Abstract
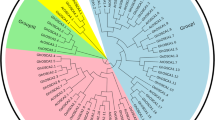
Photosynthetic fixation of CO2 is more efficient in C4 than in C3 plants. Rice is a C3 plant and a potential target for genetic engineering of the C4 pathway. It is known that genes encoding C4 enzymes are present in C3 plants. However, no systematic analysis has been conducted to determine if these C4 gene family members are expressed in diverse rice genotypes. In this study, we identified 15 genes belonging to the five C4 gene families in rice genome through BLAST search using known maize C4 photosynthetic pathway genes. Phylogenetic relationship of rice C4 photosynthetic pathway genes and their isoforms with other grass genomes (Brachypodium, maize, Sorghum and Setaria), showed that these genes were highly conserved across grass genomes. Spatiotemporal, hormone, and abiotic stress specific expression pattern of the identified genes revealed constitutive as well as inductive responses of the C4 photosynthetic pathway in different tissues and developmental stages of rice. Expression levels of C4 specific gene family members in flag leaf during tillering stage were quantitatively analyzed in five rice genotypes covering three species, viz. Oryza sativa, ssp. japonica (cv. Nipponbare), Oryza sativa, ssp. indica (cv IR64, Swarna), and two wild species Oryza barthii and Oryza australiensis. The results showed that all the identified genes expressed in rice and exhibited differential expression pattern during different growth stages, and in response to biotic and abiotic stress conditions and hormone treatments. Our study concludes that C4 photosynthetic pathway genes present in rice play a crucial role in stress regulation and might act as targets for C4 pathway engineering via CRISPR-mediated breeding.




Similar content being viewed by others
Abbreviations
- As:
-
arsenic
- BAP:
-
6-benzylaminopurine
- BSC:
-
bundle sheath cells
- Cd:
-
cadmium
- CDS:
-
coding DNA sequence
- Cr:
-
chromium
- GA3 :
-
gibberellic acid
- IAA:
-
indole-3-acetic acid
- MC:
-
mesophyll cell
- mdh :
-
malate dehydrogenase
- MPSS:
-
massively parallel signature sequencing
- NAA:
-
1-naphthaleneacetic acid
- nadp-me :
-
malic enzyme
- OAA:
-
oxaloacetate
- Pb:
-
lead
- PEP:
-
phosphoenolpyruvate
- PEPC:
-
phosphoenol pyruvate carboxylase
- ppdk :
-
pyruvate orthophosphate dikinase
- RuBisCo:
-
ribulosebishosphate carboxylase/oxygenase
- RUE:
-
radiation use efficiencies
References
Hibberd, J. M., Sheehy, J. E., & Langdale, J. A. (2008). Using C4 photosynthesis to increase the yield of rice-rationale and feasibility. Current Opinion in Plant Biology, 11(2), 228–231. https://doi.org/10.1016/j.pbi.2007.11.002.
Elert, E. (2014). Rice by the numbers: A good grain. Nature, 514(7524), S50–S51. https://doi.org/10.1038/514S50a.
Lobell, D. B., Schlenker, W., & Costa-Roberts, J. (2011). Climate trends and global crop production since 1980. Science, 333(6042), 616–620. https://doi.org/10.1126/science.1204531 (New York, N.Y.).
Welch, J. R., Vincent, J. R., Auffhammer, M., Moya, P. F., Dobermann, A., & Dawe, D. (2010). Rice yields in tropical/subtropical Asia exhibit large but opposing sensitivities to minimum and maximum temperatures. Proceedings of the National Academy of Sciences of the United States of America, 107(33), 14562–14567. https://doi.org/10.1073/pnas.1001222107.
Bansal, K. C., Viret, J. F., Haley, J., Khan, B. M., Schantz, R., & Bogorad, L. (1992). Transient expression from cab-m1 and rbcS-m3 promoter sequences is different in mesophyll and bundle sheath cells in maize leaves. Proceedings of the National Academy of Sciences of the United States of America, 89(8), 3654–3658. https://doi.org/10.1073/pnas.89.8.3654.
Bansal, K. C., & Bogorad, L. (1993). Cell type-preferred expression of maize cab-m1: Repression in bundle sheath cells and enhancement in mesophyll cells. Proceedings of the National Academy of Sciences of the United States of America, 90(9), 4057–4061. https://doi.org/10.1073/pnas.90.9.4057.
Sheehy, J. E., Ferrer, A. B., Mitchell, P. L., Pablico, P., & Dionora, M. J. A. (2008). How the rice crop works and why it needs a new engine. In Charting New Pathways to C4 Rice pp. 3–26. doi:https://doi.org/10.1142/9789812709523_0001
Sage, R. F., Christin, P. A., & Edwards, E. J. (2011). The C4 plant lineages of planet Earth. Journal of Experimental Botany, 62(9), 3155–3169. https://doi.org/10.1093/jxb/err048.
Ludwig, M. (2013). Evolution of the C4 photosynthetic pathway: Events at the cellular and molecular levels. Photosynthesis Research, 117(1–3), 147–161. https://doi.org/10.1007/s11120-013-9853-y.
Kiniry, J. R. R., Jones, C. A. A., O’toole, J. C. C., Blanchet, R., Cabelguenne, M., & Spanel, D. A. A. (1989). Radiation-use efficiency in biomass accumulation prior to grain-filling for five grain-crop species. Field Crops Research, 20(1), 51–64. https://doi.org/10.1016/0378-4290(89)90023-3.
Mitchell, P. L., & Sheehy, J. E. (2006). Supercharging rice photosynthesis to increase yield. New Phytologist, 171(4), 688–693. https://doi.org/10.1111/j.1469-8137.2006.01855.x.
Long, S. P., Zhu, X.-G. G., Naidu, S. L., & Ort, D. R. (2006). Can improvement in photosynthesis increase crop yields? Plant, Cell & Environment, 29(3), 315–330. https://doi.org/10.1111/j.1365-3040.2005.01493.x.
Kajala, K., Covshoff, S., Karki, S., Woodfield, H., Tolley, B. J., Dionora, M. J. A., Mogul, R. T., Mabilangan, A. E., Danila, F. R., Hibberd, J. M., & Quick, W. P. (2011). Strategies for engineering a two-celled C(4) photosynthetic pathway into rice. Journal of Experimental Botany, 62(9), 3001–3010. https://doi.org/10.1093/jxb/err022.
Miyao, M., Masumoto, C., Miyazawa, S. I., & Fukayama, H. (2011). Lessons from engineering a single-cell C4 photosynthetic pathway into rice. Journal of Experimental Botany, 62(9), 3021–3029. https://doi.org/10.1093/jxb/err023.
Langdale, J. A. (2011). C4 cycles: Past, present, and future research on C4 photosynthesis. Plant Cell, 23(11), 3879–3892. https://doi.org/10.1105/tpc.111.092098.
von Caemmerer, S., Quick, W. P., & Furbank, R. T. (2012). The development of C4 rice: Current progress and future challenges. Science, 336(6089), 1671–1672. https://doi.org/10.1126/science.1220177.
Hibberd, J. M., & Quick, W. P. (2002). Characteristics of C4 photosynthesis in stems and petioles of C3 flowering plants. Nature, 415(6870), 451–454. https://doi.org/10.1038/415451a.
Muthusamy, S. K., Singh, S. K., Singh, I., Singh, A. K., Chinnusamy, V., & Bansal, K. C. (2013). Expression analysis of C4 photosynthetic pathway related genes in seven rice genotypes during grain filling stage. Proc Indian Natl Sci Acad, 79(1), 91–98.
Imaizumi, N., Samejima, M., & Ishihara, K. (1997). Characteristics of photosynthetic carbon metabolism of spikelets in rice. Photosynthesis Research, 52(2), 75–82. https://doi.org/10.1023/A:1005887931498.
Rangan, P., Furtado, A., & Henry, R. J. (2016). New evidence for grain specific C4 photosynthesis in wheat. Scientific Reports, 6(1), 31721. https://doi.org/10.1038/srep31721.
Langdale, J. A., & Nelson, T. (1991). Spatial regulation of photosynthetic development in C4 plants. Trends Genet: TIG, 7(6), 191–196. https://doi.org/10.1016/0168-9525(91)90435-S.
Sage, R. F. (2004). The evolution of C4 photosynthesis. New Phytol, 161(2), 341–370. https://doi.org/10.1111/j.1469-8137.2004.00974.x.
Makino, A., Sakuma, I., Sudo, E., & Mae, T. (2003). Differences between maize and rice in N-use efficiency for photosynthesis and protein allocation. Plant and Cell Physiology, 44(9), 952–956. https://doi.org/10.1093/pcp/pcg113.
Raines, C. A. (2011). Increasing photosynthetic carbon assimilation in C3 plants to improve crop yield: Current and future strategies. Plant Physiology, 155(1), 36–42. https://doi.org/10.1104/pp.110.168559.
Matsuoka, M., Furbank, R. T., Fukayama, H., & Miyao, M. (2001). Molecular engineering of C4 photosynthesis. Annual Review of Plant Physiology and Plant Molecular Biology, 52(1), 297–314. https://doi.org/10.1146/annurev.arplant.52.1.297.
Emanuelsson, O., Brunak, S., von Heijne, G., & Nielsen, H. (2007). Locating proteins in the cell using TargetP, SignalP and related tools. Nature Protocols, 2(4), 953–971. https://doi.org/10.1038/nprot.2007.131.
Horton, P., Park, K.-J. J., Obayashi, T., Fujita, N., Harada, H., Adams-Collier, C. J., & Nakai, K. (2007). WoLF PSORT: protein localization predictor. Nucleic Acids Research, 35(Web Server issue), W585–W587. https://doi.org/10.1093/nar/gkm259.
Goodstein, D. M., Shu, S., Howson, R., Neupane, R., Hayes, R. D., Fazo, J., Mitros, T., Dirks, W., Hellsten, U., Putnam, N., & Rokhsar, D. S. (2012). Phytozome: A comparative platform for green plant genomics. Nucleic Acids Research, 40(Database issue), D1178–D1186. https://doi.org/10.1093/nar/gkr944.
Edgar, R. C. (2004). MUSCLE: Multiple sequence alignment with high accuracy and high throughput. Nucleic Acids Research, 32(5), 1792–1797. https://doi.org/10.1093/nar/gkh340.
Letunic, I., Doerks, T., & Bork, P. (2012). SMART 7: Recent updates to the protein domain annotation resource. Nucleic Acids Research, 40(Database issue), D302–D305. https://doi.org/10.1093/nar/gkr931.
Muthusamy, S. K., Dalal, M., Chinnusamy, V., & Bansal, K. C. (2016). Differential regulation of genes coding for organelle and cytosolic ClpATPases under biotic and abiotic stresses in wheat. Frontiers in Plant Science, 7, 929. https://doi.org/10.3389/FPLS.2016.00929.
Katiyar, A., Smita, S., Muthusamy, S. K., Chinnusamy, V., Pandey, D. M., & Bansal, K. C. (2015). Identification of novel drought-responsive microRNAs and trans-acting siRNAs from Sorghum bicolor (L.) Moench by high-throughput sequencing analysis. Frontiers in Plant Science, 6, 506. https://doi.org/10.3389/fpls.2015.00506.
Hruz, T., Laule, O., Szabo, G., Wessendorp, F., Bleuler, S., Oertle, L., et al. (2008). Genevestigator v3: A reference expression database for the meta-analysis of transcriptomes. Advances in Bioinformatics, 2008, 420747–420745. https://doi.org/10.1155/2008/420747.
Muthusamy, S. K., Dalal, M., Chinnusamy, V., & Bansal, K. C. (2017). Genome-wide identification and analysis of biotic and abiotic stress regulation of small heat shock protein (HSP20) family genes in bread wheat. Journal of Plant Physiology, 211, 100–113. https://doi.org/10.1016/j.jplph.2017.01.004.
Nakano, M., Nobuta, K., Vemaraju, K., Tej, S. S., Skogen, J. W., & Meyers, B. C. (2006). Plant MPSS databases: Signature-based transcriptional resources for analyses of mRNA and small RNA. Nucleic Acids Research, 34(90001), D731–D735. https://doi.org/10.1093/nar/gkj077.
Livak, K. J., & Schmittgen, T. D. (2001). Analysis of relative gene expression data using real-time quantitative PCR and the 2(−Delta Delta C(T)) method. Methods, 25(4), 402–408. https://doi.org/10.1006/meth.2001.1262 (San Diego, Calif.).
Tamura, K., Peterson, D., Peterson, N., Stecher, G., Nei, M., & Kumar, S. (2011). MEGA5: Molecular evolutionary genetics analysis using maximum likelihood, evolutionary distance, and maximum parsimony methods. Molecular Biology and Evolution, 28(10), 2731–2739. https://doi.org/10.1093/molbev/msr121.
Price, G. D., von Caemmerer, S., Evans, J. R., Yu, J. W., Lloyd, J., Oja, V., Kell, P., Harrison, K., Gallagher, A., & Badger, M. R. (1994). Specific reduction of chloroplast carbonic anhydrase activity by antisense RNA in transgenic tobacco plants has a minor effect on photosynthetic CO2 assimilation. Planta, 193(3), 331–340. https://doi.org/10.1007/BF00201810.
DiMario, R. J., Clayton, H., Mukherjee, A., Ludwig, M., & Moroney, J. V. (2017). Plant carbonic anhydrases: Structures, locations, evolution, and physiological roles. Molecular Plant, 10(1), 30–46. https://doi.org/10.1016/j.molp.2016.09.001.
Moroney, J. V., Bartlett, S. G., & Samuelsson, G. (2001). Carbonic anhydrases in plants and algae: Invited review. Plant, Cell and Environment, 24(2), 141–153. https://doi.org/10.1046/j.1365-3040.2001.00669.x.
Wang, X., Gowik, U., Tang, H., Bowers, J., Westhoff, P., & Paterson, A. (2009). Comparative genomic analysis of C4 photosynthetic pathway evolution in grasses. Genome Biology, 10(6), R68. https://doi.org/10.1186/gb-2009-10-6-r68.
Vidal, J., & Chollet, R. (1997). Regulatory phosphorylation of C4 PEP carboxylase. Trends in Plant Science, 2(6), 230–237. https://doi.org/10.1016/S1360-1385(97)01046-7.
Masumoto, C., Miyazawa, S.-I. S.-I., Ohkawa, H., Fukuda, T., Taniguchi, Y., Murayama, S., et al. (2010). Phosphoenolpyruvate carboxylase intrinsically located in the chloroplast of rice plays a crucial role in ammonium assimilation. Proceedings of the National Academy of Sciences, 107(11), 5226–5231. https://doi.org/10.1073/pnas.0913127107.
Chollet, R., Vidal, J., & O’Leary, M. H. (1996). Pyruvate carboxylase: A ubiquitous, highly regulated enzyme in plants. Annual Review of Plant Physiology and Plant Molecular Biology, 47(1), 273–298. https://doi.org/10.1146/annurev.arplant.47.1.273.
Sánchez, R., & Cejudo, F. (2003). Identification and expression analysis of a gene encoding a bacterial-type phosphoenolpyruvate carboxylase from Arabidopsis and rice. Plant Physiology, 132(June), 949–957. https://doi.org/10.1104/pp.102.019653.1997.
Doubnerová, V., & Ryšlavá, H. (2011). What can enzymes of C4 photosynthesis do for C3 plants under stress? Plant Science, 180(4), 575–583. https://doi.org/10.1016/j.plantsci.2010.12.005.
Chi, W., Yang, J., Wu, N., & Zhang, F. (2004). Four rice genes encoding NADP malic enzyme exhibit distinct expression profiles. Bioscience, Biotechnology, and Biochemistry, 68(9), 1865–1874. https://doi.org/10.1271/bbb.68.1865.
Cairoll, L. J., Dunaway-Mariano, D., Smith, C. M., & Chollet, R. (1990). Determination of the catalytic pathway of C4-leaf pyruvate, orthophosphate dikinase from maize. FEBS Letters, 274(1–2), 178–180. https://doi.org/10.1016/0014-5793(90)81358-U.
Parsley, K., & Hibberd, J. M. (2006). The Arabidopsis PPDK gene is transcribed from two promoters to produce differentially expressed transcripts responsible for cytosolic and plastidic proteins. Plant Molecular Biology, 62(3), 339–349. https://doi.org/10.1007/s11103-006-9023-0.
Dong, X., Li, Y., Chao, Q., Shen, J., Gong, X., Zhao, B., & Wang, B. (2016). Analysis of gene expression and histone modification between C4 and non-C4 homologous genes of PPDK and PCK in maize. Photosynthesis Research, 129(1), 71–83. https://doi.org/10.1007/s11120-016-0271-9.
Hibberd, J. M., & Covshoff, S. (2010). The regulation of gene expression required for C4 photosynthesis. Annual Review of Plant Biology, 61(1), 181–207. https://doi.org/10.1146/annurev-arplant-042809-112238.
Aubry, S., Brown, N. J., & Hibberd, J. M. (2011). The role of proteins in C3 plants prior to their recruitment into the C4 pathway. Journal of Experimental Botany, 62(9), 3049–3059. https://doi.org/10.1093/jxb/err012.
Wang, Q., Zhang, Q., Fan, D., & Lu, C. (2006). Photosynthetic light and CO2 utilization and C4 traits of two novel super-rice hybrids. Journal of Plant Physiology, 163(5), 529–537. https://doi.org/10.1016/j.jplph.2005.04.035.
Wang, P., Vlad, D., & Langdale, J. A. (2016). Finding the genes to build C4 rice. Current Opinion in Plant Biology, 31, 44–50. https://doi.org/10.1016/j.pbi.2016.03.012.
Xu, J., Bräutigam, A., Weber, A. P. M., & Zhu, X. G. (2016). Systems analysis of cis-regulatory motifs in C4photosynthesis genes using maize and rice leaf transcriptomic data during a process of de-etiolation. Journal of Experimental Botany, 67(17), 5105–5117. https://doi.org/10.1093/jxb/erw275.
Chang, T. T. (1976). The origin, evolution, cultivation, dissemination, and diversification of Asian and African rices. Euphytica, 25(1), 425–441. https://doi.org/10.1007/BF00041576.
Khush, G. S. (1997). Origin, dispersal, cultivation and variation of rice. Plant Molecular Biology, 35(1/2), 25–34. https://doi.org/10.1023/A:1005810616885.
Tsuzuki, M., Miyachi, S., & Edwards, G. E. (1985). Localization of carbonic anhydrase in mesophyll cells of terrestrial C3 plants in relation to CO2 assimilation. Plant and Cell Physiology, 26(5), 881–891. https://doi.org/10.1093/oxfordjournals.pcp.a076983.
Rumeau, D., Cuine, S., Fina, L., Gault, N., Nicole, M., & Peltier, G. (1996). Subcellular distribution of carbonic anhydrase in Solanum tuberosum L leaves—Characterization of two compartment-specific isoforms. Planta, 199(1), 79–88. https://doi.org/10.1007/BF00196884.
Yu, S., Zhang, X., Guan, Q., Takano, T., & Liu, S. (2007). Expression of a carbonic anhydrase gene is induced by environmental stresses in rice (Oryza sativa L.). Biotechnology Letters, 29(1), 89–94. https://doi.org/10.1007/s10529-006-9199-z.
Matsuoka, M., & Hata, S. (1987). Comparative studies of phosphoenolpyruvate carboxylase from c(3) and c(4) plants. Plant Physiology, 85(4), 947–951.
Yeo, M. E., Yeo, A. R., & Flowers, T. J. (1994). Photosynthesis and photorespiration in the genus Oryza. Journal of Experimental Botany, 45(5), 553–560. https://doi.org/10.1093/jxb/45.5.553.
Cornic, G., Bukhov, N. G., Wiese, C., Bligny, R., & Heber, U. (2000). Flexible coupling between light-dependent electron and vectorial proton transport in illuminated leaves of C3 plants. Role of photosystem I-dependent proton pumping. Planta, 210(3), 468–477. https://doi.org/10.1007/PL00008154.
Saccardy, K., Cornic, G., Brulfert, J., & Reyss, A. (1996). Effect of drought stress on net CO2 uptake by Zea leaves. Planta, 199(4), 589–595. https://doi.org/10.1007/BF00195191.
Bandyopadhyay, A., Datta, K., Zhang, J., Yang, W., Raychaudhuri, S., Miyao, M., & Datta, S. K. (2007). Enhanced photosynthesis rate in genetically engineered indica rice expressing pepc gene cloned from maize. Plant Science, 172(6), 1204–1209. https://doi.org/10.1016/j.plantsci.2007.02.016.
Cheng, Y., & Long, M. (2007). A cytosolic NADP-malic enzyme gene from rice (Oryza sativa L.) confers salt tolerance in transgenic Arabidopsis. Biotechnology Letters, 29(7), 1129–1134. https://doi.org/10.1007/s10529-007-9347-0.
Brar, D. S., & Khush, G. S. (2006). Cytogenetic manipulation and germplasm enhancement of rice (Oryza sativa L.). In: Singh RJ, Jauhar PP (eds) Genetic resources, chromosome engineering and crop improvement. CRC Press, Boca Raton, FL, 115–158. https://doi.org/10.1201/9780203489260.ch5.
Liu, S., Cheng, Y., Zhang, X., Guan, Q., Nishiuchi, S., Hase, K., & Takano, T. (2007). Expression of an NADP-malic enzyme gene in rice (Oryza sativa. L) is induced by environmental stresses; over-expression of the gene in Arabidopsis confers salt and osmotic stress tolerance. Plant Molecular Biology, 64(1–2), 49–58. https://doi.org/10.1007/s11103-007-9133-3.
Moons, A., Valcke, R., & Van Montagu, M. (1998). Low-oxygen stress and water deficit induce cytosolic pyruvate orthophosphate dikinase (PPDK) expression in roots of rice, a C3 plant. Plant Journal, 15(1), 89–98. https://doi.org/10.1046/j.1365-313X.1998.00185.x.
Henry, R. J., Rice, N., Waters, D. L. E., Kasem, S., Ishikawa, R., Hao, Y., Dillon, S., Crayn, D., Wing, R., & Vaughan, D. (2010). Australian Oryza: Utility and conservation. Rice, 3(4), 235–241. https://doi.org/10.1007/s12284-009-9034-y.
Wang, L., Czedik-Eysenberg, A., Mertz, R. A., Si, Y., Tohge, T., Nunes-Nesi, A., Arrivault, S., Dedow, L. K., Bryant, D. W., Zhou, W., Xu, J., Weissmann, S., Studer, A., Li, P., Zhang, C., LaRue, T., Shao, Y., Ding, Z., Sun, Q., Patel, R. V., Turgeon, R., Zhu, X., Provart, N. J., Mockler, T. C., Fernie, A. R., Stitt, M., Liu, P., & Brutnell, T. P. (2014). Comparative analyses of C4 and C3 photosynthesis in developing leaves of maize and rice. Nature Biotechnology, 32(11), 1158–1165. https://doi.org/10.1038/nbt.3019.
Reyna-Llorens, I., Burgess, S. J., Reeves, G., Singh, P., Stevenson, S. R., Williams, B. P., Stanley, S., & Hibberd, J. M. (2018). Ancient duons may underpin spatial patterning of gene expression in C4 leaves. Proceedings of the National Academy of Sciences, 115(8), 1931–1936. https://doi.org/10.1073/pnas.1720576115.
Heimann, L., Horst, I., Perduns, R., Dreesen, B., Offermann, S., & Peterhansel, C. (2013). A common histone modification code on C4 genes in maize and its conservation in Sorghum and Setaria italica. Plant Physiology, 162(1), 456–469. https://doi.org/10.1104/pp.113.216721.
Reeves, G., Grangé-Guermente, M. J., & Hibberd, J. M. (2017). Regulatory gateways for cell-specific gene expression in C4 leaves with Kranz anatomy. Journal of Experimental Botany, 68(2), 107–116. https://doi.org/10.1093/jxb/erw438.
Acknowledgements
This work was supported by the Indian Council of Agricultural Research (ICAR)-sponsored National Agricultural Innovation Project (NAIP) project. SKM gratefully acknowledges the Indian Agricultural Research Institute (IARI) for IARI-Junior Research Fellowship grant and Department of Science and Technology (DST), Government of India for DST-INSPIRE fellowship grant. The authors acknowledge National Phytotron Facility, IARI, for providing space for growing plants.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Muthusamy, S.K., Lenka, S.K., Katiyar, A. et al. Genome-Wide Identification and Analysis of Biotic and Abiotic Stress Regulation of C4 Photosynthetic Pathway Genes in Rice. Appl Biochem Biotechnol 187, 221–238 (2019). https://doi.org/10.1007/s12010-018-2809-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12010-018-2809-0