Abstract
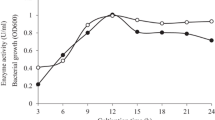
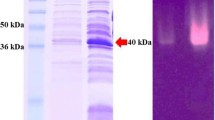
A DNA fragment of 2,042 bp containing a novel β-mannanase gene, man5A, was identified from the genome of the mannan-degrading bacterium Bacillus circulans CGMCC1554. The open reading frame of man5A comprised 978 bp encoding a protein of 326 amino acids with a predicted molecular weight of 32 kDa. The amino acid sequence of the encoded mannanase, MAN5A, showed the highest identity (78.5%) to β-mannanases belonging to glycosyl hydrolases family 5. The gene man5A was efficiently expressed in Escherichia coli and Pichia pastoris with the highest activity of 541 U/ml in a 3-L fermenter. Recombinant MAN5A purified from E. coli had a high specific activity of 4,839 U/mg, which is much higher than that of enzymes that showed high sequence identity. The enzyme showed maximum activity at pH 7.6 and 60 °C and resistance to trypsin. After hydrolysis of LBG, oligomannosides accounted for 76% of the hydrolysis products. All these properties collectively make MAN5A a better candidate than current mannanases for use in the food and feed industry.




Similar content being viewed by others
References
Suurmakki, A., Heijnesson, A., Buchert, J., Tenkanan, M., Viikari, L., & Westermark, V. (1996). Journal of Pulp and Paper Science, 22, 78–83.
Kuhad, R. C., Singh, A., & Eriksson, K. E. (1997). Advances in Biochemical Engineering/Biotechnology, 57, 45–125. doi:10.1007/BFb0102072.
Puls, J. (1997). Macromolecular Symposia, 120, 183–196.
Lee, J. T., Bailey, C., & Cartwright, A. L. (2003). Poultry Science, 82, 1925–1931.
Sachslehner, A., Foidl, G., Foidl, N., & Gubitz, G. J. (2000). Biotechnology, 80, 127–134. doi:10.1016/S0168-1656(00)00253-4.
Buchert, J., Salminen, J., Sika-aho, M., Ranua, M., & Viikari, L. (1993). Holzforschung, 47, 473–478.
Suurnakki, A., Tenkanen, M., Buchert, J., & Viikari, L. (1997). Advances in Biochemical Engineering/Biotechnology, 57, 261–287. doi:10.1007/BFb0102077.
Cann, I. K., Kocherginskaya, S., King, M. R., White, B. A., & Mackie, R. I. (1999). Journal of Bacteriology, 181, 1643–1651.
Luthi, E., Jasmat, N. B., Grayling, R. A., Love, D. R., & Bergquist, P. L. (1991). Applied and Environmental Microbiology, 57, 694–700.
Yoon, K. H., & Lim, B. L. (2007). Journal of Microbiology and Biotechnology, 17, 1688–1694.
Henrik, S., Matti, S. A., Mafja, T., & Lusa, V. (1995). Applied and Environmental Microbiology, 61, 1090–1097.
Bewley, D. J., Burton, A. R., Morohashi, Y., & Fincher, B. G. (1997). Planta, 203, 454–459. doi:10.1007/s004250050214.
Xu, B., Sellos, D., & Janson, J. C. (2002). European Journal of Biochemistry, 269, 1753–1760. doi:10.1046/j.1432-1327.2002.02824.x.
Henrissat, B. (1991). The Biochemical Journal, 280, 309–316.
Henrissat, B., & Bairoch, A. (1993). Journal of Biochemistry, 293, 781–788.
Stoll, D., LeNours, J., Anderson, L., Stalbrand, H., & LoLeggio, L. (2005). Biochemistry, 44, 12700–12708. doi:10.1021/bi050779v.
Dhawan, S., & Kaur, J. (2007). Critical Reviews in Biotechnology, 27, 197–216. doi:10.1080/07388550701775919.
Nelson, N. (1944). The Journal of Biological Chemistry, 153, 375–380.
Li, Y., Yang, P., Meng, K., Wang, Y., Luo, H., Wu, N., et al. (2008). Journal of Microbiology and Biotechnology, 18, 160–166.
Sumner, J. B., & Somers, G. F. (1949). Laboratory expression in biological chemistry. New York: Academic.
Wang, S. Y., Wu, S. J., Thottappilly, G., Locy, R., & Singh, N. (2001). Journal of Bioscience and Bioengineering, 92, 59–66. doi:10.1263/jbb.92.59.
Yang, P., Shi, P., Wang, Y., Bai, Y., Meng, K., Luo, H., et al. (2007). Journal of Microbiology and Biotechnology, 17, 58–66.
Yoshida, S., Sako, Y., & Uchida, A. (1998). Bioscience, Biotechnology, and Biochemistry, 62, 514–520. doi:10.1271/bbb.62.514.
Hakamada, Y., Kobayashi, T., Hitomi, J., Kawai, S., & Ito, S. (1994). Journal of Fermentation and Bioengineering, 78, 105–108. doi:10.1016/0922-338X(94)90188-0.
Baird, D. S., Hefford, M. A., Johnson, D. A., Sung, L. W., Yaguchi, M., & Seligy, V. L. (1990). Biochemical and Biophysical Research Communications, 169, 1035–1039. doi:10.1016/0006-291X(90)91998-8.
Belaich, A., Fierobe, H. P., Baty, D., Busetta, B., Bagnara-Tardif, C., Gaudin, C., et al. (1992). Journal of Bacteriology, 174, 4677–4682.
Guiseppi, A., Cami, B., Aymeric, J. L., Ball, G., & Creuzet, N. (1998). Molecular Microbiology, 2, 159–164. doi:10.1111/j.1365-2958.1988.tb00017.x.
Macarron, R., Beeumen, J. V., Henrissat, B., Mata, I., & Claeyssens, M. (1993). FEBS Letters, 316, 137–140. doi:10.1016/0014-5793(93)81202-B.
Wang, Q., Tull, D., Meinke, A., Gilkes, R. N., Warren, J. A. R., Aebersold, R., et al. (1993). The Journal of Biological Chemistry, 268, 14096–14102.
Hilge, M., Gloor, S. M., Rypniewski, W., Sauer, O., Heightman, T. D., Zimmermann, W., et al. (1998). Structure (London, England), 6, 1433–1444. doi:10.1016/S0969-2126(98)00142-7.
Yan, X., An, X., Gui, L., & Liang, D. (2008). Journal of Molecular Biology, 379, 535–544. doi:10.1016/j.jmb.2008.03.068.
Zakaria, M. M., Yamamoto, S., & Yagi, T. (1998). FEMS Microbiology Letters, 158, 25–31.
Jiang, Z., Wei, Y., Li, D., Li, L., Chai, P., & Kusakabe, I. (2006). Carbohydrate Polymers, 66, 88–96. doi:10.1016/j.carbpol.2006.02.030.
Macauley-Patrick, S., Fazenda, M. L., McNeil, B., & Harvey, L. M. (2005). Yeast (Chichester, England), 22, 249–270. doi:10.1002/yea.1208.
Chen, X., Cao, Y., Ding, Y., Lu, W., & Li, D. (2007). Journal of Biotechnology, 128, 452–461. doi:10.1016/j.jbiotec.2006.11.003.
Tang, C. M., Waterman, L. D., Smith, M. H., & Thurston, C. F. (2001). Applied and Environmental Microbiology, 67, 2298–2303. doi:10.1128/AEM.67.5.2298-2303.2001.
Wong, K. K. Y., & Saddler, J. N. (1993). In M. P. Coughlan & G. P. Hazlewood (Eds.), Hemicellulose and hemicellulases (pp. 127–143). London: Portland.
Acknowledgments
This work was supported by the Chinese National High Technology Research and Development Program (863 Program, Grant No. 2007AA100601) and the Chinese Agricultural Microorganism Collection and Share Program (No. 2005DKA21201).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Yang, P., Li, Y., Wang, Y. et al. A Novel β-mannanase with High Specific Activity from Bacillus circulans CGMCC1554: Gene Cloning, Expression and Enzymatic Characterization. Appl Biochem Biotechnol 159, 85–94 (2009). https://doi.org/10.1007/s12010-008-8364-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12010-008-8364-3