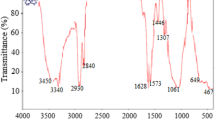
Abstract
Use of nanoparticles as carriers of anticancer drugs is a suitable way for targeted drug delivery and reduction of the side effects. This research focuses on a novel drug carrier for therapeutic goals by the bacterial magnetic nanoparticles (magnetosomes). The unique characteristics of magnetosomes make them ideal nanobiotechnological materials. In this study, magnetic nanoparticles of Alphaproteobacterium MTB-KTN90 were labeled with the radioisotope rhenium-188 and optimized the factors affecting the labeling efficiency. The results showed that the labeling efficiency of magnetosomes with rhenium-188 was more than 96%. The optimum concentration of bacterial nanoparticles was 133 mg/ml and the best time for maximum efficiency labeling was 60 min. The labeling stability showed that the 188Re-nanoparticle complexes have good stability in 29 h. The results of magnetic nanoparticles bacterial cytotoxicity on cancer cells AsPC1 did not show significant toxicity to concentration of 100 μg/μl. Finally, the biogenic magnetic nanoparticles labeled with rhenium-188 can be introduced as a valuable candidate for the targeted therapy of tumor with reducing radiation to surrounding healthy tissues.








Similar content being viewed by others
References
Aghamiri, S. M. R., Akbari-Karadeh, S., Tajer-Mohammad-Ghazvini, P., & Ghorbanzadeh-Mashkani, S. (2018). Effect of temperature and reducing agent on labeling of magnetosomes with 188Re and biodistribution study of labeled magnetic nanoparticles. Modares Journal of Biotechnology, 9, 179–185.
Alphandéry, E. (2014). Applications of magnetosomes synthesized by magnetotactic bacteria in medicine. Frontiers in Bioengineering and Biotechnology, 2, 5.
Alphandéry, E., Guyot, F., & Chebbi, I. (2012). Preparation of chains of magnetosomes, isolated from Magnetospirillum magneticum strain AMB-1 magnetotactic bacteria, yielding efficient treatment of tumors using magnetic hyperthermia. International Journal of Pharmaceutics, 434(1-2), 444–452.
Bazylinski, D. A., Lefèvre, C. T., & Schüler, D. (2013). In E. Rosenberg, E. F. DeLong, S. Lory, E. Stackebrandt, & F. Thompson (Eds.), The prokaryotes (pp. 453–494). Berlin: Springer.
Bouziotis, P., Psimadas, D., Tsotakos, T., Stamopoulos, D., & Tsoukalas, C. (2012). Radiolabeled iron oxide nanoparticles as dual-modality SPECT/MRI and PET/MRI agents. Current Topics in Medicinal Chemistry, 12(23), 2694–2702.
Chunfu, Z., Jinquan, C., Duanzhi, Y., Yongxian, W., Yanlin, F., & Jiajü, T. (2004). Preparation and radiolabeling of human serum albumin (HSA)-coated magnetite nanoparticles for magnetically targeted therapy. Applied Radiation and Isotopes, 61(6), 1255–1259.
Elcey, C., Kuruvilla, A. T., & Thomas, D. (2014). Synthesis of magnetite nanoparticles from optimized iron reducing bacteria isolated from iron ore mining sites. International Journal of Current Microbiology and Applied Sciences, 3, 408–417.
Erdal, E., Demirbilek, M., Yeh, Y., Akbal, Ö., Ruff, L., Bozkurt, D., Cabuk, A., Senel, Y., Gumuskaya, B., Algın, O., Colak, S., Esener, S., & Denkbas, E. B. (2018). A comparative study of receptor-targeted magnetosome and HSA-coated Iron oxide nanoparticles as MRI contrast-enhancing agent in animal cancer model. Applied Biochemistry and Biotechnology, 185(1), 91–113.
Fernandez-Fernandez, A., Manchanda, R., & McGoron, A. J. (2011). Theranostic applications of nanomaterials in cancer: drug delivery, image-guided therapy, and multifunctional platforms. Applied Biochemistry and Biotechnology, 165(7-8), 1628–1651.
Ghorbanzadeh-Mashkani, S., Tajer-Mohammad-Ghazvini, P., Nozad-Golikand, A., Kasra-Kermanshahi, R., & Davarpanah, M.-R. (2013). Synthesis of sterile and pyrogen free biogenic magnetic nanoparticles: biotechnological potential of magnetotactic bacteria for production of nanomaterials. World Academy of Science, Engineering and Technology- International Journal of Biological, Biomolecular, Agricultural, Food and Biotechnological Engineering, World Academy of Science, Engineering and Technology, 7, 133–137.
Grünberg, K., Wawer, C., Tebo, B. M., & Schüler, D. (2001). A large gene cluster encoding several magnetosome proteins is conserved in different species of magnetotactic bacteria. Applied and Environmental Microbiology, 67(10), 4573–4582.
Häfeli, U., Pauer, G., Failing, S., & Tapolsky, G. (2001). Radiolabeling of magnetic particles with rhenium-188 for cancer therapy. Journal of Magnetism and Magnetic Materials, 225(1-2), 73–78.
Huang, F. Y., Lee, T. W., Chang, C. H., Chen, L. C., Hsu, W. H., Chang, C. W., & Lo, J. M. (2015). Evaluation of 188Re-labeled PEGylated nanoliposome as a radionuclide therapeutic agent in an orthotopic glioma-bearing rat model. International Journal of Nanomedicine, 10, 463–473.
Jain, R. (2001). New approaches for the treatment of cancer. Advanced Drug Delivery Reviews, 46(1-3), 149–168.
Koo, O. M., Rubinstein, I., & Onyuksel, H. (2005). Role of nanotechnology in targeted drug delivery and imaging: a concise review. Nanomedicine: nanotechnology, biology and medicine, 1(3), 193–212.
Laan, A. C., Santini, C., Jennings, L., de Jong, M., Bernsen, M. R., & Denkova, A. G. (2016). Radiolabeling polymeric micelles for in vivo evaluation: a novel, fast, and facile method. EJNMMI Research, 6(1), 12–12.
Liang, S., Wang, Y., Yu, J., Zhang, C., Xia, J., & Yin, D. (2007). Surface modified superparamagnetic iron oxide nanoparticles: as a new carrier for bio-magnetically targeted therapy. Journal of Materials Science: Materials in Medicine, 18(12), 2297–2302.
Liu, G., & Hnatowich, D. J. (2007). Labeling biomolecules with radiorhenium: a review of the bifunctional chelators. Anticancer Agents in Medicinal Chemistry, 7(3), 367–377.
Loudos, G., Kagadis, G. C., & Psimadas, D. (2011). Current status and future perspectives of in vivo small animal imaging using radiolabeled nanoparticles. European Journal of Radiology, 78(2), 287–295.
Mathuriya, A. S. (2015). Magnetotactic bacteria for cancer therapy. Biotechnology Letters, 37(3), 491–498.
Matsunaga, T., Suzuki, T., Tanaka, M., & Arakaki, A. (2007). Molecular analysis of magnetotactic bacteria and development of functional bacterial magnetic particles for nano-biotechnology. Trends in Biotechnology, 25(4), 182–188.
Phillips, W. T., Bao, A., Brenner, A. J., & Goins, B. A. (2014). Image-guided interventional therapy for cancer with radiotherapeutic nanoparticles. Advanced Drug Delivery Reviews, 76, 39–59.
Sun, J.-B., Duan, J.-H., Dai, S.-L., Ren, J., Zhang, Y.-D., Tian, J.-S., & Li, Y. (2007). In vitro and in vivo antitumor effects of doxorubicin loaded with bacterial magnetosomes (DBMs) on H22 cells: The magnetic bio-nanoparticles as drug carriers. Cancer Letters, 258(1), 109–117.
Sun, J., Tang, T., Duan, J., Xu, P.-X., Wang, Z., Zhang, Y., Wu, L., & Li, Y. (2010). Biocompatibility of bacterial magnetosomes: acute toxicity, immunotoxicity and cytotoxicity. Nanotoxicology, 4(3), 271–283.
Tajer Mohammad Ghazvini, P. (2014). Isolation of magnetic nanoparticles producer bacteria for evaluation in bioremediation processes, PhD thesis, Alzahra University.
Tajer Mohammad Ghazvini, P., Kasra Kermanshahi, R., Nozad Golikand, A., & Sadeghizadeh, M. (2014). Isolation and characterization of a novel magnetotactic bacterium from Iran: Iron uptake and producing magnetic nanoparticles in Alphaproteobacterium MTB-KTN90. Jundishapur journal of microbiology, 7, e19343.
Truong, N. P., Whittaker, M. R., Mak, C. W., & Davis, T. P. (2015). The importance of nanoparticle shape in cancer drug delivery. Expert Opinion on Drug Delivery, 12(1), 129–142.
Wu, Y., Chu, M., Shi, B., & Li, Z. (2011). A novel magneto-fluorescent nano-bioprobe for cancer cell targeting, imaging and collection. Applied Biochemistry and Biotechnology, 163(7), 813–825.
Xiang, L., Bin, W., Huali, J., Wei, J., Jiesheng, T., Feng, G., & Ying, L. (2007). Bacterial magnetic particles (BMPs)-PEI as a novel and efficient non-viral gene delivery system. The Journal of Gene Medicine, 9(8), 679–690.
Xiang, L., Wei, J., Jianbo, S., Guili, W., Feng, G., & Ying, L. (2007). Purified and sterilized magnetosomes from Magnetospirillum gryphiswaldense MSR-1 were not toxic to mouse fibroblasts in vitro. Letters in Applied Microbiology, 45(1), 75–81.
Yan, L., Zhang, S., Chen, P., Liu, H., Yin, H., & Li, H. (2012). Magnetotactic bacteria, magnetosomes and their application. Microbiological Research, 167(9), 507–519.
Yeong, C.-H., Cheng, M.-H., & Ng, K.-H. (2014). Therapeutic radionuclides in nuclear medicine: current and future prospects. Journal of Zhejiang University Science B, 15(10), 845–863.
Zhang, L., Chen, H., Wang, L., Liu, T., Yeh, J., Lu, G., Yang, L., & Mao, H. (2010). Delivery of therapeutic radioisotopes using nanoparticle platforms: potential benefit in systemic radiation therapy. Nanotechnology, Science and Applications, 3, 159–170.
Zhou, W., Zhang, Y., Ding, X., Liu, Y., Shen, F., Zhang, X., Deng, S., Xiao, H., Yang, G., & Peng, H. (2012). Magnetotactic bacteria: promising biosorbents for heavy metals. Applied Microbiology and Biotechnology, 95(5), 1097–1104.
Acknowledgments
This manuscript was a part of the MSc. thesis by S. Akbari-Karadeh, under the supervision of Dr. S.M.R. Aghamiri and Dr. P. Tajer-Mohammad-Ghazvini; and advisory of S. Ghorbanzadeh-Mashkani. The authors would like to thank Department of Medical Radiation Engineering, Shahid Beheshti University and also Materials and Nuclear Fuel Research School, Nuclear Science and Technology Research Institute, Tehran, Iran for the supports through this study. The authors are grateful to Dr. Reza Dabbagh and Dr. Behrooz Alirezapour for their valuable contributions to this project.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Akbari-Karadeh, S., Aghamiri, S.M.R., Tajer-Mohammad-Ghazvini, P. et al. Radiolabeling of Biogenic Magnetic Nanoparticles with Rhenium-188 as a Novel Agent for Targeted Radiotherapy. Appl Biochem Biotechnol 190, 540–550 (2020). https://doi.org/10.1007/s12010-019-03079-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12010-019-03079-x