Abstract
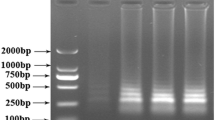
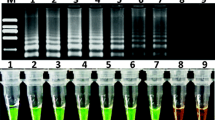
Genetically modified (GM) livestock have the potential to contribute to improving the environment and human health, with consumption of fewer resources and reduced waste production. However, the transgene process also poses risks. The safety assessment and control of transgenic animal products have drawn wide attention, and the relevant regulations and technology are being developed. Quick testing technology plays a significant role in on-site and customs sampling. Nowadays, loop-mediated isothermal amplification (LAMP) was widely applied in nucleic acid analysis because of its simplicity, rapidity, high efficiency and specificity. In this study, a specific, sensitive detection system for detecting sFAT-1 transgenic pigs was designed. A set of six primers including two loop primers was designed for the target sequence. The DNA samples were amplified in less than 1 h at the optimized temperature and detecting by both Nephelometer LA-320c and unaided eyes directly adding calcein. The detection limit of sFAT-1 LAMP was as low as 1.26 ng/μL. Furthermore, blind tests of transgenic and non-transgenic DNA samples were all correctly detected. Hence, the results in this study demonstrated that LAMP is a very useful tool for transgenic detection.





Similar content being viewed by others
References
Yashodhara, B. M., Umakanth, S., Pappachan, J. M., et al. (2009). Omega-3 fatty acids: a comprehensive review of their role in health and disease. Postgraduate Medical Journal, 85, 84–90.
Goldberg, R. J., & Katz, J. (2007). A meta-analysis of the analgesic effects of omega-3 polyunsaturated fatty acid supplementation for inflammatory joint pain. Pain, 129, 210–223.
Kabir, M., Skurnik, G., Naour, N., et al. (2007). Treatment for 2 mo with n 3 polyunsaturated fatty acids reduces adiposity and some atherogenic factors but does not improve insulin sensitivity in women with type 2 diabetes: a randomized controlled study. American Journal of Clinical Nutrition, 86, 1670–1679.
Kuriki, K., Hirose, K., Wakai, K., et al. (2007). Breast cancer risk and erythrocyte compositions of n-3 highly unsaturated fatty acids in Japanese. International Journal of Cancer, 121, 377–385.
Puri, B. K., Leavitt, B. R., Hayden, M. R., et al. (2005). Ethyl-EPA in Huntington disease: a double-blind, randomized, placebocontrolled trial. Neurology, 65, 286–292.
Von Schacky, C., Angerer, P., Kothny, W., et al. (1999). The effect of dietary omega-3 fatty acids on coronary atherosclerosis. A randomized, double-blind, placebo-controlled trial. Annals of Internal Medicine, 130, 554–562.
Tang, M., Qian, L., Jiang, S., et al. (2014). Functional and safety evaluation of transgenic pork rich in omega-3 fatty acids. Transgenic Research, 23, 557–571.
Spychalla, J. P., Kinney, A. J., & Browse, J. (1997). Identification of an animal omega-3 fatty acid desaturase by heterologous expression in Arabidopsis. Proceedings of the National Academy of Sciences of the United States of America, 94(4), 1142–1147.
Tang, M. X., Zheng, X. M., Cheng, W. K., et al. (2011). Safety assessment of sFat-1 transgenic pigs by detecting their co-habitant microbe in intestinal tract. Translational Research, 20, 749–758.
Singh, R. K., Singh, V. K., Raghavendrarao, S., et al. (2015). Expression of finger millet EcDehydrin7 in transgenic tobacco confers tolerance to drought stress. Applied Biochemistry and Biotechnology, 177, 207–216.
Zhu, H., Wen, F., & Li, P. (2014). Validation of a reference gene (BdFIM) for quantifying transgene copy numbers in brachypodium distachyon by real-time PCR. Applied Biochemistry and Biotechnology, 172, 3163–3175.
Wang, X., Jiang, D., & Yang, D. (2014). Fast-tracking determination of homozygous transgenic lines and transgene stacking using a reliable quantitative real-time PCR assay. Applied Biochemistry and Biotechnology, 175, 996–1006.
Tian, F., Guan, Q., Wang, X., et al. (2015). Influence of different processing treatments on the detectability of nucleic acid and protein targets in transgenic soybean meal. Applied Biochemistry and Biotechnology, 172, 3686–3700.
Lee, D., La Mura, M., Allnutt, T. R., et al. (2009). Detection of genetically modified organisms (GMOs) using isothermal amplification of target DNA sequences. BMC Biotechnology, 9, 7.
Notomi, T., Okayama, H., Masubuchi, H., et al. (2000). Loop-mediated isothermal amplification of DNA. Nucleic Acids Research, 28(12), E63.
Lee, D. K., Kim, E. J., & Kilgore, P. E. (2015). Clinical evaluation of a loop-mediated isothermal amplification (LAMP) assay for rapid detection of Neisseria meningitidis in cerebrospinal fluid. PLoS One, 10(4), e0122922.
Notomi, T., Mori, Y., & Tomita, N. (2014). Loop-mediated isothermal amplification (LAMP): principle, features, and future prospects. Journal of Microbiology, 53(1), 1–5.
Liu, M., Luo, Y., Tao, R., He, R., Jiang, K., Wang, B., & Wang, L. (2009). Sensitive and rapid detection of genetic modified soybean (Roundup Ready) by loop-mediated isothermal amplification. Bioscience Biotechnology and Biochemistry, 73, 2365–2369.
Zhai, S., Liu, C., Zhang, Q., Tao, C., & Liu, B. (2012). Detection of two exogenous genes in transgenic cattle by loop-mediated isothermal amplification. Transgenic Research, 21(6), 1367–1373.
Nagamine, K., Hase, T., & Notomi, T. (2002). Accelerated reaction by loop-mediated isothermal amplification using loop primers. Molecular and Cellular Probes, 16, 223–229.
Hu, M., Guo, Y., & Chen, H. (2013). Exon array analysis of alternative splicing of genes in SOD1G93A transgenic mice. Applied Biochemistry and Biotechnology, 170, 301–319.
Mori, Y., Hirano, T., & Notomi, T. (2006). Sequence specific visual detection of LAMP reactions by addition of cationic polymers. BMC Biotechnology, 6, 3. doi:10.1186/1472-6750-6-3.
Tao, C., Zhang, Q., Zhai, S., & Liu, B. (2013). Detection of HbsAg and hATIII genetically modified goats (Caprahircus) by loop-mediated isothermal amplification. Molecular Biology Reports, 40(11), 6177–6182.
Acknowledgments
This work was supported by the National Natural Science Foundation of China (31501931), the 948 Program of China (2011-G11), and the Agricultural Science and Technology Innovation Program (ASTIP-IAS05).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Tao, C., Yang, Y., Li, X. et al. Rapid and Sensitive Detection of sFAT-1 Transgenic Pigs by Visual Loop-Mediated Isothermal Amplification. Appl Biochem Biotechnol 179, 938–946 (2016). https://doi.org/10.1007/s12010-016-2041-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12010-016-2041-8