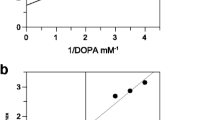
Abstract
Tyrosinase is the rate-limiting enzyme for melanin production in plant and mammalian cells. Upregulation of this enzyme results in hyperpigmentation disorders. In order to treat pigmentation problems, novel skin whitening compounds are extremely screened. It is found that fatty acids based on their saturation levels either increase or decrease tyrosinase enzyme activity. Thus, fatty acids and their compositions are promising candidates for the treatment of hyperpigmentation or hypopigmentation disorders. Microalgae are rich in both saturated and unsaturated fatty acids, as well. In this study, C. cohnii and S. limacinum fatty acids were evaluated as tyrosinase inhibitor candidates. Mushroom tyrosinase activity studies displayed that both extracts increase tyrosinase enzyme activity dose-dependently. On the other hand, S. limacinum at 200 µg ml−1 concentration almost decreased half of tyrosinase enzyme activity in B16-F10 cells. Besides, it was 3 times more efficient for tyrosinase enzyme activity inhibition and 2 times more effective to decrease melanin synthesis compared to C. cohnii. Considering low toxicity to B16-F10 melanoma and healthy keratinocyte cells (HaCaT), S. limacinum fatty acids could be a suitable source for lipid-based tyrosinase inhibitory functional cosmetics products.





Similar content being viewed by others
Data Availability
Not applicable.
References
Chang, T. S. (2012). Natural melanogenesis inhibitors acting through the down-regulation of tyrosinase activity. Materials, 5(9), 1661–1685. https://doi.org/10.3390/ma5091661
D’Mello, S. A. N., Finlay, G. J., Baguley, B. C., & Askarian-Amiri, M. E. (2016). Signaling pathways in melanogenesis. International Journal of Molecular Sciences, 17(7), 1–18. https://doi.org/10.3390/ijms17071144
Heo, S. J., Ko, S. C., Cha, S. H., Kang, D. H., Park, H. S., Choi, Y. U., & Jeon, Y. J. (2009). Effect of phlorotannins isolated from Ecklonia cava on melanogenesis and their protective effect against photo-oxidative stress induced by UV-B radiation. Toxicology in Vitro. https://doi.org/10.1016/j.tiv.2009.05.013
Pillaiyar, T., Namasivayam, V., Manickam, M., & Jung, S. H. (2018). Inhibitors of melanogenesis: An updated review. Journal of Medicinal Chemistry, 61(17), 7395–7418. https://doi.org/10.1021/acs.jmedchem.7b00967
Oh, T. I., Yun, J. M., Park, E. J., Kim, Y. S., Lee, Y. M., & Lim, J. H. (2017). Plumbagin suppresses α-MSH-induced melanogenesis in B16F10 mouse melanoma cells by inhibiting tyrosinase activity. International Journal of Molecular Sciences, 18(2). https://doi.org/10.3390/ijms18020320
Chan, C. F., Huang, C. C., Lee, M. Y., & Lin, Y. S. (2014). Fermented broth in tyrosinase- and melanogenesis inhibition. Molecules, 19(9), 13122–13135. https://doi.org/10.3390/molecules190913122
Kubglomsong, S., Theerakulkait, C., Reed, R. L., Yang, L., Maier, C. S., & Stevens, J. F. (2018). Isolation and identification of tyrosinase-inhibitory and copper-chelating peptides from hydrolyzed rice-bran-derived albumin. Journal of Agricultural and Food Chemistry, 66(31), 8346–8354. research-article. https://doi.org/10.1021/acs.jafc.8b01849
Wang, Z. J., Si, Y. X., Oh, S., Yang, J. M., Yin, S. J., Park, Y. D., & Qian, G. Y. (2012). The effect of fucoidan on tyrosinase: Computational molecular dynamics integrating inhibition kinetics. Journal of Biomolecular Structure and Dynamics, 30(4), 460–473. https://doi.org/10.1080/07391102.2012.682211
Ephrem, E., Elaissari, H., & Greige-Gerges, H. (2017). Improvement of skin whitening agents efficiency through encapsulation: Current state of knowledge. International Journal of Pharmaceutics, 526(1–2), 50–68. https://doi.org/10.1016/j.ijpharm.2017.04.020
Kose, A., & Oncel, S. S. (2015). Properties of microalgal enzymatic protein hydrolysates: Biochemical composition, protein distribution and FTIR characteristics. Biotechnology Reports, 6, 137–143. https://doi.org/10.1016/J.BTRE.2015.02.005
Garc ıa, J. L., de Vicente, M., & Gal an, B. (n.d.). Microalgae, old sustainable food and fashion nutraceuticals. https://doi.org/10.1111/1751-7915.12800
Nishigaki, I., Peramaiyan, R., Ramachandran, V., Gnapathy, E., Dhanapal, S., & Yutaka, N. (2010). Cytoprotective role of astaxanthin against glycated protein/iron chelate-induced toxicity in human umbilical vein endothelial cells. Phytotherapy Research, 24(June), 54–59. https://doi.org/10.1002/ptr
Jain, S., Prajapat, G., Abrar, M., Ledwani, L., Singh, A., & Agrawal, A. (2017). Cyanobacteria as efficient producers of mycosporine-like amino acids. Journal of Basic Microbiology. https://doi.org/10.1002/jobm.201700044
Pratoomthai, B., Songtavisin, T., Gangnonngiw, W., & Wongprasert, K. (n.d.). In vitro inhibitory effect of sulfated galactans isolated from red alga Gracilaria fisheri on melanogenesis in B16F10 melanoma cells. https://doi.org/10.1007/s10811-018-1469-3
Rao, A. R., Sindhuja, H. N., Dharmesh, S. M., Sankar, K. U., Sarada, R., & Ravishankar, G. A. (2013). Effective inhibition of skin cancer, tyrosinase, and antioxidative properties by astaxanthin and astaxanthin esters from the green alga haematococcus pluvialis. Journal of Agricultural and Food Chemistry, 61(16), 3842–3851. https://doi.org/10.1021/jf304609j
Custódio, L., Soares, F., Pereira, H., Rodrigues, M. J., Barreira, L., Rauter, A. P., & Varela, J. (2015). Botryococcus braunii and Nannochloropsis oculata extracts inhibit cholinesterases and protect human dopaminergic SH-SY5Y cells from H2O2-induced cytotoxicity. Journal of Applied Phycology, 27(2), 839–848. https://doi.org/10.1007/s10811-014-0369-4
Levy-Ontman, O., Huleihel, M., Hamias, R., Wolak, T., & Paran, E. (2017). An anti-inflammatory effect of red microalga polysaccharides in coronary artery endothelial cells. Atherosclerosis, 264, 11–18. https://doi.org/10.1016/j.atherosclerosis.2017.07.017
Isleten-Hosoglu, M., & Elibol, M. (2017). Improvement of medium composition and cultivation conditions for growth and lipid production by Crypthecodinium cohnii. Romanian Biotechnological Letters, 22(6), 13086–13095.
Sahin, D., Tas, E., & Altindag, U. H. (2018). Enhancement of docosahexaenoic acid (DHA) production from Schizochytrium sp. S31 using different growth medium conditions. AMB Express, 8(1). https://doi.org/10.1186/s13568-018-0540-4
Ando, H., Watabe, H., Valencia, J. C., Yasumoto, K., Furumura, M., Funasaka, Y., & Hearing, V. J. (2004). Fatty acids regulate pigmentation via proteasomal degradation of tyrosinase. Journal of Biological Chemistry, 279(15), 15427–15433. https://doi.org/10.1074/jbc.m313701200
Balcos, M. C., Kim, S. Y., Jeong, H., Yun, H., Baek, K. J., Kwon, N. S., & Kim, D. (2014). Docosahexaenoic acid inhibits melanin synthesis in murine melanoma cells in vitro through increasing tyrosinase degradation. Nature Publishing Group, 1, 489–495. https://doi.org/10.1038/aps.2013.174
Cui, H. X., Duan, F. F., Jia, S. S., Cheng, F. R., & Yuan, K. (2018). Antioxidant and tyrosinase inhibitory activities of seed oils from Torreya grandis Fort. ex Lindl. BioMed Research International, 2018. https://doi.org/10.1155/2018/5314320
Yildiztekin, F., Nadeem, S., Erol, E., Yildiztekin, M., Tuna, A. L., & Ozturk, M. (2016). Antioxidant, anticholinesterase and tyrosinase inhibition activities, and fatty acids of Crocus mathewii – A forgotten endemic angiosperm of Turkey. Pharmaceutical Biology, 54(9), 1557–1563. https://doi.org/10.3109/13880209.2015.1107746
Isleten-Hosoglu, M., Gultepe, I., & Elibol, M. (2012). Optimization of carbon and nitrogen sources for biomass and lipid production by Chlorella saccharophila under heterotrophic conditions and development of Nile red fluorescence based method for quantification of its neutral lipid content. Biochemical Engineering Journal, 61, 11–19. https://doi.org/10.1016/J.BEJ.2011.12.001
Chan, Y. Y., Kim, K. H., & Cheah, S. H. (2011). Inhibitory effects of Sargassum polycystum on tyrosinase activity and melanin formation in B16F10 murine melanoma cells. Journal of Ethnopharmacology. https://doi.org/10.1016/j.jep.2011.07.050
Sun, L., Guo, Y., Zhang, Y., & Zhuang, Y. (2017). Antioxidant and anti-tyrosinase activities of phenolic extracts from rape bee pollen and inhibitory melanogenesis by cAMP/MITF/TYR pathway in B16 mouse melanoma cells. Frontiers in Pharmacology, 8(MAR), 1–9. https://doi.org/10.3389/fphar.2017.00104
Fistarol, S. K., & Itin, P. H. (2010). Disorders of pigmentation, 2010(Band 8), 187–202. https://doi.org/10.1111/j.1610-0387.2009.07137.x
Chang, T. S. (2009). An updated review of tyrosinase inhibitors. International Journal of Molecular Sciences, 10(6), 2440–2475. https://doi.org/10.3390/ijms10062440
Wang, L. X., Qian, J., Zhao, L. N., & Zhao, S. H. (2018). Effects of volatile oil from ginger on the murine B16 melanoma cells and its mechanism. Food and Function, 9(2), 1058–1069. https://doi.org/10.1039/c7fo01127j
Tel, G., Öztürk, M., Duru, M. E., Doǧan, B., & Harmandar, M. (2013). Fatty acid composition, antioxidant, anticholinesterase and tyrosinase inhibitory activities of four Serratula species from anatolia. Records of Natural Products, 7(2), 86–95.
Shabani, F., & Sariri, R. (2010). Increase of melanogenesis in the presence of fatty acids. Pharmacologyonline, 1, 314–323.
Jeon, S., Kim, N. H., Koo, B. S., Kim, J. Y., & Lee, A. Y. (2009). Lotus (Nelumbo nuficera) flower essential oil increased melanogenesis in normal human melanocytes. Experimental and Molecular Medicine, 41(7), 517–524. https://doi.org/10.3858/emm.2009.41.7.057
Chaikul, P., Sripisut, T., Chanpirom, S., Sathirachawan, K., & Ditthawuthikul, N. (2017). Melanogenesis inhibitory and antioxidant effects of Camellia oleifera seed oil. Advanced Pharmaceutical Bulletin, 7(3), 473–477. https://doi.org/10.15171/apb.2017.057
Matsuura, R., Ukeda, H., & Sawamura, M. (2006). Tyrosinase inhibitory activity of citrus essential oils. Journal of Agricultural and Food Chemistry, 54(6), 2309–2313. https://doi.org/10.1021/jf051682i
Zajdel, A., Wilczok, A., Chodurek, E., Gruchlik, A., & Dzierzewicz, Z. (2013). Polyunsaturated fatty acids inhibit melanoma cell growth in vitro. Acta Poloniae Pharmaceutica - Drug Research, 70(2), 365–369.
Tan, R., Wang, F., Fan, C., & Zhang, X. (2018). Function suppresses B16F10 melanoma lung metastasis by autophagy induction. 6179–6186. https://doi.org/10.1039/c8fo01617h
Mericli, F., Becer, E., Kabadayi, H., Hanoglu, A., Hanoglu, D. Y., Yavuz, D. O., & Vatansever, S. (2017). Fatty acid composition and anticancer activity in colon carcinoma cell lines of Prunus dulcis seed oil. Pharmaceutical Biology, 55(1), 1239–1248. https://doi.org/10.1080/13880209.2017.1296003
Schauss, A. G. (2016). Advances in the study of the health benefits and mechanisms of action of the pulp and seed of the Amazonian palm fruit, Euterpe oleracea Mart., known as “Açai.” Fruits, Vegetables, and Herbs: Bioactive Foods in Health Promotion, 48, 179–220. https://doi.org/10.1016/B978-0-12-802972-5.00010-X
Santos, A., Andres, E., Medina, S. P. H., Ferrari, C. R., & Lourenc, C. B. (2012). Schinus terebinthifolius Raddi extract and linoleic acid from Passiflora edulis synergistically decrease melanin synthesis in B16 cells and reconstituted epidermis. 435–440. https://doi.org/10.1111/j.1468-2494.2012.00736.x
Kose, A., & Oncel, S. S. (2017). Algae as a promising resource for biofuel industry: Facts and challenges. International Journal of Energy Research, 41(7), 924–951. https://doi.org/10.1002/er.3699
Borowitzka, M. A. (2013). High-value products from microalgae-their development and commercialisation. Journal of Applied Phycology, 25(3), 743–756. https://doi.org/10.1007/s10811-013-9983-9
Shigeta, Y., Imanaka, H., Ando, H., Ryu, A., Oku, N., Baba, N., & Makino, T. (2004). Skin whitening effect of linoleic acid is enhanced by liposomal formulations. Biological and Pharmaceutical Bulletin, 27(4), 591–594. https://doi.org/10.1248/bpb.27.591
Acknowledgements
This research study was done at Ege University Bioengineering Department, Microalgal Bioprocess lab. The author would like to thank Assoc. Prof. Dr. Suphi S. Oncel and Prof. Dr. Murat Elibol for their valuable contribution.
Author information
Authors and Affiliations
Contributions
Ayse Kose (AK) designed the experiments, conducted the analysis, and wrote the manuscript.
Corresponding author
Ethics declarations
Ethics Approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Consent to Participate
Not applicable.
Consent to Publish
Not applicable.
Competing Interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Kose, A. Chemical Composition and Tyrosinase Inhibitory Activities of Fatty Acids Obtained from Heterotrophic Microalgae, S. limacinum and C. cohnii. Appl Biochem Biotechnol 195, 369–385 (2023). https://doi.org/10.1007/s12010-022-04143-9
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12010-022-04143-9