Abstract
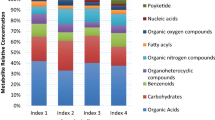
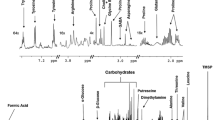
Mungbean is an important legume mainly cultivated in Southeast Asia known for cheap source of food protein. Yellow mosaic disease (YMD) of mungbean is one of the most damaging diseases caused by mungbean yellow mosaic virus (MYMV) and mungbean yellow mosaic India virus (MYMIV) in India. The genetic basis of YMD resistance of mungbean is not well studied yet. Our present studies aimed to explore the genetic basis of YMD resistance through molecular, biochemical and metabolomics approach. Molecular analysis of YMV-infected mungbean plant materials revealed the presence of MYMIV. Chlorophyll contents were estimated as mosaic symptoms that cause chlorosis and necrosis in infected leaves. Chlorophyll a, b and total chlorophyll content were significantly reduced by 27–55% in infected samples compared non-infected control samples. 1H NMR-based metabolomic profiling of virus-infected mungbean were carried out, and we found that vital changes occurred during the development of MYMIV infection in mungbean. A total of fifty metabolites were identified in mungbean leaf samples. Principal component analysis (PCA) and partial least square discriminant analysis (PLS-DA) separated the severely infected sample from the non-infected samples. Orthogonal partial least discrimination analysis (OPLS-DA) revealed significant differences in MYMIV-infected and non-infected control samples. The featured metabolites in MYMIV infected and control samples were amino acids, carbohydrates, and organic acids. Relative abundance of sucrose, γ-amino butyric acid (GABA), proline, alanine, phenylalanine, tryptophan, pyruvate, ascorbate, and citrates were found as differential metabolites. Our results suggest that metabolic changes in infected mungbean samples is related to the viral acquisition. The present study may help in better understanding the metabolic alterations during biotic stress in mungbean.







Similar content being viewed by others
Data Availability
All data generated or analyzed during this study are included in this manuscript.
References
Pratap, A., Douglas, C., Prajapati, U., Kumari, G., War, A. R., Tomar, R., Pandey, A. K., & Dubey, S. (2020). Breeding progress and future challenges: Biotic stresses. In the mung bean genome, Compendium of plant genomes (pp. 55–80). Springer.
Pandey, A. K., Burlakoti, R. R., Kenyon, L., & Nair, R. M. (2018). Perspectives and challenges for sustainable management of fungal diseases of mungbean [Vigna radiata (L.) R. Wilczek var. radiata]: A review. Frontiers in Environmental Science, 6, 53.
Nair, R. M., Götz, M., Winter, S., Giri, R. R., Boddepalli, V. N., Sirari, A., Bains, T. S., Taggar, G. K., Dikhsit, H. K., Aski, M., Boopathi, M., Swain, D., Rathore, A., Kumar, V. A., Lii, E. C., & Kenyon, L. (2017). Identification of mungbean lines with tolerance or resistance to yellow mosaic in fields in India where different begomovirus species and different Bemisia tabaci cryptic species predominate. European Journal of Plant Pathology, 149, 349–365.
Haq, Q. M. I., Arif, A., & Malathi, V. G. (2010). Engineering resistance against mungbean yellow mosaic India virus using antisense RNA. Indian Journal Virology, 21(1), 82–85.
Kumar, S., Tanti, B., Mukherjee, S. K., & Sahoo, L. (2017). Molecular characterization and infectivity of Mungbean Yellow Mosaic India virus associated with yellow mosaic disease of cowpea and mungbean. Biocatalyst and Agricultural Biotechnology, 11, 183–191.
Karthikeyan, A., Shobhana, V. G., Sudha, M., Raveendran, M., Senthil, N., Pandiyan, M., & Nagarajan, P. (2014). Mung bean yellow mosaic virus (MYMV): A threat to green gram (Vigna radiata) production in Asia. International Journal of Pest Management, 60(4), 314–324.
Bolton, M. D. (2009). Primary metabolism and plant defense–fuel for the fire. Molecular Plant Microbe Interaction, 22(5), 487–497.
Villa-Ruano, N., Valle, R. V., Vallejo, L. G. Z., Hernandez, N. P., Ponce, M. V., Adame, V. M. A., & Mortinez, E. B. (2018). 1H NMR based metabolomics profiling for identification of metabolites in Capsicum annuum cv. Mirasol infected by beet mild curly top virus (BMCTV). Food Research International, 106, 870–877.
Chen, L., Wu, J., Li, Z., Liu, Q., Zhao, Z., & Yang, H. (2019). Metabolomics analysis of energy regulated germination and sprouting of organic mun bean (Vigna radiata) using NMR spectroscopy. Food Chemistry, 15, 87–97.
Srivastava, S., Bisht, H., Sidhu, O. P., Srivastava, A., Singh, P. C., Pandey, R., Raj, S., Roy, R., & Nautiyal, C. (2012). Changes in the metabolome and histopathology of Amaranthus hypochondriacus L. in response to Ageratum enation virus infection. Phytochemistry, 80, 8–16.
Haible, D., Kober, S., & Jeske, H. (2006). Rolling circle amplification revolutionizes diagnosis and genomics of geminiviruses. Journal of Virological Methods, 135(1), 9–16.
Jeffery, S. W., & Humphrey, G. F. (1975). New spectrophotometric equation for determining chlorophylls a, b, c and c2 in higher plants, algae and natural phytoplankton. Biochemie and Physiologie der Pflanzen, 167(2), 191–194.
Chen, L., Sun, D., Zhang, X., Shao, D., Lu, Y., & An, Y. (2021). Transcriptome analysis of yellow passion fruit in response to cucumber mosaic virus infection. PLoS ONE, 16, e0247127.
Zhou, Y., Kim, S. Y., Lee, J. S., Shin, B. K., Seo, J. A., Kim, Y. S., Lee, D. Y., & Choi, H. K. (2021). Discrimination of the geographical origin of soybean using NMR based metabolomics. Foods, 10(2), 435.
Kundu, S., Chakraborty, D., Kumdu, A., & Pal, A. (2013). Proteomics approach combined with biochemical attributes to elucidate compatible and incompatible plant-virus interactions between Vigna mungo and Mungbean Yellow Mosaic India Virus. Proteome Science, 11, 15.
Bhattacharyya, D., Gnanasekaran, P., Kumar, R. K., Kushwaha, N. K., Sharma, V. K., Yusuf, M. A., & Chakraborty, S. (2015). A geminivirus betasatellite damages the structural and functional integrity of chloroplasts leading to symptom formation and inhibition of photosynthesis. Journal of Experimental Botany, 66(19), 5881–5895.
Hameed, S., Akhtar, K. P., Hameed, A., Gulzar, T., Kiran, S., Yousuf, S., Abbas, G., Asghar, M. J., & Sarwar, N. (2017). Biochemical changes in the leaves of mungbean (Vigna radiata) plants infected by phytoplasma. Turkish Journal of Biochemistry, 42(6), 591–599.
Pesti, R., Kontra, L., Paul, K., Vass, I., Csorba, T., Havelda, Z., & Varallyay, E. (2019). Differential gene expression and physiological changes during acute or persistent plant virus interactions may contribute to viral symptom differences. PLoS ONE, 14, e0216618.
Less, H., Angelovici, R., Tzin, V., & Galili, G. (2011). Coordinated gene networks regulating Arabidopsis plant metabolism in response to various stresses and nutritional cues. The Plant Cell, 23(4), 1264–1271.
Shalitin, D., & Wolf, S. (2000). Cucumber mosaic virus infection affected sugar transport in melon plants. Plant Physiology, 123(2), 597–604.
Addy, H. S., Nurmalasari, Wahudi, A. H. S., Sholeh, A., Anugrah, C., Iriyanto, E. E. S., Darmanto, W., & Sugiharto, B. (2017). Detection and response of sugarcane against the infection of sugarcane mosaic virus (SCMV) in Indonesia. Agronomy. 7, 50.
Mandal, R., Kathiria, P., Psychogios, N., Bouatra, S., Krishnamurthy, R., Wishart, D., & Kovalchuk, I. (2012). Progeny of tobacco mosaic virus-infected Nicotiana tabacum plants exhibit trans-generational changes in metabolic profiles. Biocatalyst and Agricultural Biotechnology, 1(2), 115–123.
Zanini, A. A., Feo, L. D., Luna, D. F., Paccioretti, P., Callavino, A., & Rodriguez, M. S. (2020). Cassava common mosaic virus infection causes alterations in chloroplast ultrastructure, function and carbohydrate metabolism of cassava plants. Plant pathology, 70(1), 195–203.
Li, X., An, M., Xia, Z., Bai, X., & Wu, Y. (2017). Transcriptome analysis of watermelon (Citrullus lantus) fruits in response to cucumber green mottle mosaic virus (CGMMV) infection. Scientific Reports, 7, 16747.
Li, L. M., Li, X., Zhao, X. X., Mao, H., Sun, J. H., & Wu, Y. H. (2018). The relationships between sugar metabolism and blood-flesh of watermelon infected with Cucumber green mottle mosaic virus. Journal of Plant Protection, 45(3), 447–454.
Kogovsek, P., Pompe-Novak, M., Petek, M., Fragner, L., Weckwerth, W., & Gruden, K. (2016). Primary metabolism, phenylpropanoids and antioxidant pathways are regulated in potato as a response to potato virus Y infection. PLoS ONE, 11(1), e0146135.
Guo, L., Su, Q., Yin, J., Yang, Z., Xie, W., Wang, S., Wu, Q., Cui, H., & Zhang, Y. (2019). Amino acid utilization may explain why Bemisia tabaci Q and B differ in their performance on plants infected by the tomato yellow leaf curl virus. Frontiers in Physiology, 10, 489.
Tarkowski, L. P., De-Poel, B. V., Hofte, M., & den Ende, W. V. (2019). Sweet immunity: Inulin boosts resistance of lettuce (Lactuca sativa) against grey mold (Botrytis cinerea) in an ethylene-dependent manner. International Journal Molecular Science, 20(5), 1052.
Wang, G., Kong, J., Cui, D., Zhao, H., Niu, Y., Xu, M., Jiang, G., Zhao, Y., & Wang, W. (2018). Resistance against Ralstonia solanacearum in tomato depends on the methionine cycle and the γ-aminobutyric acid metabolic pathway. The Plant Journal, 97(6), 1032–1047.
Yang, J., Sun, C., Zhang, Y., Fu, D., Zheng, X., & Yu, T. (2017). Induced resistance in tomato fruit by γ-aminobutyric acid for the control of Alternaria rot caused by Alternaria alternata. Food Chemistry, 221, 1014–1020.
Miyashita, Y., & Good, A. G. (2008). Contribution of the GABA shunt to hypoxia-induced alanine accumulation in roots of Arabidopsis thaliana. Plant Cell Physiology, 49(1), 92–102.
Qamar, A., Mysore, K. S., & Senthil, K. M. (2015). Role of proline and pyrroline-5-carboxylate metabolism in plant defence against invading pathogens. Frontiers of Plant Science, 6, 503.
Wu, G. (2009). Amino acids, metabolism, functions, and nutrition. Amino Acids, 37, 1–17.
Wang, H., Liu, D., Sun, J., & Zhang, A. (2005). Asparagine synthetase gene TaASN1 from wheat is up-regulated by salt stress, osmotic stress and ABA. Journal of Plant Physiology, 162, 81–89.
Qiao, W., & Fan, L. M. (2008). Nitric oxide signalling in plant responses to abiotic stresses. Journal of Integrative Plant Biology, 50, 1238–1246.
Chuang, C., Prasanth, K. R., & Nagy, P. D. (2017). The glycolytic pyruvate kinase is recruited directly into the viral replicase complex to generate ATP for RNA synthesis. Cell host & microbe, 22(5), 639–652.
Ryšlavá, H., Müller, K., Semorádová, Š, Synková, H., & Čeřovská, N. (2003). Photosynthesis and activity of phosphoenolpyruvate carboxylase in Nicotiana tabacum L. leaves infected by Potato virus A and Potato virus Y. Photosynthetica, 41(3), 357–363.
Funding
This study was supported by Department of Biotechnology, Government of India, for research grant (BT/PR13560/COE/34/44/2015) and Central Instruments Facility, Indian Institute of Technology Guwahati for providing NMR facility.
Author information
Authors and Affiliations
Contributions
Lingaraj Sahoo: conceptualization, experimental design, writing—review and editing. Devendra Kumar Maravi: methodology, investigation, data analysis, writing—original draft, writing—review and editing. Sanjeev Kumar: methodology, writing—original draft, writing—review and editing, formal analysis. All authors have read and agreed for the manuscript.
Corresponding author
Ethics declarations
Ethical Approval
Since this study does not involve use of any animal, human being or cell line as such, there is no ethical issue.
Consent to Participate
Not applicable.
Consent for Publication
Not applicable.
Conflict of Interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Maravi, D.K., Kumar, S. & Sahoo, L. NMR-Based Metabolomic Profiling of Mungbean Infected with Mungbean Yellow Mosaic India Virus. Appl Biochem Biotechnol 194, 5808–5826 (2022). https://doi.org/10.1007/s12010-022-04074-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12010-022-04074-5