Abstract
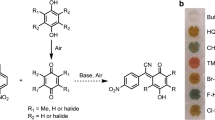
Cytochrome P450 BM-3 (A74G/F87V/L188Q) could catalyze indole to produce indigo. To further improve this capability, random mutagenesis was performed on the heme domain of P450 BM-3 (A74G/F87V/L188Q) with error-prone PCR. A single mutant V445A was selected out from the error-prone library and exhibited the highest specific activity toward indole among the mutants obtained. The kinetic parameters of V445A were also highly improved. Compared with the parent enzyme, the turnover rate (k cat) of V445A was increased by 7.5 times, while its K m value decreased by 9.2 %. Consequently, the catalytic efficiency (k cat/K m) of V445A was raised to 8.2 times than that of the parent enzyme. Moreover, alanine was confirmed as the best amino acid substitution by saturated mutagenesis in Val445 position. Three-dimensional structure analysis was also used to rationalize the effect on the enzyme properties of the mutation. This study showed that random mutagenesis was efficient to identify mutants with potential values in industry and increased our insight into P450 BM-3.





Similar content being viewed by others
References
Božič, M., Díaz-González, M., Tzanov, T., Guebitz, G. M., & Kokol, V. (2009). Enzyme and Microbial Technology, 45, 317–323.
Papanastasiou, M., Allen, N. S., McMahon, A., Naegel, L. C., Edge, M., & Protopappas, S. (2012). Dyes and Pigments, 92, 1192–1198.
Blackburn, R. S., Bechtold, T., & John, P. (2009). Coloration Technology, 125, 193–207.
Meksi, N., Ben, T. M., Kechida, M., & Mhenni, M. F. (2012). Journal of Cleaner Production, 24, 149–158.
Whitehouse, C. J., Bell, S. G., & Wong, L. L. (2012). Chemical Society Reviews, 41, 1218–1260.
Zhang, Z. G., Liu, Y., Guengerich, F. P., Matse, J. H., Chen, J., & Wu, Z. L. (2009). Journal of Biotechnology, 139, 12–18.
Huang, W., Johnston, W. A., Hayes, M. A., De Voss, J. J., & Gillam, E. M. (2007). Archives of Biochemistry and Biophysics, 467, 193–205.
Gillam, E. M., Notley, L. M., Cai, H. L., De Voss, J. J., & Guengerich, F. P. (2000). Biochemistry, 39, 13817–13824.
van Vugt-Lussenburg, B. M. A., Stjernschantz, E., Lastdrager, J., Oostenbrink, C., Vermeulen, N. P. E., & Commandeur, J. N. M. (2007). Journal of Medicinal Chemistry, 50, 455–461.
Li, Q. S., Schwaneberg, U., Fischer, P., & Schmid, R. D. (2000). Chemical-European Journal, 6, 1531–1536.
Narhi, L. O., & Fulco, A. J. (1986). Journal of Biological Chemistry, 261, 7160–7169.
Warman, A. J., Robinson, J. W., Luciakova, D., Lawrence, A. D., Marshall, K. R., Warren, M. J., et al. (2012). FEBS Journal, 279, 1675–1693.
Munro, A. W., Leys, D. G., McLean, K. J., Marshall, K. R., Ost, T. W. B., Daff, S., et al. (2002). Trends in Biochemical Sciences, 27, 250–257.
Li, H. M., Mei, L. H., Urlacher, V. B., & Schmid, R. D. (2008). Applied Biochemistry and Biotechnology, 144, 27–36.
Maurer, S. C., Schulze, H., Schmid, R. D., & Urlacher, V. B. (2003). Advanced Synthesis and Catalysis, 345, 802–810.
Omura, T., & Sato, R. (1964). Journal of Biological Chemistry, 239, 2370–2378.
Urlacher, V. B., Makhsumkhanov, A., & Schmid, R. D. (2006). Applied Microbiology and Biotechnology, 70, 53–59.
Choi, H. S., Kim, J. K., Cho, E. H., Kim, Y. C., Kim, J. I., & Kim, S. W. (2003). Biochemical and Biophysical Research Communications, 306, 930–936.
Sevrioukova, I. F., Li, H., Zhang, H., Peterson, J. A., & Poulos, T. L. (1999). Proceedings of the National Academy of Sciences, 96, 1863–1868.
Sven, H., Michael, S., Bert, S., & Svante, W. (1987). Journal of Medicinal Chemistry, 30, 1126–1135.
Oliver, C. F., Modi, S., Primrose, W. U., Lian, L. Y., & Roberts, G. C. (1997). Biochemistry Journal, 327, 537–544.
Hutzler, J. M., Wienkers, L. C., Wahlstrom, J. L., Carlson, T. J., & Tracy, T. S. (2003). Archives Biochemistry Biophysics, 410, 16–24.
Hu, S., Huang, J., Mei, L. H., Yu, Q., Yao, S. J., & Jin, Z. H. (2010). Journal of Molecular Catalysis B: Enzymatic, 67, 29–35.
Liao, Y., Zeng, M., Wu, Z. F., Chen, H., Wang, H. N., Wu, Q., et al. (2011). Applied Biochemistry Biotechnology, 166, 549–562.
Acknowledgments
This work was financially supported by the National Natural Science Foundation of China (30970638, 21176220, and 31240054) and Natural Science Foundation of Zhejiang Province (LZ13B060002).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Pengpai, Z., Sheng, H., Lehe, M. et al. Improving the Activity of Cytochrome P450 BM-3 Catalyzing Indole Hydroxylation by Directed Evolution. Appl Biochem Biotechnol 171, 93–103 (2013). https://doi.org/10.1007/s12010-013-0353-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12010-013-0353-5