Abstract
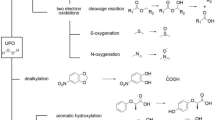
This study presents the first report of the purification and characterization of a monooxygenase enzyme from Pseudomonas putida A (ATCC 12633) that is responsible for the oxidation of physiologically relevant quaternary ammonium compounds, the tetradecyltrimethylammonium bromide. The degradation of tetradecyltrimethylammonium bromide by P. putida A (ATCC 12633) is initiated by N-dealkylation and catalysed by tetradecyltrimethylammonium monooxygenase (TTABMO), resulting in the formation of tetradecylalkanal and trimethylamine. Based on sequence analysis, the gene for TTABMO (ttbmo) corresponded to an ORF named PP2033 in the genome of P. putida KT2440. Mutation in ttabmo blocked the utilization of tetradecyltrimethylammonium bromide by Pseudomonas putida A (ATCC 12633) as carbon and nitrogen sources. The enzyme can be highly overexpressed in P. putida Δttabmo-T7 in active form and purified as a hexahistidine fusion protein. Like the native enzyme, the his-TTABMO was found to be a monomer with molecular mass of 40 kDa, the isoelectric point 7.3, that catalyses the breakdown of tetradecyltrimethylammonium bromide and utilized NADPH and FAD as cofactor. The biochemical properties and the analysis of the respective protein sequence revealed that TTABMO represents a typical flavoprotein monooxygenase, which is member of a flavoprotein family that is distinct from Baeyer–Villiger monooxygenases.




Similar content being viewed by others
References
Cross, J. (1994), in, Cationic surfactants, analytical and biological evaluation,surfactant science series (Singer, E.J., ed.), Marcel Dekker, New York, pp. 6–15.
Holah, J.T., Taylor, J.H., Dawson, D.J. & Hall, K.E. (2002) Symposium series (society for applied microbiology) pp. 111S–120S.
van Ginkel, C. G. (1991). Chemosphere, 23, 281–289.
Nishiyama, N., Toshima, Y., & Ikeda, Y. (1995). Chemosphere, 30, 593–603.
Oh, S., Tandukar, M., Pavlostathis, S. G., Chain, P. S., & Konstantinidis, K. T. (2013). Environmental Microbiology, 15, 2850–2864.
Takenaka, S., Tonoki, T., Taira, K., Murakami, S., & Aoki, K. (2007). Applied and Environmental Microbiology, 73, 1797–1802.
Liffourrena, A. S., López, F. G., Salvano, M. A., Domenech, C. E., & Lucchesi, G. I. (2008). Journal of Applied Microbiology, 104, 396–402.
Liffourrena, A. S., Salvano, M. A., & Lucchesi, G. I. (2010). Archives of Microbiology, 192, 471–476.
Lucchesi, G.I., Liffourrena, A.S. Boeris, P.S. & Salvano, M.A. (2010), in Current Research, Technology and education topics in applied microbiology and microbial biotechnology (Méndez-Vilas, A., ed.), Badajoz, Formatex, Spain, pp 1297–1303.
Lucchesi, G. I., Lisa, A. T., & Domenech, C. E. (1989). FEMS Microbiology Letter, 57, 335–338.
Hanahan, D. (1983). Journal of Molecular Biology, 166, 557–580.
Skipski, V.P. & Barclay, M. (1969), in: Methods in Enzymology, Vol 14 (Lowenstein, J.M., ed.), Academic Press, New York, pp 530–591.
Laemmli, U. K. (1970). Nature, 227, 680–685.
Sambrook, J., & Russell, D. R. (Eds.). (2001). Molecular cloning: a laboratory manual (2nd ed.). NY: Cold Spring Harbor Laboratory.
Kovach, M. E., Elzer, P. H., Hill, D. S., Robertson, G. T., Farris, M. A., Roop, R. M., & Peterson, K. M. (1995). Gene, 166, 175–176.
Kaniga, K., Delor, I., & Cornelis, G. R. (1991). Gene, 109, 137–141.
Lee, J. E., & Ahn, T. I. (2000). Research in Microbiology, 151, 605–618.
Liffourrena, A. S., Boeris, P. S., Salvano, M. A., & Lucchesi, G. I. (2009). Analytical Biochemistry, 384, 343–347.
Bradford, M. (1976). Analytical Biochemistry, 72, 248–254.
Ferreira, E., Giménez, R., Aguilera, L., Guzmán, K., Aguilar, J., Badia, J., & Baldomà, L. (2013). Research in Microbiology, 164, 145–154.
Suh, J. K., Poulsen, L. L., Ziegler, D. M., & Robertus, J. D. (1996). Archives of Biochemistry and Biophysics, 336, 268–274.
Nelson, K. E., Weinel, I. T., Paulsen, R. J., Dodson, H., Hilbert, V. A. O., Martins dos Santos, D. E., et al. (2002). Environmental Microbiology, 4, 799–808.
Schmidt, A., Schiesswohl, M., Völker, U., Hecker, M., & Schumann, W. (1992). Journal of Bacteriology, 174, 3993–3999.
Poulsen, L. L., & Ziegler, D. M. (1995). Chemico-Biological Interactions, 96, 57–73.
van Berkel, W. J. H., Kamerbeek, N. M., & Fraaije, M. W. (2006). Journal of Biotechnology, 124, 670–689.
Massey, V. J. (1994). Biological Chemistry, 269, 22459–22462.
Alfieri, A., Malito, E., Orru, R., Fraaije, M. W., & Mattevi, A. (2008). PNAS, 105, 86572–86577.
Dym, O., & Eisenberg, D. (2001). Protection Science, 10, 1712–1728.
Crozier-Reabe, K., & Moran, G. R. (2012). International Journal of Molecular Sciences, 13, 15601–15639.
Fraaije, M. W., Kamerbeek, N. M., van Berkel, W. J., & Janssen, D. B. (2002). FEBS Letters, 518, 43–47.
Riebel, A., Dudek, H. M., de Gonzalo, G., Stepniak, P., Rychlewski, L., & Fraaije, M. W. (2012). Applied Microbiology and Biotechnology, 95, 1479–1489.
Palleroni, N.J. (1992), in Pseudomonas, Molecular Biology and Biotechnology (E. Galli, S. Silver, and B. Witholt, eds.), American Society for Microbiology, Washington DC, USA, pp 105–115.
Hoang, T. T., Kutchma, A. J., Becher, A., & Schweizer, H. P. (2000). Plasmid, 43, 59–72.
Acknowledgments
GIL is Career Members of the Consejo Nacional de Investigaciones Científicas y Técnicas (CONICET). ASL is grateful for fellowships from CONICET. This work was supported by grants from CONICET, MinCyT Córdoba and SECYT–UNRC of Argentina.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Liffourrena, A.S., Lucchesi, G.I. Identification, Cloning and Biochemical Characterization of Pseudomonas putida A (ATCC 12633) Monooxygenase Enzyme necessary for the Metabolism of Tetradecyltrimethylammonium Bromide. Appl Biochem Biotechnol 173, 552–561 (2014). https://doi.org/10.1007/s12010-014-0862-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12010-014-0862-x