Abstract
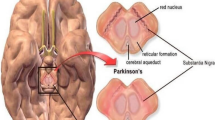
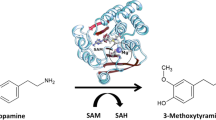
Catechol-O-methyltransferase (COMT) has a vital role for degradation of dopamine, a neurotransmitter, and this dopamine performs an important function in our mental and physical health. The scarcity of dopamine may lead to Parkinson’s disease, and inhibition of COMT can stop dopamine metabolism. Here, we have carried out genomics and proteomics analyses of COMT in order to facilitate new inhibitors of COMT. For genomics analyses, we performed codon composition investigation of COMT gene which shows A+T content which is 53.3 %. For proteomics analyses, conservation patterns and residues (highly conserved amino acids GLU64, LEU65, GLY66, CYS69, GLY70, ALA77, GLU90, THR99, SER119, ASP136, LEU140, ASP141, THR164, ASN170, VAL171, and ILE172), binding grooves, binding pockets, binding and conformation with substrate, evaluation of amino acid composition (15 % leucine rich), high scoring hydrophobic segments, high scoring transmembrane segments, tandem and periodic repeats, and disulfide bonds (three numbers), sequence logos (maximum stack height of 3 b and minimum stack height of <0.5 b) have been investigated for COMT protein. Furthermore, using COMT sequences of different species (class Mammalia, class Amphibia, and class Pisces), a phylogenetic tree has been constructed to examine the evolutionary relationship among different species.









Similar content being viewed by others
References
Dauer, W., & Przedborski, S. (2003). Neuron, 39, 889–909.
Hornykiewicz, O. (1970). Rivista di Patologia Nervosa e Mentale, 91, 281–286.
Gold, B. G., & Nutt, J. G. (2002). Current Opinion in Pharmacology, 2, 82–86.
Matsumoto, M., Weickert, C. S., Akil, M., Lipska, B. K., Hyde, T. M., Herman, M. M., Kleinman, J. E., & Weinberger, D. R. (2003). Neuroscience, 116, 127–137.
Brooks, D. J. (2001). Journal of Neural Transmission, 108, 1283–1298.
Wu, D. C., Jackson-Lewis, V., Vila, M., Tieu, K., Teismann, P., Vadseth, C., Choi, D. K., Ischiropoulos, H., & Przedborski, S. (2002). Journal of Neuroscience, 22, 1763–1771.
Olanov, C. W., Schapira, A. H., & Agid, Y. (2003). Annals of Neurology, 53, S1–S2.
Serra, P. A., Esposito, G., Enrico, P., Mura, M. A., Migheli, R., et al. (2000). British Journal of Pharmacology, 130, 937–945.
Werner, P., Mytilineou, C., Cohen, G., & Yahr, M. D. (1994). European Journal of Pharmacology, 263, 157–162.
Nutt, J. G. (1990). Neurology, 40, 340–345.
Sulzer, D., Bogulavsky, J., Larsen, K. E., Behr, G., Karatekin, E., et al. Proceedings of the National Academy of Sciences of the United States of America, 97, 1869–1874
Graham, D. G. (1978). Molecular Pharmacology, 14, 633–643.
Hald, A., & Lotharius, J. (2005). Experimental Neurology, 193, 279–290.
Zhou, C., Huang, Y., & Przedborski, S. (2008). Annals of the New York Academy of Sciences, 1147, 93–104.
Melnikova, V. I., Lavrenteva, A. V., Kudrin, V. S., Raevskii, K. S., & Ugryumov, M. V. (2005). Neuroscience and Behavioral Physiology, 35, 809–813.
Molinoff, P. B., & Axelrod, J. (1971). Annual Review of Biochemistry, 40, 465–500.
Mannisto, P. T., Ulmanen, I., Lundstrom, K., Taskinen, J., Tenhunen, J., Tilgman, C., & Kaakkola, S. (1992). Progress in Drug Research, 39, 291–348.
Westerink, B. H. C. (1985). Neurochemistry International, 7, 221–227.
Bortolato, M., Chen, K., & Shih, J. C. (2008). Advanced Drug Delivery Reviews, 60, 1527–1533.
Mannisto, P. T., & Kaakkola, S. (1999). Pharmacological Reviews, 51, 593–628.
Raevskii, K. S., Gainetdinov, R. R., Budygin, E. A., Mannisto, P., & Wightman, M. Neuroscience and Behavioral Physiology, 32, 183–188.
Okereke, C. S. (2002). Journal of Pharmacy and Pharmaceutical Sciences, 5, 146–161.
Rinne, U. K., Larsen, J. P., Siden, A., & Worm-Petersen, J. (1998). Neurology, 51, 1309–1314.
Rajput, A. H., Martin, W., Saint-Hillaire, M. H., Dorflinger, E., & Pedder, S. Neurology, 49, 1066–1071.
Lees, A. J. (2008). CNS Neuroscience and Therapeutics, 14, 83–93.
Muto, A., & Osawa, S. (1987). Proceedings of the National Academy of Sciences of the United States of America, 84, 166–169.
Yakovchuk, P., Protozanova, E., & Frank-Kamenetskii, M. D. (2006). Nucleic Acids Research, 34, 564–574.
Jermiin, L. S., Graur, D., Lowe, R. M., & Crozier, R. H. Journal of Molecular Evolution, 39, 160–173.
Hashimoto, T., Nakamura, Y., Nakamura, F., Shirakura, T., Adachi, J., Goto, N., Okamoto, K., & Hasegawa, M. (1994). Molecular and Biological Evolution, 11, 65–71.
Zeeberg, B. (2002). Genome Research, 12, 944–955.
Tao, X., & Dafu, D. (1998). FEBS Letters, 434, 93–96.
Sayers, E. W., Barrett, T., Benson, D. A., Bolton, E., Bryant, S. H., et al. (2011). Nucleic Acids Research, 39, D38–D51.
Leavitt, A., Chmelnitskym, I., Carmeli, Y., & Navon-Venezia, S. Antimicrobial Agents and Chemotherapy, 54, 4493–4496
Brendel, V., Bucher, P., Nourbakhsh, I., Blaisdell, B. E., & Karlin, S. (2002). Proceedings of the National Academy of Sciences of the United States of America, 89, 2002–2006.
Cheng, J., Randall, A. Z., Sweredoski, M. J., & Baldi, P. (2005). Nucleic Acids Research, 33, W72–W76.
Herraez, A. (2006). Biochemistry and Molecular Biology Education, 34, 255–261.
Hanson, R. M. (2010). Journal of Applied Crystallography, 43, 1250–1260.
DeLano, W. L. (2002). The PyMOL molecular graphics System.
Ashkenazy, H., Erez, E., Martz, E., Pupko, T., & Ben-Tal, N. (2010). Nucleic Acids Research, 38, W529–W533.
Glaser, F., Pupko, T., Paz, I., Bell, R. E., Bechor-Shental, D., Martz, E., & Ben-Tal, N. (2003). Bioinformatics, 19, 163–164.
Tseng, Y. Y., Chen, Z. J., & Li, W. H. (2010). Nucleic Acids Research, 38, D288–D295.
Smith, T. F., & Waterman, M. S. (1981). Journal of Molecular Biology, 147, 195–197.
Tseng, Y. Y., Dupree, C., Chen, Z. J., & Li, W. H. (2009). Nucleic Acids Research, 37, W384–W389.
Schneider, T. D. & Stephens, R. M. Nucleic Acids Res, 18, 6097–6100.
Crooks, G. E., Hon, G., Chandonia, J. M., & Brenner, S. E. (2004). Genome Research, 14, 1188–1190.
Dereeper, A., Guignon, V., Blanc, G., Audic, S., Buffet, S., Chevenet, F., Dufayard, J. F., Guindon, S., Lefort, V., Lescot, M., & Claverie, J. M. (2008). Nucleic Acids Research, 36, W465–W469.
Axelrod, J., Senoh, S., & Witkop, B. (1958). O-methylation of catechol amines in vivo. Journal of Biological Chemistry, 233, 697–701.
Axelrod, J., & Tomchick, R. (1958). Journal of Biological Chemistry, 233, 702–705.
Guldberg, H. C., & Marsden, C. A. (1975). Pharmacological Reviews, 27, 135–206.
Mannisto, P. T., & Kaakkola, S. (1989). Trends in Pharmacological Sciences, 10, 54–56.
Mannisto, P. T., & Kaakkola, S. (1990). Pharmacology and Toxicology, 66, 317–323.
Dingemanse, J. (1997). Drug Development Research, 42, 1–25.
Purushotham, D., Ganguly, C., & Chakraborty, C. (2011). Letters in Drug Design & Discovery, 8, 246–252.
Grossman, M. H., Emanuel, B. S., & Budarf, M. L. (1992). Genomics, 12, 822–825.
Winqvist, R., Lundstrom, K., Salminen, M., Laatikainen, M., & Ulmanen, I. (1992). Cytogenetics and Cell Genetics, 59, 253–257.
Salminen, M., Lundstrom, K., Tilgmann, C., Savolainen, R., Kalkkinen, N., & Ulmanen, I. (1990). Gene, 93, 241–247.
Lundstrom, K., Salminen, M., Jalanko, A., Savolainen, R., & Ulmanen, I. (1991). DNA and Cell Biology, 10, 181–189.
Rutherford, K., Le Trong, I., Stenkamp, R. E., & Parson, W. W. (2008). Journal of Molecular Biology, 380, 120–130.
Malherbe, P., Bertocci, B., Caspers, P., Zurcher, G., & Da Prada, M. (1992). Journal of Neurochemistry, 58, 1782–1789.
Vidgren, J., Ovaska, M., Tenhunen, J., Tilgmann, C., Lotta, T., & Mannisto, P. T. (1999). In X. Cheng & R. M. Blumenthal (Eds.), Structure and function of AdoMet-dependent methyltransferases (pp. 55–91). Singapore: World Scientific.
Vidgren, J., Svensson, L. A., & Liljas, A. (1994). Nature, 368, 354–358.
Vidgren, J., Tilgmann, C., Lundstrom, K., & Liljas, A. (1991). Proteins: Structure, Function, and Genetics, 11, 233–236.
Vidgren, J., & Ovaska, M. (1997). In P. Veerapandian (Ed.), Structure-based drug design (pp. 343–363). New York: Marcel Dekker, Inc.
Deo, N. (1974). Prentice Hall Series in Automatic Computation.
Mittal, R., Jain, B. N., & Patney, R. K. (1994). Microelectronics Reliability, 34, 1301–1310.
Chakraborty, C., Agoramoorthy, G., & Hsu, M. J. (2011). PLoS One, 6, e16580.
Author information
Authors and Affiliations
Corresponding author
Additional information
Chiranjib Chakraborty and Soumen Pal equally contributed to this work.
Rights and permissions
About this article
Cite this article
Chakraborty, C., Pal, S., Doss, C.G.P. et al. In Silico Analyses of COMT, an Important Signaling Cascade of Dopaminergic Neurotransmission Pathway, for Drug Development of Parkinson’s Disease. Appl Biochem Biotechnol 167, 845–860 (2012). https://doi.org/10.1007/s12010-012-9725-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12010-012-9725-5