Abstract
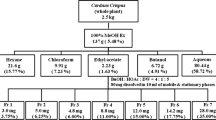
The inhibitory effects of vanadium-binding proteins (VBPs) from the blood plasma and the intestine of sea squirt on adipogenesis in 3T3-L1 adipocytes were examined. 3T3L-1 cells treated with VBP blood plasma decreased markedly the lipid content in maturing pre-adipocytes in a dose-dependent manner, whereas VBP intestine did not show significant effects on lipid accumulation. Both VBPs did not have significant effect on cell viability. In order to demonstrate the anti-adipogenic effects of VBP blood plasma, the expressions of several adipogenic transcription factors and enzymes were investigated by Reverse Transcriptase-Polymerase Chain Reaction. VBP blood plasma down-regulated the expressions of transcription factors; PPAR-γ, C/EBP-α, SREBP1, and FAS, but did not have significant effects on the expressions of lipolytic enzymes; HSL and LPL. Both the crude and purified VBPs significantly increased the mRNA levels of Wnt10b, FZ1, LRP6, and β-catenin, while decreased the expression of GSK-3β. Hence, VBP blood plasma inhibited adipogenesis by activating WNT/β-catenin pathway via the activation of Wnt10b. Based on the findings, VBP blood plasma decreased lipid accumulation which was mediated by decreasing adipogenesis, not by lipolysis. Therefore, VBP blood plasma could be used to treat obesity.






Similar content being viewed by others
References
Siersbæk, R., Nielsen, R., & Mandrup, S. (2012). Transcriptional networks and chromatin remodeling controlling adipogenesis. Trends in Endocrinology and Metabolism, 23(2), 56–64.
Rosen, E. D., & Spiegelman, B. M. (2006). Adipocytes as regulators of energy balance and glucose homeostasis. Nature, 444(7121), 847–853.
Bae, K. H., Kim, W. K., & Lee, S. C. (2012). Involvement of protein tyrosine phosphatases in adipogenesis: New anti-obesity targets? BMB Reports, 45(12), 700–706.
Farmer, S. R. (2008). Molecular determinants of brown adipocyte formation and function. Genes & Development, 22(10), 1269–1275.
Bouzid, T., Hamel, F. G., & Lim, J. Y. (2016). Journal of Diabetes Research, 5, 75–85.
Lefterova, E. D., & Lazar, M. A. (2009). New developments in adipogenesis. Trends in Endocrinology and Metabolism, 20(3), 107–114.
Gregoire, F. M., Smas, C. M., & Sul, H. S. (1998). Understanding Adipocyte Differentiation. Physiological Reviews, 78(3), 783–809.
Rosen, E. D., Hsu, C. H., Wang, X., Sakai, S., Freeman, M. W., Gonzalez, F. J., & Spiegelman, B. M. (2002). C/EBPalpha induces adipogenesis through PPARgamma: a unified pathway. Genes & Development, 16(1), 22–26.
Kim, J. B., Wright, H. M., Wright, M., & Spiegelman, B. M. (1998). ADD1/SREBP1 activates PPAR through the production of endogenous ligand. Proceedings of the National Academy of Sciences of the United States of America, 95(8), 4333–4337.
Wu, Z., Rosen, E. D., Brun, R., Hauser, S., Adelmant, G., Troy, A. E., AcKeon, C., Darlington, G., & Spiegelmsn, B. M. (1999). Molecular Cell, 3(2), 151–158.
Kim, J. B., Wright, H. M., Wright, M., & Spiegelman, B. M. (1998). Genes & Development, 10, 1096–1007.
Yokoyama, C., Spiegelmsn, B. M., Wang, X., Briggs, M. R., Admon, A., Wu, J., Hua, X., Goldstein, J. L., & Brown, M. S. (1993). SREBP-1, a basic-helix-loop-helix-leucine zipper protein that controls transcription of the low density lipoprotein receptor gene. Cell, 75(1), 187–197.
Pessoa, J. C., Garribba, E., Santos, M. F. A., & Santos-Silva, T. (2015). Coordination Chemistry Reviews, 301(302), 49–86.
Yoshihara, M., Ueki, T., Yamaguchi, N., Kaminom, K., & Michibata, H. (2008). Biochimica et Biophysica Acta, 1780, 56–63.
Yoshinaga, M., Ueki, T., & Michibata, H. (2007). Metal binding ability of glutathione transferases conserved between two animal species, the vanadium-rich ascidian Ascidia sydneiensis samea and the schistosome Schistosoma japonicum. Biochimica et Biophysica Acta, 1770(9), 1413–1418.
Ueki, T., Adachi, T., Kawano, S., Aoshima, M., & Yamaguchi, N. (2003). Vanadium-binding proteins (vanabins) from a vanadium-rich ascidian Ascidia sydneiensis samea. Biochimica et Biophysica Acta, 1626(1–3), 43–50.
McNeill, J. H., Yeun, V. G., Dai, S., & Orvig, C. (1995). Increased potency of vanadium using organic ligands. Molecular and Cellular Biochemistry, 153(1-2), 175–180.
Srivastava, A. K. (1999). Molecular and Cell Biology, 206, 177–182.
Liu, Y., Xu, H., Xu, J., Guo, Y., Xue, Y., Wang, J., & Xue, C. (2015). Vanadium-binding protein from vanadium-enriched sea cucumber Apostichopus japonicus inhibits adipocyte differentiation through activating WNT/β-catenin pathway. Journal of Functional Foods, 17, 504–513.
Ueki, T., & Michibata, H. (2011). Molecular mechanism of the transport and reduction pathway of vanadium in ascidians. Coordination Chemistry Reviews, 255(19-20), 2249–2257.
Gunasinghe, M. A., & Kim, S. M. (2018). Antioxidant and antidiabetic activities of vanadium binding proteins purified from the sea squirt Halocynthia roretzi. Journal of Food Science and Technology, 55(5), 1840–1849.
Kruger, N. J. (1994). In methods in molecular biology. In J. M. Walker (Ed.), The Bradford method for protein quantitation (Vol. 32, pp. 9–15). Totowa: Humana.
Laemmli, U. K., & Favre, M. (1973). Maturation of the head of bacteriophage T4. Journal of Molecular Biology, 80(4), 575–592.
Feng, Z., Yu, H. N., Cui, X. M., Wang, Z. C., Shen, S. R., & Das, U. N. (2014). Effect of yellow capsicum extract on proliferation and differentiation of 3T3-L1 preadipocytes. Nutrition, 30(3), 319–325.
Cortizo, A. M., Bruzzone, L., Molinuevo, S., & Etcheverry, S. B. (2000). A possible role of oxidative stress in the vanadium-induced cytotoxicity in the MC3T3E1 osteoblast and UMR106 osteosarcoma cell lines. Toxicology, 147(2), 89–99.
Crans, D. C., Smee, J. J., Gaidamauskas, E., & Yang, L. (2004). The chemistry and biochemistry of vanadium and the biological activities exerted by vanadium compounds. Chemical Reviews, 104(2), 849–902.
Nechay, B. R. (1984). Mechanisms of action of vanadium. Annual Review of Pharmacology and Toxicology, 24(1), 501–524.
Peters, K. G., Davis, M. G., Howard, B. W., Pokross, M., Rastogi, V., Diven, C., Greis, K. D., Eby-Wilkens, E., Maier, M., Evdokimov, A., Soper, S., & Genbauffe, F. (2003). Mechanism of insulin sensitization by BMOV (bis maltolato oxo vanadium); unliganded vanadium (VO4) as the active component. Journal of Inorganic Biochemistry, 96(2-3), 321–330.
Green, H., & Kehinde, O. (1975). An established preadipose cell line and its differentiation in culture II. Factors affecting the adipose conversion. Cell, 5(1), 19–27.
Yeh, W. C., Cao, Z., Classon, M., & McKnight, S. L. (1995). Cascade regulation of terminal adipocyte differentiation by three members of the C/EBP family of leucine zipper proteins. Genes & Development, 9(2), 168–181.
Summers, S. A., Yin, V. P., Whiteman, E. L., Garza, L. A., Cho, H., Tuttle, R. L., & Birnbaum, M. J. (1999). Annals of the New York Academy of Sciences, 892, 169–186.
Farmer, S. R. (2006). Transcriptional control of adipocyte formation. Cell Metabolism, 4(4), 263–273.
Tong, Q., & Hotamisligil, G. S. (2001). Molecular mechanisms of adipocyte differentiation. Reviews in Endocrine & Metabolic Disorders, 2(4), 349–355.
Zechner, R1., Zimmermann, R., Eichmann, T. O., Kohlwein, S. D., Haemmerle, G., Lass, A., & Madeo, F. (2012). FAT SIGNALS - lipases and lipolysis in lipid metabolism and signaling. Cell Metabolism, 15(3), 279–291.
Tontonoz, P., & Spiegelman, B. M. (2008). Fat and beyond: the diverse biology of PPARγ. Annual Review of Biochemistry, 77(1), 289–312.
Kim, S. P., Nam, S. H., & Friedman, M. (2015). Food funct. Annual Review of Biochemistry, 6, 2939–2948.
Linhart, H. G., Ishimura-Oka, K., DeMayo, F., Kibe, T., Repka, D., Poindexter, B., & Darlington, G. J. (2001). C/EBP is required for differentiation of white, but not brown, adipose tissue. Proceedings of the National Academy of Sciences of the United States of America, 98(22), 12532–12537.
Fajas, L., Schoonjans, K., Gelman, L., Kim, J. B., Najib, J., Martin, G., Fruchart, J. C., Briggs, M., Spiegelman, B. M., & Auwerx, J. (1999). Regulation of peroxisome proliferator-activated receptor γ expression by adipocyte differentiation and determination factor 1/sterol regulatory element binding protein 1: implications for adipocyte differentiation and metabolism. Molecular and Cellular Biochemistry, 19(8), 5495–5503.
Clevers, H., Loh, K. M., & Nusse, R. (2014). An integral program for tissue renewal and regeneration: Wnt signaling and stem cell control. Science, 346(6205), 1248012. https://doi.org/10.1126/science.1248012.
Komiya, Y., & Habas, R. (2008). Wnt signal transduction pathways. Organogenesis, 4(2), 68–75.
MacDonald, B. T., Tamai, K., & He, X. (2009). Wnt/β-catenin signaling: components, mechanisms, and diseases. Developmental Cell, 17(1), 9–26.
Pakula, H., Xiang, D., & Li, Z. (2017). A tale of two signals: AR and WNT in development and tumorigenesis of prostate and mammary gland. Cancers, 9(12), 14. https://doi.org/10.3390/cancers9020014.
Liu, J., & Farmer, S. R. (2004). Regulating the balance between peroxisome proliferator-activated receptor γ and β-catenin signaling during adipogenesis. The Journal of Biological Chemistry, 279(43), 45020–45027.
Moldes, M., Zuo, Y., Morrison, R. F., Silva, D., Park, B. H., Liu, J., & Farmer, S. R. (2003). Peroxisome-proliferator-activated receptor γ suppresses Wnt/β-catenin signalling during adipogenesis. The Biochemical Journal, 376(3), 607–613.
Bennett, C. N., Ross, S. E., Longo, K. A., Bajnok, L., Hemati, N., Johnson, K. W., Harrison, S. D., & MacDougald, O. A. (2002). Regulation of Wnt signaling during adipogenesis. The Journal of Biological Chemistry, 277(34), 30998–31004.
Acknowledgements
This research was part of the project entitled “Future Marine Technology Development” funded by the Ministry of Oceans and Fisheries, Republic of Korea.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Gunasinghe, M.A., Kim, A.T. & Kim, S.M. Inhibitory Effects of Vanadium-Binding Proteins Purified from the Sea Squirt Halocynthia roretzi on Adipogenesis in 3T3-L1 Adipocytes. Appl Biochem Biotechnol 189, 49–64 (2019). https://doi.org/10.1007/s12010-019-02982-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12010-019-02982-7