Abstract
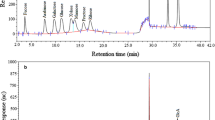
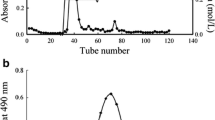
In this study, two extracellular polysaccharides (TEPS1 and TEPS2) were isolated from the endophytic fungus (Talaromyces purpureogenus) and purified by DEAE-Sepharose Fast Flow column using NaCl as gradient eluent. The HPLC analysis displayed that TEPS1 was composed of mannose (38.70%), ribose (25.02%), glucose (19.34%), and galactose (16.94%) while the TEPS2 composed by mannose (100%). The NMR results indicated that TEPS1 exhibited α-glycosidic configurations. The both polysaccharides, TEPS1 and TEPS2 were exhibited a good antioxidant activity in terms of DPPH, ABTS, and •OH scavenging. However, TEPS1 showed a higher antioxidant activity than TEPS2. The IC50 of TEPS1 were 32.16, 192.57, and 54.67 μg·mL−1, for DPPH, ABTS, and •OH radical scavenging, respectively. Furthermore, TEPS1 showed the high cellular antioxidant and wound healing activity in the human embryonic kidney (HEK293) cell line. Overall, these two polysaccharides were promising in antioxidant activity.










Similar content being viewed by others
Data Availability
The data generated and analyzed during the current study are available from the corresponding author on reasonable request.
References
Liang, X.-X., Gao, Y.-Y., Pan, Y., Zou, Y.-F., He, M., He, C.-L., Li, L.-X., Yin, Z.-Q., & Lv, C. (2019). Purification, chemical characterization and antioxidant activities of polysaccharides isolated from Mycena dendrobii. Carbohydrate Polymers, 203, 45–51.
Kungel, P. T. A. N., Correa, V. G., Corrêa, R. C. G., Peralta, R. A., Soković, M., Calhelha, R. C., Bracht, A., Ferreira, I. C. F. R., & Peralta, R. M. (2018). Antioxidant and antimicrobial activities of a purified polysaccharide from yerba mate (Ilex paraguariensis). International Journal of Biological Macromolecules, 114, 1161–1167.
Li, H., Cao, K., Cong, P., Liu, Y., Cui, H., & Xue, C. (2018). Structure characterization and antitumor activity of the extracellular polysaccharide from the marine fungus Hansfordia sinuosae. Carbohydrate Polymers, 190, 87–94.
Yang, J. P., Hsu, T., Lin, F., Hsu, W., & Chen, Y. (2012). Potential antidiabetic activity of extracellular polysaccharides in submerged fermentation culture of Coriolus versicolor LH1. Carbohydrate Polymers, 90, 174–180.
Wang, Z., Xue, R., Cui, J., Wang, J., Fan, W., Zhang, H., & Zhan, X. (2019). Antibacterial activity of a polysaccharide produced from Chaetomium globosum CGMCC 6882. International Journal of Biological Macromolecules, 125, 376–382.
Yu, W., Chen, G., Zhang, P., & Chen, K. (2016). Purification, partial characterization and antitumor effect of an exopolysaccharide from Rhizopus nigricans. International Journal of Biological Macromolecules, 82, 299–307.
Freitas, F., Torres, C. A. V., & Reis, M. A. M. (2017). Engineering aspects of microbial exopolysaccharide production. Bioresource Technology, 245, 1674–1683.
Wang, Y., Li, Y., Li, S., Li, Q., Fan, W., Kiatoukosin, L., & Chen, J. (2019). Extracellular polysaccharides of endophytic fungus Alternaria tenuissima F1 from Angelica sinensis: Production conditions, purification, and antioxidant properties. International Journal of Biological Macromolecules, 133, 172–183.
Li, S.-C., Yang, X.-M., Ma, H.-L., Yan, J.-K., & Guo, D.-Z. (2015). Purification, characterization and antitumor activity of polysaccharides extracted from Phellinus igniarius mycelia. Carbohydrate Polymers, 133, 24–30.
Kumari, M., Taritla, S., Sharma, A., & Jayabaskaran, C. (2018). Antiproliferative and antioxidative bioactive compounds in extracts of marine-derived endophytic fungus Talaromyces purpureogenus. Frontiers in Microbiology, 9, 1777.
Wang, W., Wan, X., Liu, J., Wang, J., Zhu, H., Chen, C., & Zhang, Y. (2018). Two new terpenoids from Talaromyces purpurogenus. Mar Drugs, 16(5), 150.
Sahu, M. K., Kaushik, K., Das, A., & Jha, H. (2020). In vitro and in silico antioxidant and antiproliferative activity of rhizospheric fungus Talaromyces purpureogenus isolate-ABRF2. Bioresources and Bioprocessing, 7, 14.
Masuko, T., Minami, A., Iwasaki, N., Majima, T., Nishimura, S.-I., & Lee, Y. C. (2005). Carbohydrate analysis by a phenol–sulfuric acid method in microplate format. Analytical Biochemistry, 339, 69–72.
Bradford, M. M. (1976). A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Analytical Biochemistry, 72, 248–254.
Mariadoss, A. V., Park, S., Saravanakumar, K., Sathiyaseelan, A., & Wang, M.-H. (2021). Ethyl Acetate fraction of Helianthus tuberosus L. induces anti-diabetic, and wound-healing activities in insulin-resistant human liver cancer and mouse fibroblast cells. Antioxidants, 10(1), 99.
Cai, L., Zou, S., Liang, D., & Luan, L. (2018). Structural characterization, antioxidant and hepatoprotective activities of polysaccharides from Sophorae tonkinensis Radix. Carbohydrate Polymers, 184, 354–365.
An, L., Si, C., Wang, G., Sui, W., & Tao, Z. (2019). Enhancing the solubility and antioxidant activity of high-molecular-weight lignin by moderate depolymerization via in situ ethanol/acid catalysis. Industrial Crops and Products, 128, 177–185.
Saravanakumar, K., Park, S., Sathiyaseelan, A., Kim, K.-N., Cho, S.-H., Mariadoss, A. V., & Wang, M.-H. (2021). Metabolite profiling of methanolic extract of Gardenia jaminoides by LC-MS/MS and GC-MS and its anti-diabetic, and anti-oxidant activities. Pharmaceuticals, 14(2), 102.
Hu, X., Saravanakumar, K., Jin, T., & Wang, M.-H. (2019). Mycosynthesis, characterization, anticancer and antibacterial activity of silver nanoparticles from endophytic fungus Talaromyces purpureogenus. International Journal of Nanomedicine, 14, 3427–3438.
Saravanakumar, K., Vivek, R., Sithranga Boopathy, N., Yaqian, L., Kathiresan, K., & Chen, J. (2015). Anticancer potential of bioactive 16-methylheptadecanoic acid methyl ester derived from marine Trichoderma. Journal of Applied Biomedicine, 13, 199–212.
Shi, L. (2016). Bioactivities, isolation and purification methods of polysaccharides from natural products: A review. International journal of biological macromolecules, 92, 37–48.
Zhang, M., Wang, G., Lai, F., & Wu, H. (2016). Structural characterization and immunomodulatory activity of a novel polysaccharide from Lepidium meyenii. Journal of Agricultural and Food Chemistry, 64, 1921–1931.
Huang, S.-Q., Li, J.-W., Li, Y.-Q., & Wang, Z. (2011). Purification and structural characterization of a new water-soluble neutral polysaccharide GLP-F1-1 from Ganoderma lucidum. International Journal of Biological Macromolecules, 48, 165–169.
Wang, F., Wang, W., Huang, Y., Liu, Z., & Zhang, J. (2015). Characterization of a novel polysaccharide purified from a herb of Cynomorium songaricum Rupr. Food Hydrocolloids, 47, 79–86.
Li, B., Zhang, N., Wang, D. X., Jiao, L., Tan, Y., Wang, J., Li, H., Wu, W., & Jiang, D. C. (2018). Structural analysis and antioxidant activities of neutral polysaccharide isolated from Epimedium koreanum Nakai. Carbohydrate Polymers, 196, 246–253.
Ye, L., Zhang, J., Yang, Y., Zhou, S., Liu, Y., Tang, Q., Du, X., Chen, H., & Pan, Y. (2009). Structural characterisation of a heteropolysaccharide by NMR spectra. Food Chemistry, 112, 962–966.
Xiu, A., Kong, Y., Zhou, M., Zhu, B., Wang, S., & Zhang, J. (2010). The chemical and digestive properties of a soluble glucan from Agrobacterium sp. ZX09. Carbohydrate Polymers, 82, 623–628.
Wang, X., Zhang, M., Zhang, D., Wang, X., Cao, H., Zhang, Q., & Yan, C. (2019). Structural elucidation and anti-osteoporosis activities of polysaccharides obtained from Curculigo orchioides. Carbohydrate Polymers, 203, 292–301.
Zeng, Y.-J., Yang, H.-R., Ou, X.-Y., Su, H.-H., Zong, M.-H., Yang, J.-G., & Lou, W.-Y. (2019). Fungal polysaccharide similar with host Dendrobium officinale polysaccharide: Preparation, structure characteristics and biological activities. International Journal of Biological Macromolecules, 141, 460–470.
Sharma, O. P., & Bhat, T. K. (2009). DPPH antioxidant assay revisited. Food Chemistry, 113, 1202–1205.
Re, R., Pellegrini, N., Proteggente, A., Pannala, A., Yang, M., & Rice-Evans, C. (1999). Antioxidant activity applying an improved ABTS radical cation decolorization assay. Free Radical Biology and Medicine, 26, 1231–1237.
Reiter, R. J., Melchiorri, D., Sewerynek, E., Poeggeler, B., Barlow-Walden, L., Chuang, J., Ortiz, G. G., & AcuñaCastroviejo, D. (1995). A review of the evidence supporting melatonin’s role as an antioxidant. Journal of Pineal Research, 18, 1–11.
Koppenol, W. H. (1993). The centennial of the Fenton reaction. Free Radical Biology and Medicine, 15, 645–651.
Joanna, K., Michał, P. & Anna, P. (2017). Osmotic properties of polysaccharides solutions. Solubility of Polysaccharides, 23. https://doi.org/10.5772/intechopen.69864
Birnboim, H. C. (1986). DNA strand breaks in human leukocytes induced by superoxide anion, hydrogen peroxide and tumor promoters are repaired slowly compared to breaks induced by ionizing radiation. Carcinogenesis, 7, 1511–1517.
Dogan,K.H., (2019). In Wound Healing-Current Perspectives, IntechOpen. 260p
Funding
This work was supported by Brain Pool Program through the National Research Foundation of Korea (NRF) funded by the Ministry of Science, ICT and Future Planning (2017H1D3A1A01052610), National Research Foundation of Korea (2019R1A1055452) and Korea Basic Science Institute (C210300).
Author information
Authors and Affiliations
Contributions
Xiaowen Hu: data curation, formal analysis, investigation, methodology, writing—review and editing. Kandasamy Saravanakumar: conceptualization, data curation, formal analysis, investigation, methodology, visualization, writing—original draft, writing—review and editing. SeonJu Park: formal analysis, data curation. Ki-seok Han: formal analysis, investigation. Myeong-Hyeon Wang: project administration, resources, supervision, validation, writing—review and editing.
Corresponding author
Ethics declarations
Ethics Approval
This article does not contain any studies with animals or human participants.
Consent to Participate
The authors agreed to participate in this work.
Consent for Publication
The authors agreed to publish this work.
Conflict of Interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Hu, X., Saravanakumar, K., Park, S. et al. Isolation, Characterization, Antioxidant, and Wound Healing Activities of Extracellular Polysaccharide from Endophytic Fungus Talaromyces purpureogenus. Appl Biochem Biotechnol 195, 3822–3839 (2023). https://doi.org/10.1007/s12010-022-04187-x
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12010-022-04187-x