Abstract
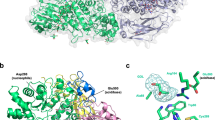
β-d-Xylosidase/α-l-arabinofuranosidase from Selenomonas ruminantium is the most active enzyme known for catalyzing hydrolysis of 1,4-β-d-xylooligosaccharides to d-xylose. Catalysis and inhibitor binding by the GH43 β-xylosidase are governed by the protonation states of catalytic base (D14, pK a 5.0) and catalytic acid (E186, pK a 7.2). Biphasic inhibition by triethanolamine of E186A preparations reveals minor contamination by wild-type-like enzyme, the contaminant likely originating from translational misreading. Titration of E186A preparations with triethanolamine allows resolution of binding and kinetic parameters of the E186A mutant from those of the contaminant. The E186A mutation abolishes the pK a assigned to E186; mutant enzyme binds only the neutral aminoalcohol \( \left( {{\text{pH}} - {\text{independent}}\;K_{\text{i}}^{\text{triethanolamine}} = 19\,{\text{mM}}} \right) \), whereas wild-type enzyme binds only the cationic aminoalcohol \( \left( {{\text{pH}} - {\text{independent}}\;K_{\text{i}}^{\text{triethanolamine}} = 0.065\,{\text{mM}}} \right) \). At pH 7.0 and 25°C, relative kinetic parameter, \( k_{\text{cat}}^{\text{4NPX}}/k_{\text{cat}}^{\text{4NPA}} \), for substrates 4-nitrophenyl-β-d-xylopyranoside (4NPX) and 4-nitrophenyl-α-l-arabinofuranoside (4NPA) of E186A is 100-fold that of wild-type enzyme, consistent with the view that, on the enzyme, protonation is of greater importance to the transition state of 4NPA whereas ring deformation dominates the transition state of 4NPX.




Similar content being viewed by others
References
Henrissat, B. (1991). Biochemical Journal, 280, 309–316.
Henrissat, B., & Davies, G. J. (1997). Current Opinion in Structural Biology, 7, 637–644.
Davies, G. J., & Sinnott, M. L. (2008). The Biochemical Journal. www.biochemj.org. doi:10.1042/BJ20080382.
Jordan, D. B., Li, X.-L., Dunlap, C. A., Whitehead, T. R., & Cotta, M. A. (2007). Applied Biochemistry and Biotechnology, 141, 51–76.
Jordan, D. B. (2008). Applied Biochemistry and Biotechnology, 146, 137–149.
Jordan, D. B., Li, X.-L., Dunlap, C. A., Whitehead, T. R., & Cotta, M. A. (2007). Applied Biochemistry and Biotechnology, 137–140, 93–104.
Jordan, D. B., Dien, B. S., Li, X.-L., & Cotta, M. A. (2006). In Renewable Energy 2006 Committee (Ed.), Proceedings of Renewable Energy 2006 (pp. 1036–1041). Japan: Makuhari Messe.
Saha, B. C. (2003). Journal of Industrial Microbiology & Biotechnology, 30, 279–291.
Gray, K. A., Zhao, L., & Emptage, M. (2006). Current Opinion in Chemical Biology, 10, 141–146.
Shallom, D., & Shoham, Y. (2003). Current Opinion in Microbiology, 6, 219–228.
Jordan, D. B., & Li, X.-L. (2007). Biochimica et Biophysica Acta, 1774, 1192–1198.
Jordan, D. B., & Braker, J. D. (2007). Archives of Biochemistry and Biophysics, 465, 231–246.
Whitehead, T. R., & Cotta, M. A. (2001). Current Microbiology, 43, 293–298.
Brunzelle, J. S., Jordan, D. B., McCaslin, D. R., Olczak, A., & Wawrzak, Z. (2008). Archives of Biochemistry and Biophysics, 474, 157–166.
Jordan, D. B., & Braker, J. D. (2009). Applied Biochemistry and Biotechnology, 155, 330–346.
Jordan, D. B., Mertens, J. A., & Braker, J. D. (2009). Biochimica et Biophysica Acta, 1794, 144–158.
Durette, P. L., & Horton, D. (1971). Advances in Carbohydrate Chemistry and Biochemistry, 26, 49–125.
Kezdy, F. J., & Bender, M. L. (1962). Biochemistry, 1, 1097–1106.
Gill, S. C., & von Hippel, P. H. (1989). Analytical Biochemistry, 182, 319–326.
Leatherbarrow, R. J. (2001). Grafit Version 5. Horley: Erithacus Software Ltd.
Zheng, Y.-J., Basarab, G. S., & Jordan, D. B. (2002). Biochemistry, 41, 820–826.
Sulzenbacher, G., Driguez, H., Henrissat, B., Scülein, M., & Davies, G. J. (1996). Biochemistry, 35, 15280–15287.
Tews, I., Perrakis, A., Oppenheim, A., Dauter, Z., Wilson, K. S., & Vorgias, C. E. (1996). Nature Structural & Molecular Biology, 3, 638–648.
Davies, G. J., Mackenzie, L., Varrot, A., Dauter, M., Brzozowski, A. M., Scülein, M., et al. (1998). Biochemistry, 37, 11707–11713.
Notenboom, V., Birsan, C., Nitz, M., Rose, D. R., Warren, R. A., & Withers, S. G. (1998). Nature Structural & Molecular Biology, 5, 812–818.
Sidhu, G., Withers, S. G., Nguyen, N. T., McIntosh, L. P., Ziser, L., & Brayer, G. D. (1999). Biochemistry, 38, 5346–5354.
Vocadlo, D. J., Davies, G. J., Laine, R., & Withers, S. G. (2001). Nature, 412, 835–838.
Ducros, V. M.-A., Zechel, D. L., Murshudov, G. N., Gilbert, H. J., Szabó, L., Stoll, D., et al. (2002). Angewandte Chemie. International Edition, 41, 2824–2827.
Larsson, A. M., Bergfors, T., Dultz, E., Irwin, D. C., Roos, A., Driguez, H., et al. (2005). Biochemistry, 44, 12915–12922.
Biarnés, X., Nieto, J., Planas, A., & Rovira, C. (2006). Journal of Biological Chemistry, 281, 1432–1441.
Author information
Authors and Affiliations
Corresponding author
Additional information
The mention of firm names or trade products does not imply that they are endorsed or recommended by the US Department of Agriculture over other firms or similar products not mentioned.
Rights and permissions
About this article
Cite this article
Jordan, D.B., Braker, J.D. β-d-Xylosidase from Selenomonas ruminantium: Role of Glutamate 186 in Catalysis Revealed by Site-Directed Mutagenesis, Alternate Substrates, and Active-Site Inhibitor. Appl Biochem Biotechnol 161, 395–410 (2010). https://doi.org/10.1007/s12010-009-8874-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12010-009-8874-7