Abstract
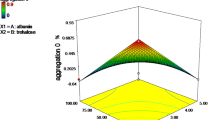
Proteins like immunoglobulin (IgGs) are prone to degradation by a variety of pathways. In this study, a stabilizing formulation for long-term storage of a panel of seven monoclonal IgGs was found using differential scanning calorimetry (DSC). In the chosen formulations, the IgGs were subjected to stress, accelerated and real-time storage, and analyzed by size exclusion chromatography to determine fragment and aggregate content, and fluorescence-activated cell sorting to measure immunoreactivity. All IgGs showed the greatest conformational stability near their isoelectric point which was enhanced by adding sorbitol, sucrose, glycine, and sodium chloride. Optimized formulations, found by DSC, containing 20 % sorbitol and 1 M glycine prevented IgG aggregation and fragmentation and conserved immunoreactivity against shear stress, multiple freeze–thaw cycles, accelerated storage at 37 °C, and 12 months storage at 4 and −20 °C. Relatively poor thermal stability of the antigen-binding fragment domain was shown to limit storage stability of IgGs. This study confirms the predictive power of DSC to find storage formulations which protect IgGs during stress and long-term storage from aggregation and degradation. Liquid formulations found in this study may have a broad utility for other IgGs.





Similar content being viewed by others
References
An, Z. (2010). Protein & Cell, 1, 319–330.
Wang, W., Nema, S., & Teagarden, D. (2010). International Journal of Pharmaceutics, 390, 89–99.
Eng, M., Ling, V., Briggs, J. A., Souza, K., Canova-Davis, E., Powell, M. F., & De Young, L. R. (1997). Analytical Chemistry, 69, 4184–4190.
Schrier, J. A., Kenley, R. A., Williams, R., Corcoran, R. J., Kim, Y., Northey, R. P., Jr., D’Augusta, D., & Huberty, M. (1993). Pharmaceutical Research, 10, 933–944.
Robert, F., Bierau, H., Rossi, M., Agugiaro, D., Soranzo, T., Broly, H., & Mitchell-Logean, C. (2009). Biotechnology and Bioengineering, 104, 1132–1141.
Vanerp, R., Adorf, M., Vansommeren, A. P. G., & Gribnau, T. C. J. (1991). Journal of Biotechnology, 20, 249–262.
Fradkin, A. H., Carpenter, J. F., & Randolph, T. W. (2009). Journal of Pharmaceutical Sciences, 98, 3247–3264.
van Beers, M. M. C., Jiskoot, W., & Schellekens, H. (2010). Journal of Interferon and Cytokine Research, 30, 767–775.
Lee, J. C., & Timasheff, S. N. (1981). Journal of Biological Chemistry, 256, 7193–7201.
Xie, G., & Timasheff, S. N. (1997). Protection Science, 6, 211–221.
Gekko, K., & Timasheff, S. N. (1981). Biochemistry, 4, 4667–4676.
Arakawa, T., & Timasheff, S. N. (1982). Biochemistry, 21, 6545–6552.
Arakawa, T., & Timasheff, S. N. (1983). Archives of Biochemistry and Biophysics, 224, 169–177.
Han, Y., Jin, B. S., Lee, S. B., Sohn, Y., Joung, J. W., & Lee, J. H. (2007). Archives of Pharmacal Research, 30, 1124–1131.
Ohtake, S., Kita, Y., & Arakawa, T. (2011). Advanced Drug Delivery Reviews, 63, 1053–1073.
Tiwari, A., & Bhat, R. (2006). Biophysical Chemistry, 124, 90–99.
Chen, B., Bautista, R., Yu, K., Zapata, G. A., Mulkerrin, M. G., & Chamow, S. M. (2003). Pharmaceutical Research, 20, 1952–1960.
Kerwin, B. A., Heller, M. C., Levin, S. H., & Randolph, T. W. (1998). Journal of Pharmaceutical Sciences, 87, 1062–1068.
Cramer, M., Frei, R., Sebald, A., Mazzoletti, P., & Maeder, W. (2009). Vox Sanguinis, 96, 219–225.
Kendrick, B. S., Chang, B. S., Arakawa, T., Peterson, B., Randolph, T. W., Manning, M. C., & Carpenter, J. F. (1997). PNAS, 94, 11917–11922.
Remmele, R. L., Nightlinger, N. S., Srinivasan, S., & Gombotz, W. R. (1998). Pharmaceutical Research, 15, 9.
Back, J. F., Oakenfull, D., & Smith, M. B. (1979). Biochemistry, 18, 5191–5196.
Lin, T. Y., & Timasheff, S. N. (1996). Protection Science, 5, 372–381.
Kreilgaard, L., Jone, L. S., Randolph, T. W., Frokjaer, S., Flink, J. M., Manning, M. C., & Carpenter, J. F. (1998). Journal of Pharmaceutical Sciences, 87, 1597–1603.
Wang, W., Wang, Y. J., & Wand, D. Q. (2008). International Journal of Pharmaceutics, 347, 31–38.
Ertel, K. D., & Carstensen, J. T. (1990). International Journal of Pharmaceutics, 61, 9–14.
Wang, W., Singh, S., Zeng, D. L., King, K., & Nema, S. (2007). Journal of Pharmaceutical Sciences, 96, 1–26.
Vermeer, A. W. P., & Norde, W. (2000). Biophysical Journal, 78, 394–404.
Garber, E., & Demarest, S. J. (2007). Biochemical and Biophysical Research Communications, 355, 751–757.
Ionescu, R. M., Vlasak, J., Price, C., & Kirchmeier, M. (2008). Journal of Pharmaceutical Sciences, 97, 1414–1426.
Chen, B. L., & Arakawa, T. (1996). Journal of Pharmaceutical Sciences, 85, 419–426.
Ahrer, K., Buchacher, A., Iberer, G., & Jungbauer, A. (2006). Journal of Biochemical and Biophysical Methods, 66, 73–86.
Szenczi, Á., Kardos, J., Medgyesi, G. A., & Závodszky, P. (2006). Biologicals, 34, 5–14.
Choo, A. B., Tan, H. L., Ang, S. N., Fong, W. J., Chin, A., Lo, J., Zheng, L., Hentze, H., Philp, R. J., Oh, S. K., & Yap, M. (2008). Stem Cells, 26, 1454–1463.
Ho, S. C. L., Bardor, M., Feng, H., Mariati, Tong, Y. W., Song, Z., Yap, M. G. S., & Yang, Y. (2012). Journal of Biotechnology, 157, 130-139.
Choo, A., Padmanabhan, J., Chin, A., Fong, W. J., & Oh, S. K. W. (2006). Journal of Biotechnology, 122, 130–141.
Eriksson, L., Johansson, E., Kettaneh-Wold, N., Wikstroem, C., Wold, S. (2008). Umetrics Academy.
He, F., Hogan, S., Latypov, R. F., Narhi, L. O., & Razinkov, V. I. (2010). Journal of Pharmaceutical Sciences, 99, 1707–1720.
Bhambhani, A., Kissmann, J. M., Joshi, S. B., Volkin, D. B., Kashi, R. S., & Middaugh, C. R. (2011). Journal of Pharmaceutical Sciences, 101, 1120–1135.
Zheng, J. Y., & Janis, L. J. (2006). International Journal of Pharmaceutics, 308, 46–51.
Li, S., Schöneich, C., & Borchardt, R. T. (1995). Biotechnology and Bioengineering, 48, 490–500.
Grinberg, V. Y., Burova, T. V., Grinberg, N. V., Shcherbakova, T. A., Guranda, D. T., Chilov, G. G., & Svedas, V. K. (2008). Biochimica et Biophysica Acta, 1784, 736–746.
Pace, C. N., Laurents, D. V., & Thomson, J. A. (1990). Biochemistry, 29, 2564–2572.
Pace, C. N., Grimsley, G. R., & Scholtz, J. M. (2009). Journal of Biological Chemistry, 284, 13285–13289.
González, M., Murature, D. A., & Fidelio, G. D. (1995). Vox Sanguinis, 68, 1–4.
Arakawa, T., Ejima, D., Tsumoto, K., Obeyama, N., Tanaka, Y., Kita, Y., & Timasheff, S. N. (2007). Biophysical Chemistry, 127, 1–8.
Welfle, K., Misselwitz, R., Hausdorf, G., Höhne, W., & Welfle, H. (1999). Biochimica et Biophysica Acta, 1431, 120–131.
Buchner, J., Renner, M., Lilie, H., Hinz, H. J., Jaenicke, R., Kiefhabel, T., & Rudolph, R. (1991). Biochemistry, 30, 6922–6929.
Acknowledgments
The authors would like to gratefully thank Yih Yean Lee, Hui Ching Hia, and Ming Wei Wu for their help with producing and purifying the IgGs. Furthermore, the authors would like to thank Christopher Tan for his help with analytical SEC and Vanessa Ding for providing hESC for experiments. This work was supported by the Biomedical Research Council of A*STAR (Agency for Science, Technology and Research), Singapore.
Author information
Authors and Affiliations
Corresponding author
Electronic Supplementary Material
Below is the link to the electronic supplementary material.
ESM 1
DOC 1,123 kb
Rights and permissions
About this article
Cite this article
Mueller, M., Loh, M.Q.T., Tee, D.H.Y. et al. Liquid Formulations for Long-Term Storage of Monoclonal IgGs. Appl Biochem Biotechnol 169, 1431–1448 (2013). https://doi.org/10.1007/s12010-012-0084-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12010-012-0084-z