Abstract
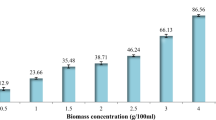
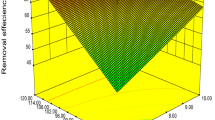
Biosorption of manganese(II) using suspended and immobilized cells of fungal Penicillium camemberti (biomass) and nano-P. camemberti (nano-biomass) was studied by evaluating the physicochemical parameters of the solution such as initial manganese ion concentration, pH, temperature, dosage, and contact time in both batch system and fixed bed column. The maximum biosorption obtained from the batch process was 91.54 and 71.08 % for nano-biomass and biomass in initial concentration of 5 ppm, respectively. The Langmuir, Freundlich, Temkin, and BET isotherms isotherm models were used in the equilibrium modeling. The correlation coefficient of more than 0.90 turned out that the adsorption process of Mn(II) on biomass and nano-biomass were in accordance with both Langmuir and Freundlich isotherms. The sorption process followed a second-order rate kinetics indicating the process to be diffusion controlled. The results also demonstrate that an intra-particle diffusion mechanism plays a significant role in the sorption process. The structure of P. camemberti was characterized by FT-IR spectrums.







Similar content being viewed by others
References
Kapoor, A., Viraraghavan, T., & Cullimore, D. R. (1999). Bioresource Technology, 70, 95–104.
Zouboulis, A. I., Loukidou, M. X., & Matis, K. I. (2004). Process Biochemistry, 39, 909–916.
Ahluwalia, S. S., & Goyal, D. (2007). Bioresource Technology, 98, 2243–2257.
Mahmoud, M. E., Yakout, A. A., Abdel-Aal, H., & Osman, M. M. (2011). Desalination, 279, 291–297.
Mukhopadhyay, M., Noronha, S. B., & Suraishkumar, G. K. (2007). Bioresource Technology, 98, 1781–1787.
Yang, Y., Wang, G., Wang, B., Li, Z., Jia, X., Zhou, Q., & Zhao, Y. (2011). Bioresource Technology, 102, 828–834.
Silva, R. M. P., Rodrguez, A. A., De-Oca, J. M. G. M., & Moreno, D. C. (2009). Bioresource Technology, 100, 1533–1538.
Marandi, R. (2011). Canadian Journal on Chemical Engineering & Technology, 2, 8–22.
Han, R., Zhang, J., Zou, W., Xiao, H., Shi, J., & Liu, H. (2006). Journal of Hazardous Materials, B133, 262–268.
Cojocaru, V. D. (2012). Journal of Magnetism and Magnetic Materials, 324, 1664–1669.
Vasoya, N. H., Vanpariya, L. H., Sakariya, P. N., Timbadiya, M. D., Pathak, T. K., Lakhani, V. K., & Modi, K. B. (2010). Ceramics International, 36, 947–954.
Preetha, B., & Viruthagiri, T. (2007). Separation and Purification Technology, 57, 126–133.
Bunluesin, S., Kruatrachue, M., Pokethitiyook, P., Upatham, S. R., & Lanza, G. (2007). Bioscience and Bioengineering, 103, 509–513.
Padmavathy, V. (2008). Bioresource Technology, 99, 3100–3109.
Yalcin, E., Cavusoglu, K., & Kınalıoglu, K. (2010). Environmental Sciences, 22, 367–373.
Yang, C., Wang, J., Lei, M., Xie, G., Zeng, G., & Luo, S. (2010). Environmental Sciences, 22, 675–680.
Malkoc, E. (2006). Hazardous Materials, B137, 899–908.
Parvathi, K., Nareshkumar, R., & Nagendran, R. (2007). Microbiol Biotechnol, 23, 671–676.
Asadi, F., Shariatmadari, H., & Mirghaffari, N. (2008). Hazardous Materials, 154, 451–458.
Park, D., Yun, Y. S., Jo, J. H., & Park, J. M. (2005). Water Research, 39, 533–540.
Vijayaraghavan, K., Teo, T. T., Balasubramanian, R., & Joshi, U. M. (2009). Hazardous Materials, 164, 1019–1023.
Yan, Z., Xuliang, F., Zhilong, Y., Yahong, L., & Weimin, C. (2008). Environmental Sciences, 20, 1288–1293.
Joo, J. H., Hassan, S. H. A., & Oh, S. E. (2010). International Biodeterioration & Biodegradation, 64, 734–741.
Bulgariu, D., & Bulgariu, L. (2012). Bioresource Technology, 103, 489–493.
Xin, X., Wei, Q., Yang, J., Yan, L., Feng, R., Chen, G., Du, B., & Li, H. (2012). Chemical Engineering Journal, 184, 132–140.
Freitas, O. M. M., Martins, R. J. E., Delerue-Mato, C. M., & Boaventura, R. A. R. (2008). Hazardous Materials, 153, 493–501.
Azila Yahaya, Y., Mat Don, M., & Bhatia, S. (2009). Journal of Hazardous Materials, 161, 189–195.
Chen, Z., Ma, W., & Han, M. (2008). Hazardous Materials, 155, 327–333.
Li, W., Zhang, L. B., Peng, J. H., Li, N., & Zhu, X. Y. (2008). Industrial Crops and Products, 27, 341–347.
Mungasavalli, D. P., Viraraghavan, T., & Jin, Y. C. (2007). Colloids and Surfaces A: Physicochemical and Engineering Aspects, 301, 214–223.
Kilic, M., Keskin, M. E., Mazlum, S., & Mazlum, N. (2008). International Journal of Mineral Processing, 87, 1–8.
Han, R., Wang, Y., Yu, W., Zou, W., Shi, J., & Liu, H. (2007). Hazardous Materials, 141, 713–718.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Khalilnezhad, R., Olya, M.E., Khosravi, M. et al. Manganese Biosorption from Aqueous Solution by Penicillium camemberti Biomass in the Batch and Fix Bed Reactors: A Kinetic Study. Appl Biochem Biotechnol 174, 1919–1934 (2014). https://doi.org/10.1007/s12010-014-1076-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12010-014-1076-y