Abstract
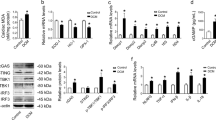
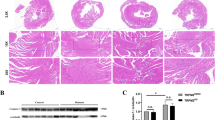
Chronic hyperglycemia induces reactive oxygen species that have an essential function in tissue injuries in cases of diabetic cardiomyopathy. The mechanism of the absent in melanoma 2 (AIM2)-associated inflammasome response in diabetic cardiomyopathy is unknown. Therefore, this study was performed to investigate the role of AIM2 and its molecular mechanisms. Diabetic rats received 1 × 108 viral injections of 5′-GGTCACCAGTTCCTCAGTT-3′ (n = 15) or 5′-TTCTCCGAACGTGTCACGT-3′ (negative control group, n = 15). Normal rats (n = 15) and diabetic rats (n = 15) were also included in the experiment. Ex vivo study was performed on primary cardiomyocytes for different concentrations of glucose. AIM2 inhibition did not affect any of the metabolic parameters (p > 0.05 for all). AIM2 protein levels were significantly increased in rats with diabetes mellitus compared with those in the control group (p < 0.0001, q = 32.044). Also, viral injection (sequence: 5′-GGTCACCAGTTCCTCAGTT-3′) decreased the diabetes mellitus-induced increase in expression of AIM2 protein levels (p < 0.0001, q = 27.129). Cardiac dysfunctions were reported in rats with diabetes mellitus characterized by several parameters (p < 0.01 for all). The diabetic myocardium of rats was reported to have higher deposits of extracellular matrix compared to the control rats (p < 0.001). These effects were downregulated by viral injection (sequence: 5′-GGTCACCAGTTCCTCAGTT-3′). Ex vivo research revealed that high glucose concentrations significantly increased AIM2 protein expression, reactive oxygen species, and cell death. AIM2 protein in diabetic cardiomyopathy is associated with reactive oxygen species production and cardiomyocyte death.






Similar content being viewed by others
Data Availability
The data used to support the findings of this study are available from the corresponding author upon request.
Abbreviations
- AIM2:
-
Absent in melanoma 2
- ANOVA:
-
Analysis of variance
- SD:
-
Standard deviation
- Q :
-
Critical value for post hoc analysis
References
Wang, X., Pan, J., Liu, H., Zhang, M., Liu, D., Lu, L., Tian, J., Liu, M., Jin, T., & An, F. (2019). AIM2 gene silencing attenuates diabetic cardiomyopathy in type 2 diabetic rat model. Life Science, 221, 249–258.
Faria, A., & Persaud, S. J. (2017). Cardiac oxidative stress in diabetes: Mechanisms and therapeutic potential. Pharmacology & Therapeutics, 172, 50–62.
Roul, D., & Recchia, F. A. (2015). Metabolic alterations induce oxidative stress in diabetic and failing hearts: Different pathways, same outcome. Antioxidants & Redox Signaling, 22(17), 1502–1514.
Palomer, X., Salvadó, L., Barroso, E., & Vázquez-Carrera, M. (2013). An overview of the crosstalk between inflammatory processes and metabolic dysregulation during diabetic cardiomyopathy. International Journal of Cardiology, 168(4), 3160–3172.
Fernandes-Alnemri, T., Yu, J. W., Datta, P., Wu, J., & Alnemri, E. S. (2009). AIM2 activates the inflammasome and cell death in response to cytoplasmic DNA. Nature, 458(7237), 509–513.
Strowig, T., Henao-Mejia, J., Elinav, E., & Flavell, R. (2012). Inflammasomes in health and disease. Nature, 481(7381), 278–286.
Dawson, M. J., & Trapani, J. A. (1996). HIN-200: A novel family of IFN-inducible nuclear proteins expressed in leukocytes. Journal of Leukocyte Biology, 60(3), 310–316.
Hakimi, M., Peters, A., Becker, A., Böckler, D., & Dihlmann, S. (2014). Inflammation-related induction of absent in melanoma 2 (AIM2) in vascular cells and atherosclerotic lesions suggests a role in vascular pathogenesis. Journal of Vascular Surgery, 59(3), 794–803.
Jorgensen, I., Rayamajhi, M., & Miao, E. A. (2017). Programmed cell death as a defence against infection. Nature Reviews Immunology, 17(3), 151–164.
Xia, Y., Gong, L., Liu, H., Luo, B., Li, B., Li, R., Li, B., Lv, M., Pan, J., & An, F. (2015). Inhibition of prolyl hydroxylase 3 ameliorates cardiac dysfunction in diabetic cardiomyopathy. Molecular and Cellular Endocrinology, 403, 21–29.
Korkmaz-Icöz, S., Al Said, S., Radovits, T., Li, S., Brune, M., Hegedűs, P., Atmanli, A., Ruppert, M., Brlecic, P., Lehmann, L. H., Lahrmann, B., Grabe, N., Yoshikawa, Y., Yasui, H., Most, P., Karck, M., & Szabó, G. (2016). Oral treatment with a zinc complex of acetylsalicylic acid prevents diabetic cardiomyopathy in a rat model of type-2 diabetes: Activation of the Akt pathway. Cardiovascular Diabetology, 15, 75.
Su, W., Zhang, Y., Zhang, Q., Xu, J., Zhan, L., Zhu, Q., Lian, Q., Liu, H., Xia, Z. Y., Xia, Z., & Lei, S. (2016). N-acetylcysteine attenuates myocardial dysfunction and postischemic injury by restoring caveolin-3/eNOS signaling in diabetic rats. Cardiovascular Diabetology, 15(1), 146.
Guo, W., Liu, X., Li, J., Shen, Y., Zhou, Z., Wang, M., Xie, Y., Feng, X., Wang, L., & Wu, X. (2018). Prdx1 alleviates cardiomyocyte apoptosis through ROS-activated MAPK pathway during myocardial ischemia/reperfusion injury. International Journal of Biological Macromolecules, 112, 608–615.
Wang, X. M., Wang, Y. C., Liu, X. J., Wang, Q., Zhang, C. M., Zhang, L. P., Liu, H., Zhang, X. Y., Mao, Y., & Ge, Z. M. (2017). BRD7 mediates hyperglycaemia-induced myocardial apoptosis via endoplasmic reticulum stress signalling pathway. Journal of Cellular and Molecular Medicine, 21(6), 1094–1105.
Liu, H., Xia, Y., Li, B., Pan, J., Lv, M., Wang, X., & An, F. (2016). Prolyl hydroxylase 3 overexpression accelerates the progression of atherosclerosis in ApoE-/- mice. Biochemical and Biophysical Research Communications, 73(1), 99–106.
Zannad, F., Cannon, C. P., Cushman, W. C., Bakris, G. L., Menon, V., Perez, A. T., Fleck, P. R., Mehta, C. R., Kupfer, S., Wilson, C., Lam, H., White, W. B., EXAMINE Investigators. (2015). Heart failure and mortality outcomes in patients with type 2 diabetes taking alogliptin versus placebo in EXAMINE: A multicentre, randomised, double-blind trial. Lancet, 385(9982), 2067–2076.
Katsuda, Y., Ohta, T., Miyajima, K., Kemmochi, Y., Sasase, T., Tong, B., Shinohara, M., & Yamada, T. (2014). Diabetic complications in obese type 2 diabetic rat models. Experimental Animals, 63(2), 121–132.
Feng, B., Chen, S., Gordon, A. D., & Chakrabarti, S. (2017). miR-146a mediates inflammatory changes and fibrosis in the heart in diabetes. Journal of Molecular and Cellular Cardiology, 105, 70–76.
Qin, W. D., Liu, G. L., Wang, J., Wang, H., Zhang, J. N., Zhang, F., Ma, Y., Ji, X. Y., Li, C., & Zhang, M. X. (2016). Poly (ADP-ribose) polymerase 1 inhibition protects cardiomyocytes from inflammation and apoptosis in diabetic cardiomyopathy. Oncotarget, 7(24), 35618–35631.
de Vasconcelos, N. M., Van Opdenbosch, N., Van Gorp, H., Parthoens, E., & Lamkanfi, M. (2019). Single-cell analysis of pyroptosis dynamics reveals conserved GSDMD-mediated subcellular events that precede plasma membrane rupture. Cell Death and Differentiation, 26(1), 146–161.
DiPeso, L., Ji, D. X., Vance, R. E., & Price, J. V. (2017). Cell death and cell lysis are separable events during pyroptosis. Cell Death Discovery, 3, 17070.
Wilson, A. J., Gill, E. K., Abudalo, R. A., Edgar, K. S., Watson, C. J., & Grieve, D. J. (2018). Reactive oxygen species signalling in the diabetic heart: Emerging prospect for therapeutic targeting. Heart, 104(4), 293–299.
Acknowledgements
The authors are thankful to the higher authorities for the facilities provided.
Funding
This work was supported by grants from the Department of Health of Hebei Province (20150148, 1020140269, 20130129).
Author information
Authors and Affiliations
Contributions
This study was done by the authors named in this article, and the authors accept all liabilities resulting from claims which relate to this article and its contents.
Corresponding author
Ethics declarations
Ethics Approval and Consent to Participate
All investigations were conducted in accordance with the National Institutes of Health Guidelines for the Protection and Handling of Laboratory Animals (USA), as approved by the institute’s local ethics board Reg. No: (3276/2020/CPCDIA/12.03.2021), as well as animal care and practice requirements.
Conflict of Interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Li, Q., Liu, Y., Meng, J. et al. Role of AIM2 Gene Knockdown Mechanism in Diabetic Cardiomyopathy: an In Vivo and Ex Vivo Study. Appl Biochem Biotechnol 195, 3533–3545 (2023). https://doi.org/10.1007/s12010-022-04306-8
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12010-022-04306-8