Abstract
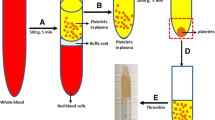
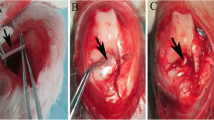
Anterior cruciate ligament (ACL) rupture is the most common sports injuries and PRP has the potential to be a kartogenin (KGN) carrier to promote collagen fibril organization and cartilage regenerative in the tendon-bone interface. This paper aimed to investigate co-injection of KGN-PRP into the bone tunnels of ACL reconstructions which could enhance tendon-bone healing graft osteointegration effectively. HPLC was used to measured release rate of KGN from KGN-PRP gel. Then, an ACL injury reconstruction model in rabbits was established and the rabbits received saline, PRP, and KGN-PRP injection onto the tendon-bone interface after reconstruction. The tissue was harvested from the tendon-bone interface at 4 weeks and 8 weeks post-surgery, and the sections were stained with Safranin O/fast green to detected tendon-bone healing. Immunochemistry staining was used to analyze VEGF, collagen I, and HIF-1α expression, and ELISA assay was used for detecting IL-6, TNF-α, and COX-2 concentrations. The expression levels of AKT/PI3K/NF-κB-related protein and mRNA were presented by Western blot and qPCR. The release rate of KGN was high within 4 h of KGN-PRP gel and followed by a slow release until 7 days. The Safranin O/fast green staining results indicated that tendon-bone interface in sham and mock group existed gap and tissue disorganization. The KGN + PRP group showed the positive color of the healing interface was more obvious and cartilage tissue began to be generated in large amounts at this interface. The maximum tensile force of KGN-PRP injection tendon-bone healing site was significantly higher than that of PRP group, and KGN-PRP effectively promoted fibro chondrogenesis and tendon-bone healing. The expression of collagen I, VEGF, and HIF-1α in regenerated tissues at the healing interface was significantly increased by KGN-PRP treatment compared with the mock and sham groups. The expressions of IL-6, TNF-α, and COX-2 after KGN-PRP treatment were significantly decreased in tendon-bone interface compared to the mock group. WB and qPCR results showed KGN-PRP treatment effectively inhibits AKT/PI3K/NF-κB activation of inflammatory pathways, thereby reducing the level of inflammation to promote wound healing. PRP is an effective carrier for KGN with the sustained release of KGN. After ACL reconstruction, injection of KGN-PRP gel significantly reduced the inflammatory response and inhibited AKT/PI3K/NF-κB activation in cartilage tissue, which promoted tendon-bone healing.





Similar content being viewed by others
Dara Availability
Not Applicable.
Abbreviations
- PRP:
-
Platelet-rich plasma
- ACL:
-
Anterior cruciate ligament
- PI3K:
-
Phosphatidylinositol-3 kinase
- AKT/PKB:
-
Protein kinase B
- NF-κB:
-
Nuclear factor-kappa B
- KGN:
-
Kartogenin
- HPLC:
-
High-performance liquid chromatography
- VEGF:
-
Vascular endothelial growth factor
- HIF1α:
-
Hypoxia-inducible factor-1α
- ELISA:
-
Enzyme-linked immunosorbent assay
- IL-6:
-
Interleukin 6
- TNF-α:
-
Tumor necrosis factor-alpha
- COX-2:
-
Cyclooxygenase-2
- WB:
-
Western blotting
- qPCR:
-
Quantitative polymerase chain reaction
- PBS:
-
Phosphate-buffered saline
- EDTA:
-
Ethylenediaminetetraacetic acid
- BCA:
-
Bicinchoninic acid
- PVDF:
-
Polyvinylidene fluoride
- hASCs:
-
Human adipose stromal cells
- rhBMP-2:
-
Recombinant human bone morphogenetic protein-2
- IL-1β:
-
Interleukin-1 beta
- ROS:
-
Reactive oxygen species
- OA:
-
Osteoarthritis
- SOX9:
-
SRY-box transcription factor 9
- Coll2:
-
Type II collagen
- ACAN:
-
Aggrecan
- MMP13:
-
Matrix metallopeptidase-13
- ERK:
-
Extracellular signal–regulated kinase
- ANGPTL4:
-
Angiopoetin-like 4
- TGF-β:
-
Transforming growth factor beta
References
Roos, P. E., Button, K., & van Deursen, R. W. M. (2014). Motor control strategies during double leg squat following anterior cruciate ligament rupture and reconstruction: An observational study. Journal of neuroengineering and rehabilitation, 11, 19–29.
Huang, H., Yin, W., Ren, S., et al. (2019). Muscular force patterns during level walking in ACL-deficient patients with a concomitant medial meniscus tear. Applied Bionics and Biomechanics, 2019, 7921785.
Bi, F., Shi, Z., Jiang, S., et al. (2014). Intermittently administered parathyroid hormone [1-34] promotes tendon-bone healing in a rat model. International Journal of Molecular Sciences, 15(10), 66–79.
Wang, D., Tan, H., Lebaschi, A. H., et al. (2018). Kartogenin enhances collagen organization and mechanical strength of the repaired enthesis in a murine model of rotator cuff repair. Arthroscopy, 34(9), 2579–2587.
Liu, F., Xu, H., & Huang, H. (2019). A novel kartogenin-platelet-rich plasma gel enhances chondrogenesis of bone marrow mesenchymal stem cells in vitro and promotes wounded meniscus healing in vivo. Stem cell research & therapy, 10(1), 201–211.
Zhang, J., Zhang, J., Zhang, N., et al. (2020). The effects of platelet-rich and platelet-poor plasma on biological characteristics of BM-MSCs in vitro. Analytical Cellular Pathology (Amsterdam), 2020, 8546231.
Zhu, J., Shao, J., Chen, Y., et al. (2021). Fibrin glue-kartogenin complex promotes the regeneration of the tendon-bone interface in rotator cuff injury. Stem Cells International, 2021, 6640424.
Zhang, J., Yuan, T., Zheng, N., et al. (2017). The combined use of kartogenin and platelet-rich plasma promotes fibrocartilage formation in the wounded rat Achilles tendon entheses. Bone & Joint Research, 6(4), 231–244.
Chen, P., Ouyang, J., Xiao, J., et al. (2018). Co-injection of human adipose stromal cells and rhBMP-2/fibrin gel enhances tendon graft osteointegration in a rabbit anterior cruciate ligament-reconstruction model. American Journal of Translational Research, 10(2), 535–544.
Ramasamy, S. K., Kusumbe, A. P., Wang, L., et al. (2014). Endothelial Notch activity promotes angiogenesis and osteogenesis in bone. Nature, 507(7492), 376–380.
Liang, B., Liang, J. M., Ding, J. N., et al. (2019). Dimethyloxaloylglycine-stimulated human bone marrow mesenchymal stem cell-derived exosomes enhance bone regeneration through angiogenesis by targeting the AKT/mTOR pathway. Stem Cell Research & Therapy, 10(1), 335.
Monaco, E., Mazza, D., Daggett, M., et al. (2021). Acute primary repair of the anterior cruciate ligament with anterolateral ligament augmentation. Arthroscopy Techniques, 10(6), e1633–e1639.
Chen, R., Zhu, H., Gu, X., et al. (2022). Effects of platelet-rich plasma on tendon-bone healing after anterior cruciate ligament reconstruction. Orthopaedic Surgery, 14(1), 88–95.
Hexter, A., Sanghani-Kerai, A., Heidari, N., et al. (2021). Mesenchymal stromal cells and platelet-rich plasma promote tendon allograft healing in ovine anterior cruciate ligament reconstruction. Knee Surgery, Sports Traumatology, Arthroscopy: Official Journal of the ESSKA, 29(11), 3678–3688.
Yin, W., Qi, X., Zhang, Y., et al. (2016). Advantages of pure platelet-rich plasma compared with leukocyte- and platelet-rich plasma in promoting repair of bone defects. Journal of Translational Medicine, 14, 73–84.
Zhou, Y., Zhang, J., Wu, H., et al. (2015). The differential effects of leukocyte-containing and pure platelet-rich plasma (PRP) on tendon stem/progenitor cells - implications of PRP application for the clinical treatment of tendon injuries. Stem Cell Research & Therapy, 6(1), 173.
Hou, M., Zhang, Y., Zhou, X., et al. (2021). Kartogenin prevents cartilage degradation and alleviates osteoarthritis progression in mice via the miR-146a/NRF2 axis. Cell Death & Disease, 12(5), 483–493.
Zeng, W., Zhang, Y., Wang, D., et al. (2021). Intra-articular injection of kartogenin-enhanced bone marrow-derived mesenchymal stem cells in the treatment of knee osteoarthritis in a rat model. The American Journal of Sports Medicine, 49(10), 2795–2809.
Liu, X., Zhu, B., Li, Y., et al. (2021). The role of vascular endothelial growth factor in tendon healing. Frontiers in Physiology, 12, 766080.
Huang, Y., Pan, M., Shu, H., et al. (2020). Vascular endothelial growth factor enhances tendon-bone healing by activating Yes-associated protein for angiogenesis induction and rotator cuff reconstruction in rats. Journal of Cellular Biochemistry, 121(3), 2343–2353.
Dyment, N. A., Kazemi, N., Aschbacher-Smith, L. E., et al. (2011). The relationships among spatiotemporal collagen gene expression, histology, and biomechanics following full-length injury in the murine patellar tendon. Journal of Orthopaedic Research, 30(1), 28–36.
Mousavizadeh, R., Scott, A., Lu, A., et al. (2015). Angiopoietin-like 4 promotes angiogenesis in the tendon and is increased in cyclically loaded tendon fibroblasts. Journal of Physiology, 594(11), 1–10.
Author information
Authors and Affiliations
Contributions
All authors contributed equally.
Corresponding author
Ethics declarations
Ethical Approval
The authors’ institution provided ethical approval for the conduct of this study 2022–091.
Consent to Participate
All authors have their consent to participate.
Consent for publication
All authors have their consent to publish their work.
Competing Interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zhang, L., Zhang, Q., Cui, L. et al. Kartogenin Combined Platelet-Rich Plasma (PRP) Promoted Tendon-Bone Healing for Anterior Cruciate Ligament (ACL) Reconstruction by Suppressing Inflammatory Response Via targeting AKT/PI3K/NF-κB. Appl Biochem Biotechnol 195, 1284–1296 (2023). https://doi.org/10.1007/s12010-022-04178-y
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12010-022-04178-y