Dang, Y. W., Lin, P., Wen, D. Y., He, R. Q., Luo, D. Z., et al. (2018). A circRNA-miRNA-mRNA network identification for exploring underlying pathogenesis and therapy strategy of hepatocellular carcinoma. J Transl Med, 16, 220.
Cai, X., Lin, L., Zhang, Q., Wu, W., & Su, A. (2020). Bioinformatics analysis of the circRNA-miRNA-mRNA network for non-small cell lung cancer. Journal Of International Medical Research, 48, 300060520929167.
Chen, L. Y., Xia, X. D., Zhao, Z. W., Gong, D., Ma, X. F., Yu, X. H., et al. (2018). MicroRNA-377 inhibits atherosclerosis by regulating triglyceride metabolism through the DNA methyltransferase 1 in apolipoprotein E-Knockout mice. Circulation Journal, 82, 2861–2871.
Yin, K., Liao, D. F., & Tang, C. K. (2010). ATP-binding membrane cassette transporter A1 (ABCA1): a possible link between inflammation and reverse cholesterol transport. Molecular Medicine, 16, 438–449.
Cao, Q., Guo, Z., Du, S., Ling, H., & Song, C. (2020). Circular RNAs in the pathogenesis of atherosclerosis. Life Sciences, 255, 117837.
Yu, X. H., Fu, Y. C., Zhang, D. W., Yin, K., & Tang, C. K. (2013). Foam cells in atherosclerosis. Clinica Chimica Acta, 424, 245–252.
An, J., Shi, H., Zhang, N., & Song, S. (2019). Elevation of circular RNA circ_0003645 forecasts unfavorable prognosis and facilitates cell progression via miR-1179/TMEM14A pathway in non-small cell lung cancer. Biochemical And Biophysical Research Communications, 511, 921–925.
Qin, M., Wang, W., Zhou, H., Wang, X., Wang, F., & Wang, H. (2020). Circular RNA circ_0003645 silencing alleviates inflammation and apoptosis via the NF-kappaB pathway in endothelial cells induced by oxLDL. Gene, 755, 144900.
Guo, Y., Huang, S., Ma, Y., Zhang, J., Wen, Y., Zhou, L., et al. (2019). MiR-377 mediates the expression of syk to attenuate atherosclerosis lesion development in ApoE(-/-) mice. Biomedicine & Pharmacotherapy, 118, 109332.
Wang, H., Wei, Z., Li, H., Guan, Y., Han, Z., Wang, H., et al. (2020). MiR-377-3p inhibits atherosclerosis-associated vascular smooth muscle cell proliferation and migration via targeting neuropilin2. Bioscience Reports, 40, BSR20193425.
O’Brien, J., Hayder, H., Zayed, Y., & Peng, C. (2018). Overview of MicroRNA Biogenesis, Mechanisms of actions, and circulation. Front Endocrinol (Lausanne), 9, 402.
Tang, X., Yin, R., Shi, H., Wang, X., Shen, D., Wang, X. (2020). LncRNA ZFAS1 confers inflammatory responses and reduces cholesterol efflux in atherosclerosis through regulating miR-654-3p-ADAM10/RAB22A axis. Int. J. Cardiol.
Acknowledgements
None.
Funding
There is no funding to report.
Author information
Authors and Affiliations
Contributions
G.S. designed research, X.Y. performed experiments, H.C. analyzed data, and wrote the manuscript. Y.X. contributed the methodology, L.W. edited the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics Approval and Consent to Participate
This study was approved by the Ethics Committee of the Second Affiliated Hospital of Hainan Medical University (Number: LW2022241).
Consent for Publication
Not applicable.
Competing Interests
The authors declare that there are no competing interests associated with the manuscript.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Xuejiao Yin and Hongdan Chen contributed equally to this work
Electronic Supplementary Material
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Yin, X., Chen, H., Sun, G. et al. Circ-C16orf62 Regulates Oxidized low-density Lipoprotein-induced Apoptosis, Inflammation, Oxidative Stress and Cholesterol Accumulation of Macrophages via Mediating RAB22A Expression by Targeting miR-377. Appl Biochem Biotechnol 195, 6586–6606 (2023). https://doi.org/10.1007/s12010-023-04320-4
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12010-023-04320-4