Abstract
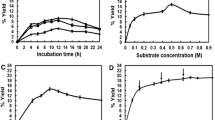
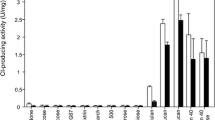
A putative α-amylase gene (accession number, CP000284) of Methylobacillus flagellatus KT ATCC51484 was cloned in Escherichia coli, and its gene product was expressed and characterized. The purified recombinant enzyme (MFAS) displayed a typical amylosucrase (ASase) activity by the demonstration of multiple activities of hydrolysis, isomerization, and polymerization although it was designated as an α-amylase. The optimal reaction temperature and pH for the sucrose hydrolysis activity of MFAS were determined to be 45 °C and pH 8.5, respectively. MFAS has relatively high thermostable characteristics compared with other ASases, as demonstrated by a half-life of 19.3 min at 50 °C. MFAS also showed polymerization activity using sucrose as a sole substrate. Glycerol was transglycosylated by the intermolecular transglycosylation activity of MFAS. Two major products were observed by thin-layer chromatography and isolated by paper chromatography and recycling HPLC. Using 1H and 13C NMR, their chemical structures were determined to be (2S)-1-O-α-d-glucosyl-glycerol or (2R)-1-O-α-d-glucosyl-glycerol and 2-O-α-d-glucosyl-glycerol, in which a glucose molecule is linked to glycerol via an α-glycosidic linkage.





Similar content being viewed by others
References
Davies, G., & Henrissat, B. (1995). Structure, 3, 853–859.
van der Maarel, M. J. E. C., van der Veen, B., Uitdehaag, J. C. M., Leemhuis, H., & Dijkhuizen, L. (2002). Journal of Biotechnology, 94, 137–155.
Bertoldo, C., & Antranikian, G. (2002). Current Opinion in Chemical Biology, 6, 151–160.
Seo, D.H., Jung, J.H., Ha, S.J., Yoo, S.H., Kim, T.J., Cha, J., & Park, C.S. (2008). Carbohydrate-active enzyme structure, function and application (Park, K.H., ed.), CRC press, Boca Raton, pp. 125–140.
Albenne, C., Skov, L. K., Mirza, O., Gajhede, M., Feller, G., D'Amico, S., et al. (2004). The Journal of Biological Chemistry, 279, 726–734.
Wang, R., Bae, J.S., Kim, J.H., Kim, B.S., Yoon, S.H., Park, C.S., & Yoo, S.H. (2012). Food Chemistry. 132, 773–779
Park, H., Choi, K., Park, Y. D., Park, C. S., & Cha, J. (2011). Journal of Life Science, 21, 1631–1635.
Daudé, D., Champion, E., Morel, S., Guieysse, D., Remaud-Siméon, M., & André, I. (2013). ChemCatChem, 5, 2288–2295.
Jung, J.H., Seo, D.H., Ha, S.J., Song, M.C., Cha, J., Yoo, S.H., Kim, T.J., Baek, N.I., Baik, M.Y., & Park, C.S. (2009). Carbohydrate Research. 344, 1612–1619
Seo, D.H., Jung, J.H., Ha, S.J., Song, M.C., Cha, J., Yoo, S.H., Kim, T.J., Baek, N.I., & Park, C.S. (2009). Journal of Molecular Catalysis B: Enzymatic. 60, 113–118
Cho, H. K., Kim, H. H., Seo, D. H., Jung, J. H., Park, J. H., Baek, N. I., et al. (2011). Enzyme Microbial Technology, 49, 246–253.
Seo, D. H., Jung, J. H., Ha, S. J., Cho, H. K., Jung, D. H., Kim, T. J., et al. (2012). Applied Microbiology and Biotechnology, 94, 1189–1197.
Potocki De Montalk, G., Remaud-Simeon, M., Willemot, R. M., Planchot, V., & Monsan, P. (1999). Journal of Bacteriology, 181, 375–381.
Pizzut-Serin, S., Potocki-Véronèse, G., van der Veen, B. A., Albenne, C., Monsan, P., & Remaud-Simeon, M. (2005). FEBS Letters, 579, 1405–1410.
Ha, S. J., Seo, D. H., Jung, J. H., Cha, J., Kim, T. J., Kim, Y. W., et al. (2009). Bioscience Biotechnology and Biochemistry, 73, 1505–1512.
Seo, D. H., Choi, H. C., Kim, H. H., Yoo, S. H., & Park, C. S. (2012). Journal of Microbiology and Biotechnology, 22, 1253–1257.
Kim, H. S., Park, H. J., Heu, S., & Jung, J. (2004). Journal of Bacteriology, 186, 411–418.
Kim, M. I., Kim, H. S., Jung, J., & Rhee, S. (2008). Journal of Molecular Biology, 380, 636–647.
Mikkat, S., Hagemann, M., & Schoor, A. (1996). Microbiology, 142, 1725–1732.
Marin, K., Zuther, E., Kerstan, T., Kunert, A., & Hagemann, M. (1998). Journal of Bacteriology, 180, 4843–4849.
Takenaka, F., Uchiyama, H., & Imamura, T. (2000). Bioscience Biotechnology and Biochemistry, 64, 378–385.
Sawai, T., & Hehre, E. J. (1962). The Journal of Biological Chemistry, 237, 2047–2052.
Takenaka, F., & Uchiyama, H. (2000). Bioscience Biotechnology and Biochemistry, 64, 1821–1826.
Smith, P. K., Krohn, R. I., Hermanson, G. T., Mallia, A. K., Gartner, F. H., Provenzano, M. D., et al. (1985). Analytical Biochemistry, 150, 76–85.
Niesen, F. H., Berglund, H., & Vedadi, M. (2007). Nature Protocols, 2, 2212–2221.
Skov, L. K., Mirza, O., Henriksen, A., De Montalk, G. P., Remaud-Simeon, M., Sarçabal, P., et al. (2001). The Journal of Biological Chemistry, 276, 25273–25278.
Guérin, F., Barbe, S., Pizzut-Serin, S., Potocki-Véronèse, G., Guieysse, D., Guillet, V., et al. (2012). The Journal of Biological Chemistry, 287, 6642–6654.
Schneider, J., Fricke, C., Overwin, H., Hofmann, B., & Hofer, B. (2009). Applied and Environmental Microbiology, 75, 7453–7460.
Nakano, H., Kiso, T., Okamoto, K., Tomita, T., Bin Abdul Manan, M., & Kitahata, S. (2003). Journal of Bioscience and Bioengineering, 95, 583–588.
Goedl, C., Sawangwan, T., Mueller, M., Schwarz, A., & Nidetzky, B. (2008). Angewandte Chemie, International Edition, 47, 10086–10089.
Schwarz, A., Thomsen, M. S., & Nidetzky, B. (2009). Biotechnology and Bioengineering, 103, 865–872.
Hinz, S. W. A., Verhoef, R., Schols, H. A., Vincken, J. P., & Voragen, A. G. J. (2005). Carbohydrate Research, 340, 2135–2143.
Masuda, T., Kawano, A., Kitahara, K., Nagashima, K., Aikawa, Y., & Arai, S. (2003). Journal of Nutritional Science and Vitaminology . (Tokyo) 49, 64–68.
Wei, W., Qi, D., Zhao, H., Lu, Z., Lv, F., & Bie, X. (2013). Food Chemistry, 141, 3085–3092.
Suhr, R., Scheel, O., & Thiem, J. (1998). Journal of Carbohydrate Chemistry, 17, 937–968.
Goedl, C., Sawangwan, T., Wildberger, P., & Nidetzky, B. (2010). Biocatalysis and Biotransformation, 28, 10–21.
Seo, S., Tomita, Y., Tori, K., & Yoshimura, Y. (1978). Journal of the American Chemical Society, 100, 3331–3339.
Cassel, S., Debaig, C., Benvegnu, T., Chaimbault, P., Lafosse, M., Plusquellec, D., et al. (2001). European Journal of Organic Chemistry, 2001, 875–896.
He, F., Hogan, S., Latypov, R. F., Narhi, L. O., & Razinkov, V. I. (2010). Journal of Pharmaceutical Sciences, 99, 1707–1720.
Goldberg, D. S., Bishop, S. M., Shah, A. U., & Sathish, H. A. (2011). Journal of Pharmaceutical Sciences, 100, 1306–1315.
Vedadi, M., Arrowsmith, C. H., Allali-Hassani, A., Senisterra, G., & Wasney, G. A. (2010). Journal of Structural Biology, 172, 107–119.
Acknowledgment
This work was supported by a National Research Foundation of Korea (NRF) grant funded by the Korean government (MEST) (No. 2013–031011) and by Business for Cooperative R&D between Industry, Academy, and Research Institute funded Korea Small and Medium Business Administration in 2013 (Grant No. C0123542).
Author information
Authors and Affiliations
Corresponding author
Additional information
Jin-Woo Jeong and Dong-Ho Seo contributed equally.
Rights and permissions
About this article
Cite this article
Jeong, JW., Seo, DH., Jung, JH. et al. Biosynthesis of Glucosyl Glycerol, a Compatible Solute, Using Intermolecular Transglycosylation Activity of Amylosucrase from Methylobacillus flagellatus KT. Appl Biochem Biotechnol 173, 904–917 (2014). https://doi.org/10.1007/s12010-014-0889-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12010-014-0889-z